Neuroprotective Effect of Chlorogenic Acids against Alzheimer’s Disease
Anggreani E*, Lee CY
Department of Food Science, Cornell University, NY, USA.
*Corresponding Author
Elfi Anggreani,
Food Science M.P.S’16, Stocking Hall,
Department of Food Science, Cornell University, Ithaca, 14853, USA.
Tel: 607-379-7473
E-mail: ea372@cornell.edu
Received: February 05, 2017; Accepted: March 06, 2017; Published: March 09, 2017
Citation: Anggreani E, Lee CY (2017) Neuroprotective Effect of Chlorogenic Acids against Alzheimer’s Disease. Int J Food Sci Nutr Diet. 6(1), 330-337. doi: http://dx.doi.org/10.19070/2326-3350-1700059
Copyright: Anggreani E© 2017. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
With increasing trend of aging population, neurodegenerative diseases, including Alzheimer’s Disease (AD) have become more prevalent in today’s society. Coffee is a popular beverage that has been linked to health benefits, including the potential to mediate AD. Coffee contains different concentrations and isomers of chlorogenic acids (CGAs), which represent a group of phenolic compounds abundant in human diet that have garnered interest due to their antioxidant activity in relation to neurological benefits. Evidence supporting the neuroprotective effects of CGAs has been observed in epidemiological studies, as well as in vitro and in vivo studies. CGAs are capable of mediating oxidative stress and attenuating cell apoptosis due to different oxidative stressors and free radicals by modulating the accumulation of reactive oxygen species and by regulating the expression of key proteins and enzymes involved in cell apoptosis. As CGAs are highly bioavailable in humans compared to other bioactive polyphenols, these compounds have the potential to exert positive effects in alleviating AD.
2.Introduction
3.CGAs Composition in Coffee and the Effect of Processing on Total Cgas Content
4.Bioavailability of Cgas in Humans
4.1 Proposed metabolic pathway of CGA and specific absorption sites for hydroxycinnamate derivatives in human digestive system
5.Neuroprotective Mechanisms of CGAs on Key Components of Alzheimer’s Disease
5.1 Antioxidant Capacity of CGAs (Chemical Assays)
5.2 Antioxidant Capacity of CGAs (Cell-Based Assays)
5.3 Antioxidant Capacity of CGAs (Animal Studies)
5.4 Antioxidant Capacity of CGAs (Human Studies)
6.Conclusions
7.References
Keywords
Alzheimer’s Disease; Chlorogenic Acids; Coffee; Antioxidant Activity.
Introduction
As population aging becomes a trend in many regions of the world, age-related neurodegenerative diseases, such as Alzheimer’s Disease (AD), have become more prevalent in today’s society. AD is the 5th leading cause of death among elderly population age 65 or older in the United States with projected number of 13.8 million cases by 2050 [1]. Contributing factors to the development of AD are believed to be the accumulation of abnormally folded amyloid beta (Aβ) plaques, consisting mainly of 40- and 42-mer Aβ protein, and the accumulation of tau protein tangles [2,3]. These accumulations lead to synaptic degeneration and neuronal damage, which are attributed to cognitive and memory impairments in AD patients. AD has also been associated with an increase in pro-inflammatory cytokines in the blood and cerebrospinal fluid, which is an inflammatory response likely as a repair mechanism that is triggered by neuronal damage [2].
Epidemiological studies have shown that coffee consumption is linked to risk reduction of brain diseases, including AD. Although caffeine content in coffee is only 1%, many studies focus on evaluating the effect of caffeine on AD prevention, which is likely due to psychoactive stimulating properties of caffeine and its ability to cross blood-brain barrier [4]. Compared to caffeine, coffee contains much higher amount of chlorogenic acids (CGAs), which is about 7-9% dry weight basis. Although some studies mention possible synergistic effect of caffeine, CGAs, and other bioactive phytochemicals in coffee to mitigate AG, the importance of CGAs may be overlooked. Moreover, studies on CGAs are necessary to further understand the neurological benefits of caffeinated coffee and decaffeinated coffee.
