Increased insulinogenic indexes after liquid food (Inslow) intake in subjects with impaired glucose tolerance and type-2 diabetes: a randomised controlled trial
Liu E2§, Kume A1§, Yan H3, Wang X4, Zheng H2, Konno T1, Ding H4, Shen H4, Sasaki H1, 5*, Ji ZS1, 2*
1 Division of Research & Development Division, Meiji Co., Ltd., 540 Naruda, Odawara, Kanagawa 250-0862, Japan.
2 Laboratory of Medical Foods, Shanghai Institute of Planned Parenthood Research, 2140 Xie-Tu Road, Shanghai 200032, China.
3 Shuguang Hospital Affiliated to Shanghai University of Traditional Chinese Medicine, 185 Pu-an Road, Shanghai 200021, China.
4 Division of Research and Development, Shanghai Meiji Health Science and Technology Co., Ltd., 285 Jianguo Road, Shanghai 200031, China.
5 Department of Nutritional and Life Science, Kanagawa Institute of Technology, 1030 Shimo-ogino, Atsugi, Kanagawa 243-0203, Japan.
§- The authors equate to this paper.
*Corresponding Author
Zai-Si Ji & Hajime Sasaki
Research & Development Division, Meiji Co., Ltd. 540 Naruda,
Odawara, Kanagawa 250-0862, Japan.
Tel.: +81-465-37-3617
Fax: +81-465-37-3624
E-mail: zai-si_ji@kta.biglobe.ne.jp and hjmsasaki@nifty.com
Received: February 14, 2016; Accepted: March 30, 2016; Published: April 07, 2016
Citation: Zai-Si Ji, Hajime Sasaki et al. (2016) Increased insulinogenic indexes after liquid food (Inslow) intake in subjects with impaired glucose tolerance and type-2 diabetes: a randomised controlled trial. Int J Clin Ther Diagn. 4(2), 110-118.
DOI : dx.doi.org/10.19070/2326-3350-1600050
Copyright: Zai-Si Ji, Hajime Sasaki© 2016. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: A carbohydrate adjusted liquid formula (Inslow) containing palatinose as the major (>50%) carbohydrate ingredient was developed. Longterm (3-5 months) consumption of Inslow was reported to improve glycemic control by reducing the postprandial plasma glucose levels of subjects with diabetes and impaired glucose tolerance (IGT). The present study aimed to elucidate the mechanism of postprandial glycaemic responses.
Methods: Two randomised crossover studies with a washout period of 1 week were conducted in 12 patients with type 2 diabetes, 13 subjects with IGT, and 11 healthy people. Before and after intake of Inslow or the standard balanced formula, plasma glucose, insulin and free fatty acid concentrations were determined.
Results: Compared with intake of the standard balanced formula, consumption of Inslow significant lowered the peak values of plasma glucose and increased the insulinogenic indexes in each group. Especially in the subjects with IGT, the increase in plasma insulin concentration was greater in the Inslow group than in the standard balanced formula group.
Conclusions: Intake of Inslow increased the insulinogenic indexes, which contributes to preventing postprandial hyperglycemia without altering the amount of total energy, preventing the transition from normal glucose tolerance to IGT and then to overt type 2 diabetes.
Trial registration: Current controlled trials NCT02641743. Registered 31 December 2015 in clinicaltrials.gov.
2.Abbreviations
3.Introduction
4.Materials and methods
4.1.Subjects
4.2.Consent to Publish
4.3.Study Design
4.4.Statistical Analysis
5.Results
5.1.Subjects’ Background Characteristics
5.2.Glucose and Insulin Responses
5.3.Insulinogenic index
5.4.Free Fatty Acid Responses
6.Discussion
7.Conclusions
8.Declarations
9.References
Keywords
Carbohydrate; Plasma Glucose; Insulin; Impaired Glucose Tolerance; Diabetes; Palatinose.