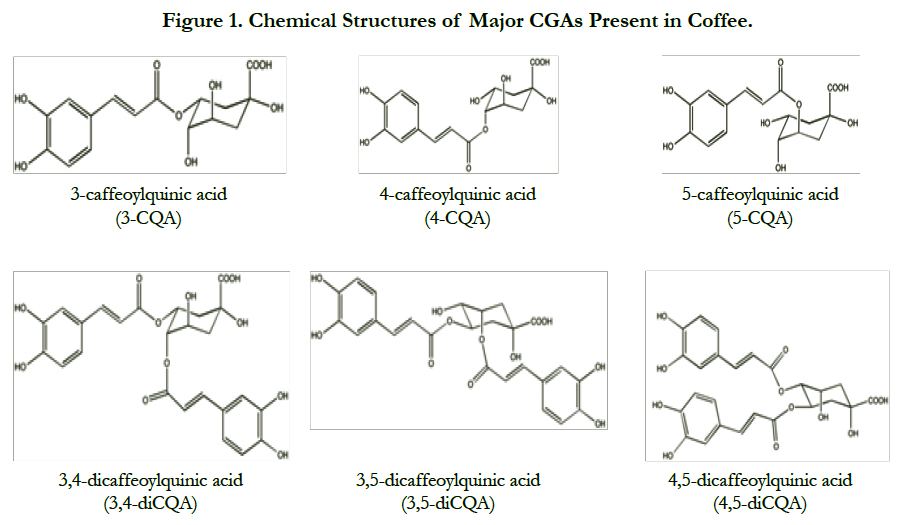
CGAs are a group of polyphenols abundant in human diet, which can be obtained by consuming fruits, vegetables, medicinal herbs, and coffee. These polyphenolic acids are esters of quinic and hydroxycinnamic acids, such as caffeic, ferulic, and p-coumaric acids. CGAs are the major phenolics in coffee, which are present in the forms of caffeoylquinic acids (CQA), feruloylquinic acids (FQA), di-caffeoylquinic acids (diCQA) and caffeoylquinic acid lactones (CQAL) isomers [5] (Figure 1).
CGAs have been the subject of scientific research due to their antioxidant capacity to scavenge free radicals, which is attributed to double bonds and hydroxyl groups in their structure. Accumulation of reactive oxygen species (ROS) in the brain plays a role in the early stage of AD. Moreover, oxidative stress has been regarded as a critical component in the pathogenesis of AD.
This review will discuss the potential of CGAs in mitigating AD by examining evidence pertaining to their antioxidant and anti-inflammatory properties. A brief discussion on CGAs composition in coffee and the effect of processing will be provided to assess types of coffee that provide better source of CGAs. Moreover, bioavailability of CGAs will also be reviewed in order to provide an understanding on whether CGAs are well absorbed in humans and which specific compounds can reach human tissues to exert positive benefits.
CGAs Composition in Coffee and the Effect of Processing on Total Cgas Content
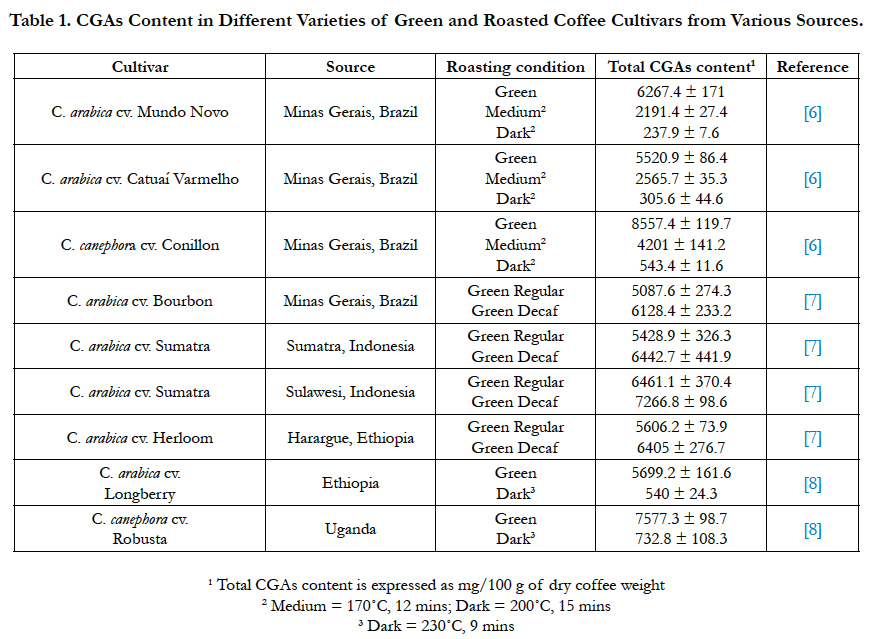
CGAs content in green coffee beans is the highest compared to other food sources, comprising of up to 14% in dry weight basis (dwb) with 45 identified CGA compounds [6]. In green coffee, CGAs are present in the form of CQA, diCQA, FQA, p-coumaroylquinic acids (p-CoQA) and caffeoylferuoylquinic acids (CFQA). Other compounds, such as diferuoylquinic acids (diFQA), di-p-coumaroylquinic acids (di-p-CoQA), and dimethoxycinnamoylquinic acids, are also present although they only constitute less than 1% of total CGAs.
CGAs content in coffee is dependent on the source of coffee beans, as well as the degree of roasting and brewing. Total CGA content in Brazilian Coffea arabica green coffee samples range from 5.5-6.3 g/100 g dwb, while Brazilian Coffea canephora sample has 8.6 g total CGAs/100 g dwb [6]. Many studies have reported that C. canephora has higher CGAs content than C. arabica. In arabica samples from Brazil, Indonesia, and Ethiopia, total CGAs prior to roasting range from 5.1-6.4 g/100 g dwb [7]. Total CGA content in Robusta green coffee was reported to be 28% higher than Arabica variety [8]. Although CGA isomers composition varies depending on coffee variety and source, the main CGA class in all samples was CQA, with 5-CQA being the predominant form. 35% of total CGAs in coffee is in the form of 5-CQA. In total, CQA and diCQA isomers make up 92-95% of total CGAs in coffee [9].