Abbreviations
impaired glucose tolerance (IGT); free fatty acid (FFA); hemoglobin (Hb); platelet blood cells (PLT); total lymphocytes count (TLC); plasma glucose (Glu); hemoglobin A1C (HbA1C); blood albumin (Alb); aspartate aminotransferase (AST); alanine aminotransferase (ALT); lactate dehydrogenase (LDH); total bilirubin (T-Bil); glutamyltranspetidase (γ-GTP); leucine aminopeptidase (LAP); blood urea nitrogen (BUN); serum creatinine (Cr); uric acid (UA); high density lipoprotein cholesterol (HDL-Cho); low density lipoprotein cholesterol (LDL-Cho); triglyceride (TG); lipoprotein lipase (LPL).
Introduction
Diabetes is, a group of metabolic diseases characterized by hyperglycemia that results from defective insulin production and/ or insulin action. Diabetes is known to be a major risk factor of microvascular complications (e.g., nephropathy, retinopathy, and neuropathy) and macrovascular complications (e.g., coronary heart disease, peripheral arterial disease, and cerebrovascular disease).
The dietary intakes of patients with diabetes are often reported as inadequate to supply the necessary amounts of energy. Persistent lack of energy intake can lead to energy malnutrition and cause various health-related problems such as impaired daily life activity, decreased cure rate, and increased incidence of complications. In connection with the identified metabolic abnormalities in diabetes, selection of dietary ingredients is important in order to prevent the development of diabetes-associated complications. In particular, the types of carbohydrate in diets are important to prevent postprandial hyperglycemia and to provide sufficient amounts of energy to prevent energy malnutrition [1].
Palatinose (isomaltulose) is an analogue of sucrose that is known for its slower digestion and absorption in the small intestine than sucrose [2, 3]. Therefore, it is useful for weakening the peak plasma glucose concentration in diabetic patients [4]. By using palatinose as the major carbohydrate ingredient (>50%), we newly developed a liquid enteral formula named “Inslow”. The intake of this formula significantly improved the postprandial glycemic responses in genetically obese Zucker rats [5]. Moreover, long term intake (30 days) of Inslow prevented the accumulation of viscera fat in mice [6]. The effect on improvement postprandial glycemic responses was also observed in subjects with impaired glucosetolerance (IGT) and diabetes who had a longterm (3–5 months) consumption of Inslow [7, 8]. In a clinical trial, increased fat oxidation was observed in healthy subjects after intake of Inslow [9]. This effect of Inslow is thought to be due to an increase in the hepatic PPAR-a and adipocyte PPAR-r gene expressions [10].
Except palatinose, various carbohydrates, nitrogen sources and fatty acids are included in Inslow, which may have a comprehensive effect in subjects. The aim of this report was to more precisely evaluate the acute effects of Inslow on postprandial glycemic responses by observing metabolic responses to the intake of either Inslow or the standard balanced formula. We describe the results from the following three groups of participants: healthy people, patients with IGT and patients with diabetes in Shanghai. This study provides the scientific and practical basis of enteral nutrition suitable for prevention and treatment of glucose metabolic disorders that lead to diabetes.
Materials and methods
Subjects between 52 and 61 years old were enrolled. Twelve patients with type 2 diabetes and 13 patients with IGT from the Shanghai University of Traditional Chinese Medicine, affiliated with Shanghai Shuguang Hospital, were selected. Eleven healthy controls were also included in this study. The patients with type 2 diabetes (symptoms of diabetes plus fasting plasma glucose level of >7.0 mmol/L and glucose level after a 75 g oral glucose tolerance test [OGTT] or casual blood glucose level of >11.1mmol/L) and IGT (glucose level after a 75 g OGTT between 7.8 and 11.1mmol/L) had no infectious diseases and were not receiving any drug of any sort. OGTT was performed after 10–12 h of overnight fasting by administering a 75 g oral glucose load for 2 min and obtaining blood samples at baseline and 2 h after glucose load for serum glucose measurements. Glucose tolerance status was defined as normal glucose tolerance (NGT), IGT or diabetes according to the World Health Organization criteria [11, 12].
The study protocol and its constraints were explained to each of the 36 participants. All the subjects provided informed consent to participate in this study. The study and its consent form were in accordance with the guidelines laid down in the Declaration of Helsinki. All procedures involving human subjects were approved by the human subjects ethical committee of the Shanghai University of Traditional Chinese Medicine, affiliated with Shanghai Shuguang Hospital.
In a randomized crossover trial, the test enteral nutrition formula (Inslow) and standard balanced formula (Meibalance) were administered to 11 healthy people, 13 patients with IGT and 12 patients with type 2 diabetes. The subjects were assessed according to the postprandial status after intake of the two test meals in random order, with a washout period of 1 week. On the day prior to the test day, the subjects were asked to stay at the same facility and to take the same supper controlled at 450 kcal controlled, after 21:00, with only water allowed as ad libitum and without exercise. Each participant provided a fasting blood sample and then requested to consume one of the test meals (200 kcal per serving of 200ml), that is, either Inslow or the standard balanced formula at 7:00 AM. Subsequent blood samples were collected at 30, 60, 90, and 120 min after meal consumption. The plasma concentrations of glucose, insulin, and free fatty acids (FFAs) were measured in each blood sample. Plasma glucose level was measured by using the hexokinase method; plasma insulin level, by using the serum radioimmunoassay method; and plasma FFA level, by using the enzyme chemical method. Routine blood and biochemical tests were performed only at fasting stage, by using the same blood samples, including red blood cell, white blood cell, total protein, alkaline phosphatase, and total cholesterol (T-Cho), in order to confirm the status of each subject. The collected blood samples were used in the analyses conducted at Shanghai Meizhong Clinical Measurement Centre.
The enteral nutrition meal Inslow was prepared with 56% palatinose, 23% dextrin, and 15% fibre as carbohydrates. The standard balanced meal included 85% dextrin as the main carbohydrate, together with sugar and fibre. The ingredients of the test enteral and control nutrition preparations are presented in Table 1.
Table 1. Composition of the test meals, Inslow and Standard balanced formula.
*1: retinol equivalent; *2: niacin equivalent; †: analytical value.
For the statistical analyses, repeated-measures analysis of variance (ANOVA) was performed. The factors included in the analyses were subject characteristics, disease stages, meal, and time. The interactions between meal and time (meal × time) were calculated for each variable (group and meal). ANOVA was followed by a least significant difference (LSD) multiple comparison post-hoc analysis. Within-subject differences adjusted for baseline and individual XXX, YYY were analysed by using repeated-measures ANOVA. The insulinogenic indexes were calculated by dividing the increment in serum insulin by the increment in plasma glucose from 0 to 30, 60, 90, and 120 min, respectively after each meal.
Results
The subjects’ baseline characteristics, including sex, age and biochemical profiles are shown in Table 2. None of the patients dropped out or were withdrawn from the study. The data represent normal health people, IGT and diabetes patients without outliers. As shown in Table 2, some of the reference indexes significantly differed between the diabetic, IGT, and healthy subjects. Fasting Glu levels were highest in the diabetic subjects and lowest in the healthy subjects (diabetes > IGT > healthy subjects), indicating that the three groups of subjects were in three different diabetic stages. Serious adverse effects such as anaemia, renal disorders, or hepatic disorders did not occur in this study.
Table 2. Baseline demographics (age, sex) and clinical characteristics of fasting blood sample. Hemoglobin (Hb); red blood cell (RBC); white blood cell (WBC); platelet blood cells (PLT); total lymphocytes count (TLC); plasma glucose (Glu); hemoglobin A1C (HbA1C); total protein (TP); blood albumin (Alb); aspartate aminotransferase (AST); alanine aminotransferase (ALT); lactate dehydrogenase (LDH); total bilirubin (T-Bil); alkaline phosphatase (ALP); glutamyltranspetidase (Υ-GTP); leucine aminopeptidase (LAP); blood urea nitrogen (BUN); serum creatinine (Cr); uric acid (UA); total cholesterol (T-Cho); high density lipoprotein cholesterol (HDL-Cho); low density lipoprotein cholesterol (LDL-Cho); triglyceride (TG).