In the roasting process, some CGAs are degraded into CGA lactones and other derivatives through pyrolysis. The main CGA lactones present in roasted coffee is cinnamoyl-1,5-γ-quinolactones. The extent of CGAs thermal degradation during roasting is dependent on roasting conditions (time-temperature combination). CGAs loss might reach up to 95% in dark roasted coffee, which is due to several mechanisms occurring during roasting process including isomerization, epimerization, lactonization and degradation [6].
At fast roasting time, the antioxidant capacity of C. arabica from Costa Rica was found to be higher than green beans. This is likely due to the generation of compounds with high antioxidant capacity during roasting [10]. Similarly, analyses comparing the antioxidant capacity of unheated and heated 5-CQA show that heated 5-CQA exhibits higher antioxidant capacity, which is attributed to the generation of 2-methoxy-4-vinylphenol, 4-vinylphenol, and pyrocatechol [11]. However, when roasting temperature reached 200°C, antioxidant capacity is significantly lowered as more CGAs are thermally degraded. Moon et al., [12] reported that 45-54% of CGAs are lost during light roast, while in higher roasts, up to 99% of CGAs can be lost. Aside from roasting, different brewing methods were also found to affect total CGAs content (Table 1).
Table 1. CGAs Content in Different Varieties of Green and Roasted Coffee Cultivars from Various Sources.
Bioavailability of CGAs in Humans
Bioavailability is important in evaluating potential benefits of CGAs on human health. CGAs must be absorbed and be able to pass through blood-brain barrier and understanding whether intact CGAs or their metabolites pass through this barrier is crucial. Understanding the metabolic fate of CGA compounds could increase credibility to their proposed neurological benefits, especially determining specific compounds that impose the benefits.
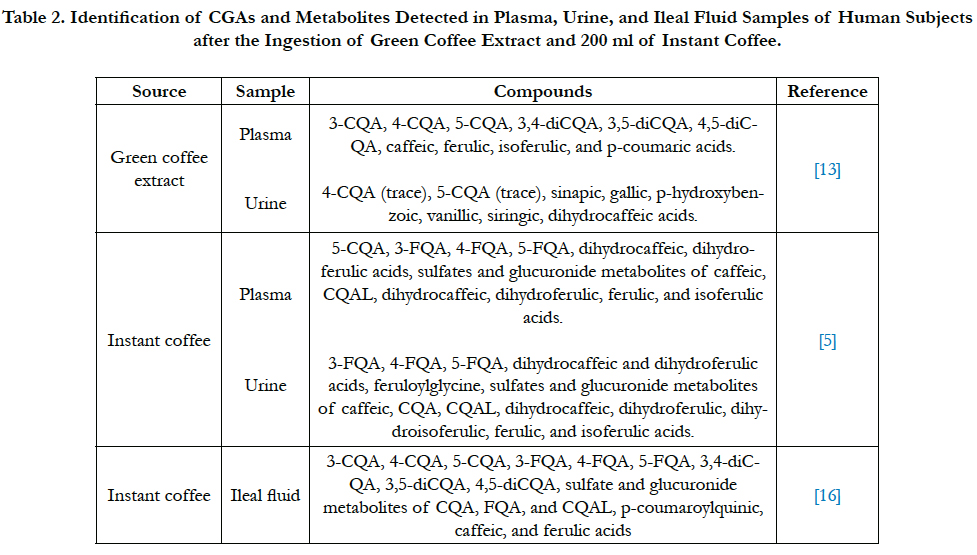
Identification and quantification of CGA and their metabolites are prerequisites to assess whether these compounds are bioavailable in humans. Plasma sample analysis of human subjects after consuming green coffee extract showed approximately 33% recovery of CGAs and their metabolites [13]. After consumption, caffeic, ferulic, isoferulic, and p-coumaric acids were recovered in plasma although these compounds were not present in the extract. Besides these compounds, dihydroferulic acid, dihydrocaffeic acid, as well as sulfated and glucuronide metabolites of several CGA compounds were detected in human plasma after instant coffee consumption [5]. There are discrepancies where after green coffee extract consumption, no feruloylquinic acid (FQA) was detected. However, FQA was detected in plasma after instant coffee consumption. Moreover, Stalmach et al., [5] did not indicate the presence of p-coumaric acid. In total, 12 hydroxycinnamate derivatives were quantified in the plasma samples [5]. In terms of pharmacokinetic profile of the compounds identified in plasma, there was a substantial variation between the subjects. Maximum concentration (Cmax) of total CGAs ranged from 1.2 to 39.7 μmol/L [13]. In a dose-response study using instant coffee, the absorption pattern shows that plasma CGA metabolites appear proportionally to ingested dose at normal range of coffee consumption [14].