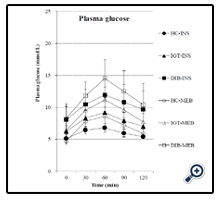
We investigated the effects of Inslow on glucose metabolism in three different diabetic stages (healthy, IGT, and type 2 diabetes). According to the results of repeated-measures ANOVA, the postprandial response to glucose and insulin were more prominent in the healthy controls and tended to diminish with the progression of diabetes. Plasma glucose concentrations increased significantly in all the groups after both meals (p < 0.0001), as shown in Figure 1. At each time point after both meals, the glucose concentrations were highest in the diabetes group, followed by the IGT group and then the healthy control group. In addition, plasma glucose concentration returned to its fasting concentration 120 min after both meals in the healthy control group and IGT group with less extent. In the diabetes group, however, it did not return to its fasting concentration 120 min after both meals. The postprandial glucose response was gradually diminished when Inslow was consumed rather than the standard balance formula in the three groups, in the order of healthy controls (p = 0.0063), patients with IGT (p = 0.0837), and patients with type 2 diabetes (p = 0.2327). Significant differences were observed among the three groups after intake of Inslow (p < 0.0001) and the standard balanced formula (p < 0.0001).
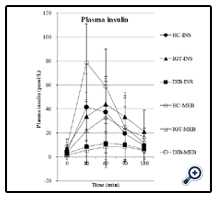
As shown in Figure 2, plasma insulin concentrations increased significantly in all the groups after both meals (p < 0.0001). In contrast to glucose profile, the insulin concentrations were highest in the healthy control group, followed by the IGT group and then the diabetes group. It is interesting that the insulin level was greater in the IGT group when Inslow was consumed than when the standard balanced formula was consumed, although the increase in plasma glucose level was attenuated. The peak of insulin secretion occurred 60 min after intake of Inslow, compared with 30 min after intake of the standard balanced formula.
To gain more quantitative insight into the effect of each enteral formula on glucose metabolism, individual postprandial values subtracted from the values in the fasting state (time 0) are shown in Figure 4. The values above or under the line in the figure indicate increased variations when the standard balanced formula or Inslow was consumed, respectively. The frequencies of the individual values are summarised in Table 3. Most of the postprandial plasma glucose variations were higher (p < 0.0001) in all the groups after intake of the standard balanced formula (Figure 4a–c), Table 3). The variation in plasma insulin levels showed a characteristic distribution along with the progression of diabetes. The insulin profile was significantly lower after Inslow intake in the healthy control group (p < 0.001) but shifted toward a greater increase after intake of Inslow in the IGT group (p = 0.0186) and showed no change in the diabetes group (p = 0.1025; Figure 4d–f; Table 3).
Figure 4 Variations of postprandial values from the fasting state (time 0) after Inslow or Meibalance (standard balanced formula) intake
Table 3. Frequencies of clinical indexes showing higher variation from the fasting state (time 0) when Inslow was consumed than Meibalance, or Meibalance was consumed than Inslow. INS: Inslow group; MEB: standard balanced formula group; HC: health control; DIB: diabetes; IGT: impaired glucose-tolerant subjects.
In order to understand comprehensively the separate effects of postprandial glucose and insulin, we assessed insulinogenic indexes. The insulinogenic index during OGTT decreased from normal glucose tolerance to IGT and to type 2 diabetes, As shown in Figure 4 (g–i), the insulinogenic indexes were higher (p < 0.0001) in all the groups after intake of Inslow (Table 3) than after intake of the standard balanced formula.
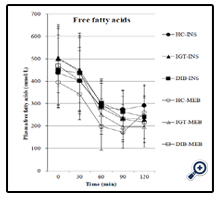
As shown in Figure 3, the plasma FFA concentrations decreased significantly in all the groups after both meals (p < 0.0001) and did not show a significant difference between the two meals in the healthy control group (p = 0.1153), IGT group (p = 0.5261), and type 2 diabetes group (p = 0.6718). Moreover, it did not show significant differences among the three groups after intakes of Inslow (p = 0.9911) and the standard balanced formula (p = 0.3951). Postprandial FFA variations showed a significant increase in the healthy control group (p = 0.0028) and diabetes group (p = 0.0002) but no significant trend in the IGT (p = 0.2393) after intake of Inslow in comparison with intake of the standard balanced formula (Figure 4j–l, Table 3).