In urine sample analysis at baseline, phenolic compounds were identified in most subjects, including 5-CQA, sinapic, gallic, phydroxybenzoic, and dihydrocaffeic acids. Excluding CQA, these other compounds represented approximately 85% of total phenolics quantified from the urine sample. However, compounds identified in the urinary excretion of subjects also substantially varied. In the urine samples, 17 hydroxycinnamate derivatives were quantified, which was approximately 29% of total CGA intake [5]. These include CQAs, FQAs, caffeic, dihydrocaffeic, ferulic, dihydroferulic, dihydro (iso)ferulic, isoferulic, feruloylglycine along with sulfates and glucoronides of several compounds. No protocatechuic, dihydroferulic, benzoic, and hippuric acids was present although previous study had identified this in urinary excretion after CGA consumption [13]. The main metabolite detected in urine after CGAs ingestion is caffeic acid [15].
In a follow-up study done by Stalmach et al., [16], 59% of CQAs was recovered in ileal fluid of ileostomy subjects. They also found high recovery of several other compounds, including FQAs (77%), CQAL metabolites (56%), p-coumaroylquinic acids (46%) and diCQA (46%). In total, 71% of CGA and their metabolites were recovered in ileal fluid However, only 8% CGAs and metabolites were recovered compared to 29% found in the previous study with healthy subjects.
Comparing the recovery of CGAs and their metabolites in plasma, urine, and ileal fluid samples, the recovery is high in plasma and ileal fluid and low in urine samples. Low urine recoveries indicate that it is not the preferred excretion route for CGA and their metabolites. Based on pharmacokinetic result of several studies, absorption of CGAs and their metabolites occur mainly in the colon. One third of CGA is potentially absorbed from the small intestine and stomach, while the rest reaches the large intestine to be metabolized by colonic microflora. The apparent bioavailability of CGAs from green coffee extract and CGA supplement are 33.3 ± 23% and 33 ± 17%, respectively [13, 17] (Table 2).
Table 2. Identification of CGAs and Metabolites Detected in Plasma, Urine, and Ileal Fluid Samples of Human Subjects after the Ingestion of Green Coffee Extract and 200 ml of Instant Coffee.
Many compounds that were not originally present in the coffee samples were recovered from plasma, urine, and ileal fluids. This suggests that combination of CGA hydrolysis, as well as complex metabolism might be occurring in human digestive system. Also preferential absorption of several compounds such as 5-CQA compared to 4-CQA, suggested that 4-CQA was metabolized/ uptake faster by organs, such as liver and adipose tissue [13]. Similarly, trace amount of parent compounds, such as CQA and FQA detected in the circulatory system, was indicative of extensive metabolic events occurring in the gastrointestinal tract. These events might involve hydrolysis of CQA to caffeic acid and other compounds due to the action of esterases present in intestinal wall [5]. Sulfated metabolites might be due to sulfation occurring in the small intestine and the liver [5].
The time when Cmax is detected (Tmax) for certain compounds, such as free dihydroferulic acid and dihydrocaffeic acid and their sulfated conjugates, was between 4.7 – 5.2 h, indicating that absorption occurred in the large intestine. Strains of Escherichia coli, Bifidobacterium lactis, Lactobacillus gasseri were found to be capable of producing esterase to break down CGAs in the colon [15]. Moreover, the studies proposed that feruloylglycine and dihydroferulic acid conversion from ferulic acid and metabolism of caffeic acid occurred in the colon [16]. For 5-CQA, two caffeoylquinic acid lactone-O-sulfates (CQAL-O-sulfates), and three FQAs, the Tmax values were between 0.6 to 1.0 h, which suggest absorption in the small intestine. However, these compounds were found to have low Cmax values ranging from 2.2 to 27 nM, which is indicative of low-level absorption [5].
These and other findings suggest that colonic metabolites CGAs might play an important role in contributing to health benefits of CGAs because two-thirds of CGAs are metabolized in the colon. Low Cmax values of certain compounds, including CQAs, indicate that some CGAs are not well absorbed in their intact form. Substantial variability from different subjects might due to some genetic factors or microbial variation of different individuals affecting metabolism and absorption processes since no correlation was found between excretion with gender, age, weight, height, or body mass index [13].
Neuroprotective Mechanisms of CGAs on Key Components of Alzheimer’s Disease
AD and other neurodegenerative diseases have been associated with oxidative neuronal death injury. Disease mechanism of AD has been linked to cellular damage due to oxidative stress in the brain, which is mediated by detrimental compounds called reactive oxygen species (ROS), including highly reactive hydroxyl radicals produced from hydrogen peroxide (H2O2) [4]. Antioxidant properties of CGAs are related to direct scavenging of hydroxyl radicals in a dose-dependent manner [18]. ROS promote apoptosis as well as oxidative damage to cellular protein, lipids, mitochondria, and DNA, subsequently leading to chronic diseases.