Discussion
Reduced insulin-secreting capacity is considered to play a more important role than insulin resistance during the transition from normal glucose tolerance to IGT and then to overt type 2 diabetic patients [13]. This is the first human study that was designed to evaluate the effects of Inslow intake on glucose metabolism in subjects with diabetes and IGT by short-term administration. The present study shows that Inslow intake, but not the standard balanced formula, had beneficial effects of significantly increasing the insulinogenic indexes in all the three groups (Figure 4).
In the healthy subjects, Inslow intake attenuated the increase in plasma glucose levels when compared with the standard balanced formula, which could be mainly attributed to the slower absorption of palatinose than sucrose and the decrease in the elevated rise of plasma insulin levels along with plasma glucose levels, which is consistent with the results of the previous study [9]. The new finding is that in subjects with IGT and diabetes with lower insulinogenic capacity than healthy subjects, plasma insulin levels recovered to amounts very close to those in the healthy controls when Inslow was consumed, or showed the same increase as that observed the standard balanced formula was consumed although the glucose levels were attenuated (Figure 2). It should be noted that their insulin levels peaked at 60 min after intake of either Inslow or the standard balanced formula, which is significantly later than the response peak (30 min) in the healthy subjects. This delay in insulin secretion might be due to impairment of the insulinsecreting responses in these patients.
The plasma T-Cho and LDL-Cho levels in the IGT group were higher than the normal ranges (< 5.17 and < 3.12 mmol/L, respectively; Table 2). This might be due to insufficient lipoprotein lipase (LPL) activities resulting from impaired glucose tolerance. The levels of T-Cho and LDL-Cho in the diabetes group were within the normal range just as in the healthy group, probably because not only LPL activity but also the glyceride synthesizing ability of the liver was decreased by liver injury in the diabetes group. Higher LPL activity was reported to be associated with greater triglyceride (TG) storage in subcutaneous adipose tissue, thereby reducing visceral adipose tissue accumulation and metabolic dysfunction [14]. Thus we speculate that the patients in both the IGT and diabetes groups had lipid metabolic dysfunction because of lower LPL activity. On the other hand, persons with adipose tissue deficiency also manifested a metabolic syndrome-like insulin resistance [15]. Thus, in take of diets with high-quality fat, neither too much nor too little, is important for controlling IGT and diabetes.
Inslow had a higher content of C18-unsaturated fatty acid than the standard balanced formula (3.3% to 2.8%), which may promote the secretion of glucagon-like peptide-1 (GLP-1) through receptors GRP40 and GPR120 in small intestinal epithelial L cells [16, 17]. GLP-1 are potent incretin hormones that enhance the glucose-dependent secretion of insulin from pancreatic beta cells and boost insulin secretion [18][19]. In addition, choline promotes GLP-1 secretion through the GPR119 receptor [20], which is a precursor to phosphatidylcholine and the tenfold higher content included in Inslow than in the standard balanced formula (18.2% to 1.8%). From the previous study results in Zucker fatty rats, 8-week consumption of a diet containing palatinose and oleic acid prevented β-cell loss, which was observed in rats consuming sugar and linoleic acid. Pancreatic islets may be the initial sites that translate the effects of different combinations of dietary carbohydrates and fats into metabolic changes [21]. In conclusion, Inslow intake improved the impaired insulin-secreting ability of the patients with diabetes and IGT. This effect may be attributed to the special composition of Inslow, including carbohydrates and fatty acids, including palatinose, dietary fibre [22], oleic acid, C18-unsaturated fatty acid and choline, which also may inhibit the progression of IGT into type 2 diabetes.
Postprandial hyperglycemia is often associated with enough energy provided through balanced ratios of protein, carbohydrate, and fat. To prevent postprandial hyperglycemia without altering the amount of total energy, a combination of carbohydrate components that does not induce hyperglycemia should be developed. Based on the results presented here, Inslow is expected to be a candidate diet that can provide sufficient energy to patients with IGT and diabetes patients with impaired glycemic responses.
Person with diabetes and IGT always develop liver and kidney complications that occur with glucose tolerance progression [23]. Hence, patients with IGT and diabetes tend to consume lowprotein diets to prevent the progression of metabolic disorders. However, long-term intake of low-protein diets enhance catabolic responses in nitrogen metabolism. High-quality protein diet is important for patients with IGT and diabetes.
Intake of essential amino acids, which are included in Inslow at 46% as the whole nitrogen content, may induce an increase in GLP-1 secretion [24, 25]. Unfortunately, the standard balance formula also contains the same 5% protein and has the same glutamine content and essential amino acid ratio in nitrogen as Inslow. Based on the above-mentioned results, the contribution of nitrogen components to the insulin-secreting capacity of patients with IGT and diabetes is thought to be similar between the two test meals. There is a possibility that the effect of Inslow on the improvement of insulin-secreting ability might be more significant when comparing to other nitrogen supplement, such as a normal milk product.
Conclusions
Intake of Inslow induced increased in the insulinogenic indexes, which contribute to the prevention of postprandial hyperglycemia without impairment of the amount of total energy, thereby preventing the transition from normal glucose tolerance to IGT and then to overt type 2 diabetes and thus enhancing the quality of life of diabetic patients.
Declarations
The study protocol and its constraints were explained to each of the 36 participants. All the subjects gave their informed consent to participate in this study. The study and its consent form were inaccordance with the guidelines laid down in the Declaration of Helsinki and all procedures involving human subjects were approved by the human subjects ethical committee of the Shanghai University of Traditional Chinese Medicine, affiliated with Shanghai Shuguang Hospital (2008-N073-01).
This work was sponsored by the Shanghai Health and Family Planning Foundation (201440324). Meiji Co., Ltd., Shanghai Institute of Planned Parenthood Research, Shanghai University of Traditional Chinese Medicine, and Shanghai Meiji Health Science and Technology Co., Ltd. provided support in the form of salaries for the authors as their researchers. However, these institutions did not play any role in the study design, data collection or analysis, decision to publish, or preparation of the manuscript. We declare the following interests: The research was conducted by using funds provided by the aforementioned institutes under a joint research agreement between them. The test enteral nutrition formula (Inslow) and standard balanced formula (Meibalance) were provided by Meiji Co., Ltd. This does not alter our adherence to all of the International Journal of food science, nutrition and dietetics policies on sharing data and materials, as detailed online in the Guide for Authors.
References
- Sheard NF, Clark NG, Brand-Miller JC, Franz MJ, Pi-Sunyer FX, et al. (2004) Dietary carbohydrate (amount and type) in the prevention and management of diabetes: a statement by the american diabetes association. Diabetes care 27(9): 2266-2271.
- Kawai K, Okuda Y, Yamashita K (1985) Changes in blood glucose and insulin after an oral palatinose administration in normal subjects. Endocrinol Jpn 32(6): 933-936.
- Lina BA, Jonker D, Kozianowski G (2002) Isomaltulose (Palatinose): a review of biological and toxicological studies. Food Chem Toxicol 40(10): 1375-1381.
- van Can JG, van Loon LJ, Brouns F, Blaak EE (2012) Reduced glycaemic and insulinaemic responses following trehalose and isomaltulose ingestion: implications for postprandial substrate use in impaired glucose-tolerant subjects. Br J Nutr 108(7): 1210-1217.
- Taketani Y, Shuto E, Arai H, Nishida Y, Tanaka R, et al. (2007) Advantage of a low glycemic index and low phosphate diet on diabetic nephropathy and aging-related diseases. J Med Invest 54(3-4): 359-365.
- Arai H, Mizuno A, Matsuo K, Fukaya M, Sasaki H, et al. (2004) Effect of a novel palatinose-based liquid balanced formula (MHN-01) on glucose and lipid metabolism in male Sprague-Dawley rats after short- and long-term ingestion. Metabolism 53(8): 977-983.
- Sakuma M, Arai H, Mizuno A, Fukaya M, Matsuura M, et al. (2009) Improvement of glucose metabolism in patients with impaired glucose tolerance or diabetes by long-term administration of a palatinose-based liquid formula as a part of breakfast. J Clin Biochem Nutr 45(2): 155-162.