Studies have shown that brain of AD patients is under considerable oxidative stress since brain has relatively low amount of antioxidant enzymes, making it susceptible to oxidative damage [4, 19]. Aβ1-42 peptide contains methionine 35 residue that contributes to oxidative stress, which leads to neurotoxicity [20]. Mitochondrial dysfunction, which is commonly associated with apoptosis, resulted from ROS accumulation and expression of pro-apoptotic proteins, including Bax. ROS accumulation and pro-apoptotic proteins can increase mitochondrial permeability causing mitochondria to release cytochrome c. This leads to the activation of cysteine proteases called caspases, which consequently cause neuronal apoptosis [21]. Endogenous antioxidant enzymes, including glutathione peroxidase and catalase, are usually able to diminish adverse effects caused by ROS for tissue to maintain a healthy redox state [22].
Chemical assays, cell cultures, and animal models are useful investigation methods to elucidate the mechanisms underlying the beneficial
effect of CGAs on cells and enzymes that are linked to AD.
1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2’-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) (ABTS), and oxygen radical absorbance capacity (ORAC) are common assays that are used to measure antioxidant capacity of phenolic compounds in vitro. CGAs have been shown to exhibit dose dependent antioxidant activity in scavenging DPPH radicals, ABTS radials, hydroxyl radicals, peroxyl radicals, as well as superoxide anions.
In DPPH and malonaldehyde-gas chromatography (MA-GC) assays the activity of 1μg/mL and 20μg/mL of 5-CQA ranged from 5.18 to 50.69% for DPPH assay, and from 1.36 to 44.41% for MA-GC assay [11]. DPPH-radical scavenging activities of 3-CQA, 4-CQA, and 5-CQA were similar, suggesting that ester bond position in the structure does not have much effect on the activity. However, compared to FQAs, CQAs have better scavenging activity, indicating that hydroxyl groups at 3-and 4-positions in the benzene rings are important in determining antioxidant activity [23]. At 10 μM, CQAs are also able to scavenge superoxide radicals by 48-51%, which can also be attributed to hydroxyl groups in the benzene ring structure [23].
As most of CGAs are extensively metabolized in the colon, it is necessary to determine the antioxidant activity of CGA metabolites. ABTS and ORAC analyses showed that caffeic, isoferulic and dihydroferulic acids have stronger antioxidant activity than CGA. Colonic metabolites tested, such as m-coumaric acid and 3-(hydroxyphenyl) propionic acid, also showed antioxidant activity in ABTS and ORAC methods although the activity is slightly lower than CGA [24].
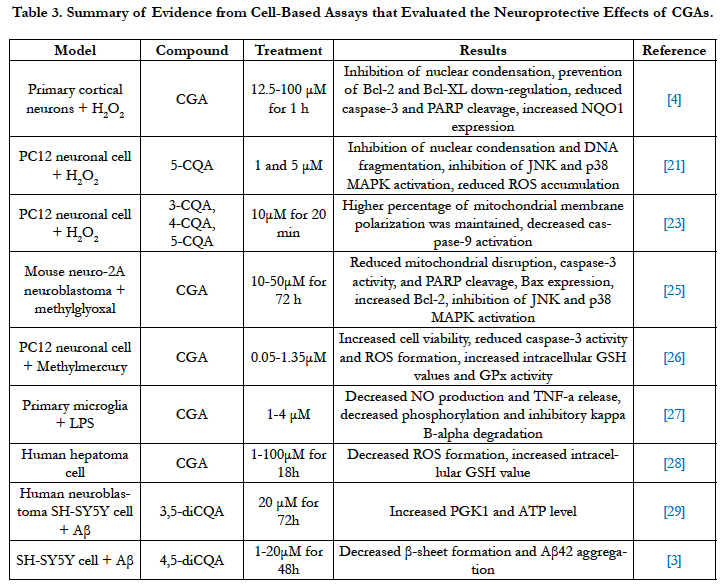
CGAs provide protective effects against cytotoxicity stimulated by various oxidative stressors including H2O2, methylglyoxal, and methylmercury. In a study using primary cortical neurons, H2O2 was shown to cause nuclear condensation in cells, which is considered as morphological characteristic of apoptosis. Pretreating cells with caffeinated coffee, decaffeinated coffee, and CGAs before they were subjected to 50 μM H2O2 for 24 h showed strong inhibition of this phenomenon in neuronal cells [4]. The proposed mechanism is through the prevention of H2O2-induced down-regulation of anti-apoptotic proteins Bcl-2 and Bcl-XL, which promotes cell survival [4]. Moreover, cell viability is increased due to prevention of caspase-3 cleavage and subsequently poly (ADP-ribose) polymerase (PARP) cleavage by caspase-3 [4]. Pretreatment also resulted in upregulation of NADPH: quinine oxidoreductase 1 (NQO1) expression in neurons, which helps to protect neurons from H2O2-induced apoptosis [4]. Compared with the control, CGAs significantly inhibit H2O2-induced increase in the number of condensed nuclei and apoptosis in primary neuronal culture. The findings show that the observed effect between caffeinated and decaffeinated coffee are similar, suggesting that CGAs have the potential to drive the effect, as they are present in both caffeinated and decaffeinated coffee [4].