- Oizumi T, Daimon M, Jimbu Y, Kameda W, Arawaka N, et al. (2007) A palatinose-based balanced formula improves glucose tolerance, serum free fatty acid levels and body fat composition. Tohoku J Exp Med 212(2): 91- 99.
- Arai H, Mizuno A, Sakuma M, Fukaya M, Matsuo K, et al. (2007) Effects of a palatinose-based liquid diet (Inslow) on glycemic control and the secondmeal effect in healthy men. Metabolism 56(1): 115-121.
- Matsuo K, Arai H, Muto K, Fukaya M, Sato T, et al. (2007) The Anti-Obesity Effect of the Palatinose-Based Formula Inslow is Likely due to an Increase in the Hepatic PPAR-alpha and Adipocyte PPAR-gamma Gene Expressions. J Clin Biochem Nutr 40(3): 234-241.
- Alberti KG, Zimmet PZ (1998) Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med 15(7): 539-553.
- American Diabetes Association (2006) Diagnosis and classification of diabetes mellitus. Diabetes Care 29(Suppl 1): S43-S48.
- Matsumoto K, Miyake S, Yano M, Ueki Y, Yamaguchi Y, et al. (1997) Glucose tolerance, insulin secretion, and insulin sensitivity in nonobese and obese Japanese subjects. Diabetes Care 20(10): 1562-1568.
- Serra MC, Ryan AS, Sorkin JD, Favor KH, Goldberg AP (2015) High adipose LPL activity and adipocyte hypertrophy reduce visceral fat and metabolic risk in obese, older women. Obesity 23(3): 602-607.
- Grundy SM (2015) Adipose Tissue and Metabolic Syndrome: Too Much, Too Little, or Neither. Eur J Clin Invest 45(11): 1209-1217.
- Hirasawa A, Tsumaya K, Awaji T, Katsuma S, Adachi T, et al. (2005) Free fatty acids regulate gut incretin glucagon-like peptide-1 secretion through GPR120. Nat Med 11(1): 90-94.
- Itoh Y, Hinuma S (2005) GPR40, a free fatty acid receptor on pancreatic beta cells, regulates insulin secretion. Hepatol Res 33(2): 171-173.
- Gromada J, Holst JJ, Rorsman P (1998) Cellular regulation of islet hormone secretion by the incretin hormone glucagon-like peptide 1. Pflugers Arch 435(5): 583-594.
- Baggio LL, Drucker DJ (2007) Biology of incretins: GLP-1 and GIP. Gastroenterology 132(6): 2131-2157.
- Lan H, Vassileva G, Corona A, Liu L, Baker H, et al. (2009) GPR119 is required for physiological regulation of glucagon-like peptide-1 secretion but not for metabolic homeostasis. J Endocrinol 201(2): 219-230.
- Sato K, Arai H, Miyazawa Y, Fukaya M, Uebanso T, et al. (2008) Palatinose and oleic acid act together to prevent pancreatic islet disruption in nondiabetic obese Zucker rats. J Med Invest 55(3-4): 183-195.
- Prosky L (2000) When is dietary fiber considered a functional food? Bio Factors 12(1-4): 289-297.
- Bedir A, Ozener IC, Emerk K (1996) Urinary leucine aminopeptidase is a more sensitive indicator of early renal damage in non-insulin-dependent diabetics than microalbuminuria. Nephron 74(1): 110-113.
- Tolhurst G, Zheng Y, Parker HE, Habib AM, Reimann F, et al. (2011) Glutamine triggers and potentiates glucagon-like peptide-1 secretion by raising cytosolic Ca2+ and cAMP. Endocrinology 152(2): 405-413.
- Reimer RA (2006) Meat hydrolysate and essential amino acid-induced glucagon-like peptide-1 secretion, in the human NCI-H716 enteroendocrine cell line, is regulated by extracellular signal-regulated kinase1/2 and p38 mitogen-activated protein kinases. J Endocrinol 191(1): 159-170.