Similar neuroprotective effect of CGAs has been observed in PC12 neuronal cell line by attenuating H2O2-induced cell death. 1μM and 5μM CGA were found to attenuate oxidative cell death. Instant decaffeinated coffee (1 and 5μg/ml) also yielded the same result in attenuating cell death. There is an observed dosedependent effect of CGAs and decaffeinated coffee in reducing the accumulation of ROS in H2O2-treated PC12 neuronal cells. Mechanism proposed is related to inhibition of nuclear condensation and DNA fragmentation in cells after pretreatment [21]. Similar to nuclear condensation, DNA fragmentation is also a morphological change associated to H2O2-induced apoptosis. Pretreatment with decaffeinated coffee and CGAs prevented the down-regulation of Bcl-XL, as well as reduced caspase-3 expression and subsequent PARP cleavage [21]. Aside from being the agent that causes oxidative stress, ROS are signaling molecules that activate protein kinase cascades including c-Jun N-terminal protein kinase (JNK) and p38 mitogen-activated protein kinase (MAPK). Pretreatments resulted in inhibition of JNK and p38 MAPK and the inhibition of these enzymes is critical because JNK and p38 MAPK are able to induce apoptosis due to H2O2 in neurons [21].
CGAs were also found to exert protective effect on mouse Neuro-2A neuroblastoma (Neuro-2A) cell subjected to methylglyoxal. High level of methylglyoxal in blood or tissue has been associated with diabetic models, which can eventually lead to diabetic neuropathy, such as AD [25]. Treatment with CGAs resulted in lower methylglyoxal-induced cell apoptosis in similar mechanisms with aforementioned studies. These mechanisms include reduction of mitochondrial disruption and caspase-3 activity, inhibition of Bax expressions, JNK and p38 MAPK activation, decreased cleavage of PARP, as well as increased Bcl-2 expression [25].
Mitochondrial membrane depolarization is one of the critical characteristics of ROS-induced neuronal cell death. Pre-exposure of H2O2-treated PC-12 cells to CQAs was shown to maintain 77% of mitochondrial membrane polarization compared to 50% of the control cells [23]. CQAs were also able to suppress H2O2- induced caspase-9 activation through maintaining the integrity of mitochondrial membrane. Similar to caspase-3, caspase-9 is a cysteine protease that plays a role in cell apoptosis.
In PC12 cells treated with methylmercury, CGAs were found to increase cell viability in a dose-dependent manner by reducing cell damage. Reduction of apoptotic cells, caspase-3 activity, and suppression of ROS formation were observed [26]. The proposed mechanism is related to the delay of cellular antioxidant consumption by glutathione peroxidase (GPx) because an increase in intracellular GSH values and activity of GPx were observed [26].
Microglia activation promotes neurotoxicity due to the release of pro-inflammatory factors, such as nitric oxide (NO), superoxide, and tumor necrosis factor-α (TNF-α) [27]. By diminishing the activation of microglia, progression of neurodegenerative diseases could be impeded. CGAs were able to suppress lipopolysaccharide (LPS)-stimulated microglia activation by reducing NO production and release of TNF-α, subsequently increasing survival of dopaminergic (DA) neuron. CGAs prevented nuclear factorkappaB (NF-αB) translocation, as well as decreased LPS-induced phosphorylation and inhibitory kappa B-alpha degradation [27].
In another study that investigated the effect of CGAs in human hepatoma cell line (HepG2), treating the cells with CGAs for 18 h without exposing the culture to pro-oxidant resulted in increased glutathione levels, while ROS formation was decreased. CGAs treatment had little effect on cell viability as caspase cascade and expression levels of Bcl-XL, Bcl-2, and Bax proteins were not affected [28]. Beneficial effect of CGAs against oxidative damage is likely due to enhanced cellular defense system related to lower ROS formation and increase in antioxidant potential related to glutathione (GSH) formation [28].
These findings suggest that CGAs provide protective effects by reducing the accumulation of intracellular ROS and by regulating the MAPK signal pathways. CGAs are also able to up-regulate Bcl-XL and down-regulate caspase-3 and caspase-9, which are key proteins and enzymes linked to apoptosis. Moreover, CGAs are able to inhibit microglia activation; hence they might inhibit the progression of AD.
In relation to Aβ, treating human neuroblastoma SH-SY5Y cells with 3,5-diCQA was shown to inhibit Aβ-induced neurotoxicity. Increased mRNA expression of phosphoglycerate kinase 1 (PGK 1) glycolytic enzyme and intracellular adenosine triphosphate (ATP) level were observed [29]. 3,5-diCQA and 4,5-diCQA were also shown to possess anti-amyloidogenic property. In human neuroblastoma SH-SY5Y cells, there is an observed dose-dependent inhibition of 42-mer amyloid beta-protein (Aβ42) aggregation. Aβ42 is important in the pathogenesis of AD due to its ability to aggregate in the form of beta sheet to induce neurotoxicity. 20 μM of 4,5-diCQA reduced Aβ42-induced neurotoxicity and restored cell viability by preventing beta sheet formation [3]. This property is related to caffeoyl group in CQA that is able to form covalent bond with Aβ42 residues, which prevents the formation of Aβ oligomers and destabilizes Aβ42 aggregates [3] (Table 3).
Table 3. Summary of Evidence from Cell-Based Assays that Evaluated the Neuroprotective Effects of CGAs.
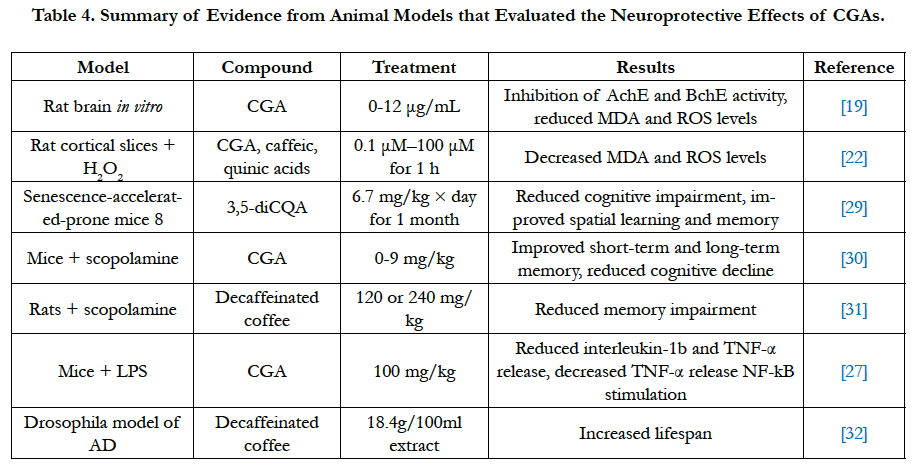
Studies conducted in rodent models further corroborate the neuroprotective effect of CGAs shown to attenuate oxidative stress in cell cultures. Pathogenesis of AD has been linked to decrease in cholinergic neurons, leading to deposition of amyloid protein and hence progression of AD [19]. As acetylcholinesterase (AChE) and butyrylcholinesterase (BchE) are key enzymes linked to AD, inhibiting the activity of these enzymes is regarded as an effective strategy against AD by enhancing cholinergic neuronal function [19]. Dose-dependent inhibition of AchE and BchE activity was observed when rats were subjected to 0-12 μg/mL CGA and caffeic acid. However, caffeic acid was found to exhibit stronger activity in inhibiting AchE and BchE activity compared to CGA. AchE and BchE break down acetylcholine and butyrylcholine in the brain, which result in reduced communication between nerve cells using acetylcholine or butyrylcholine as a chemical messenger. Thus, the inhibition of these enzymes results in a temporary improvement of AD symptoms as inhibition results in slower acetylcholine and butyrylcholine breakdown in brain and prevention of oxidative neurodegeneration [19].
In ex vivo rat cortical slices treated with H2O2, CGA was found to prevent decline in intensity of 5-triphenyltetrazolium chloride (TTC) staining in a dose-dependent manner, and reduce H2O2- induced MDA and ROS increase in brain tissues [22]. CGA had protective effect at as low as 0.1 μM, although 10 μM was required to have similar staining level as control. While caffeic acid has similar neuroprotective effect as CGA, quinic acid did not show strong protective effect against H2O2-induced alteration in the brain [22].
Administering 3,5-diCQA to senescence accelerated-prone mice (SAMP8) resulted in reduced cognitive impairment as spatial learning and memory were improved. An observed increased in intracellular ATP is likely due to overexpression of a glycolytic enzyme, phosphoglycerate kinase-1 (PGK1), which plays a role in glycolysis pathway [29]. Similarly, in mice with scopolamineinduced amnesia, CGA was found to increase spontaneous alternation behavior in Y-maze test, which indicates improvement of short-term memory [30]. Moreover, CGA treatment improved long-term memory and reversed scopolamine-induced cognitive impairments by inhibiting AchE activity and reducing MDA levels in hippocampus and frontal cortex [30]. Similarly, decaffeinated coffee attenuated scopolamine-induced memory impairment in rats by reducing TNF-α release and suppressing NF-αB stimulation in their hippocampus [31]. On the contrary, treating rats with decaffeinated coffee without scopolamine was not found to improve learning and memory. Pretreating mice with 100 mg/kg CGA before injecting LPS was found to attenuate LPS-induced release of interleukin-1β and TNF-α in injection site, which was substantia niagra (SN). CGA attenuated microglia activation in SN and protected against LPS-induced neurotoxicity [27].
In transgenic Drosophila model of AD, decaffeinated coffee suppressed the decrease in lifespan of the fly due to Aβ expression. Neuroprotective mechanism of decaffeinated coffee is related to the activation of cytoprotective transcription factor NF-E1- Related factor (Nrf2) [32]. However, other components present in decaffeinated coffee besides CGAs might contribute to its neuroprotective effect (Table 4).
Although CGAs and their metabolites are able to attenuate oxidative stress and provide neuroprotective effects in vitro and in animal models, current clinical trial is limited to studying the effect of coffee consumption on reducing oxidative damage. Consumption of CGAs enriched coffee shows changes in biomarkers of oxidative damage, including reduced lipid peroxidation measured by 8-Isoprostaglandin F2α in urine and 3-nitrotyrosine in plasma, and reduced formation of oxidized purines and pyrimidines marked by lower DNA migration in lymphocytes [33]. Moreover, lower plasma level of oxidized LDLs and malondialdehyde (MDA) were observed, indicating increased total antioxidant capacity [33]. Increased expression of several genes, including NQO1, superoxide dismutase (SOD) and glutathione-S-transferases (GST), were observed after coffee consumption although the increase was not significant [33]. In another study by Mišíket al., [34], coffee consumption reduced DNA migration due to oxidized purines. However, there was no significant change in other biomarkers, including LDLs and MDA levels.
In addition to mitigating oxidative stress, decaffeinated coffee enriched with CGA positively affects cognition in healthy elderly population. High CGA content coffee reduced headache and mental fatigues, as well as increased alertness in subjects when compared to low CGA content coffee, indicating that CGA improves brain-related function [35].
Conclusions
According to current bioavailability studies, CGAs are highly bioavailable in humans with approximately 33% absorption from the small intestine. Considering that CGAs undergo extensive metabolism in the colon, it is necessary to evaluate the role of colonic microflora and the effect of CGA metabolites in neuroprotection. CGA metabolites, including caffeic and dihydroferulic acids show high antioxidant capacity in vitro. Studies evaluating the antioxidant capacity of CGAs in modulating oxidative stress in vitro and in vivo show promising results in regards to the potential of reducing the risk of neurodegenerative diseases. However, there is currently a limited number of animal studies and human clinical trials that evaluate CGAs in relation to AD. Further research is required to assess neuroprotective benefits of CGAs by using metabolomics and human clinical studies.
References
- Schowalter WR (1978) Mechanics of Non-Newtonian Fluids. Pergamon Press, Oxford- Frankfurt.
- Nicosia MA, Robbins JA (2007) The Usefulness of the Line Spread Test as a Measure of Liquid Consistency. Dysphagia. 22(4): 306-311.
- Pelletier CA (1997) A comparison of consistency and taste of five commercial thickeners. Dyphagia. 12: 74–78.
- Glassburn D, Deem J (1998) Thickener viscosity in dysphagia management: Variability among speech-language pathologists. Dysphagia. 13(4): 218–222.
- Martinez-Padilla, L.P. Food Suspensions and Food Engineering (2005) Food Engineering-Vol II 3.4:1-338.
- Paik N-J, Han TR, Park JW, Lee EK, Park MS, et al., (2004) Categorization of dysphagia diets with the line spread test. Arch Phys Med Rehabil.85(5): 857-861.
- Mann L, Wong K (1996) Development of an objective method for assessing viscosity of formulated foods and beverages for the dysphagic diet. J Am Diet Assoc. 96(6): 585–588.
- Adams MC, Birdsall EL (1946) New consistometer measures corn consistency. Food Industries. 18: 78–80.
- Grawemeyer EA, Pfund MC (1943) Line spread as an objective test for consistency. J Food Sci. 8(2): 105–108.
- Jun X, Michael N (2007) Rheological properties of batter systems containing different combinations of flours and hydrocolloids. J Sci Food Agric. 87(7): 1292–1300.
- Acosta K, Cavender G, Kerr W (2011) Sensory and physical properties of muffins made with waxy whole wheat flour. J Food Quality . 34(5): 343–351.
- Johnson FCS (1988) Utilization of American-produced rice in muffins for gluten-sensitive individuals. Fam Consum Sci Res J. 17(2): 175-183.
- Kim CS, Walker CE (1992) Interaction between starches, sugars and emulsifiers in high-ratio cake model systems. Cereal Chem. 69(2): 206-212.
- Roura SI, Del Valle CE, Aguero La, Davidovich LA (2007) Changes in apparent viscosity and vitamin c retention during thermal treatment of butternut squash (cucurbita moschata duch) pulp: effect of ripening stage. J of Food Quality. 30(4): 538-551.
- Dhanasekharan M, Huang Ha, Kokinp JL (1999) Comparison of observed rheological properties of hard wheat flour dough with predictions of the giesekus-leonov, white-metzner and phan-thien tanner models. J Texture Studies 30(6): 603-623.