Deep Neck Space Infections: Scope, Dangers and Predictors of Outcome in an Emerging Economy
Kokong DD1*, Adoga AA1, Akhiwu BI2, Rowland A2, Mugu JG3, Embu HY4, Ramyil VM5
1 Department of Oto-Rhinolaryngology, Head & Neck Surgery, College of Medicine, University of Jos & Jos University Teaching Hospital, Plateau State, Nigeria.
2 Department of Oral & Maxillofacial Surgery, College of Medicine, University of Jos & Jos University Teaching Hospital, Plateau State, Nigeria.
3 Department of Oto-Rhinolaryngology, Head & Neck Surgery, Jos University Teaching Hospital, Plateau State, Nigeria.
4 Department Of Anaesthesia, College of Medicine, University of Jos & Jos University Teaching Hospital, Plateau state, Nigeria.
5 Department of General Surgery, College of Medicine, University of Jos & Jos University Teaching Hospital, Plateau State, Nigeria.
*Corresponding Author
Dr. Daniel D. Kokong,
Department of ORL-Head & Neck Surgery (ORL-HNS),
Jos University Teaching Hospital, PMB 2076, Jos, Plateau State, Nigeria.
Tel: +2348023816024
E-mail: dankokong@yahoo.com
Received: February 17, 2017; Accepted: March 28, 2017; Published: March 30, 2017
Citation: Kokong DD, Adoga AA, Akhiwu BI, Rowland A, Mugu JG, et al., (2017) Deep Neck Space Infections: Scope, Dangers and Predictors of Outcome in an Emerging Economy. Int J Clin Exp Otolaryngol. 3(2), 56-62.
Copyright: Kokong DD© 2017. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: Study the scope, dangers and predictors of outcome of deep neck space infections in our setting.
Study Design: Retrospective cohort.
Setting: Public Academic Tertiary Hospital.
Method: Case files retrieved utilizing ICD-10 version 10 codes that met the inclusion criteria for deep neck space infections (DNSI) from January 1, 2012- June 30, 2016 were analyzed.
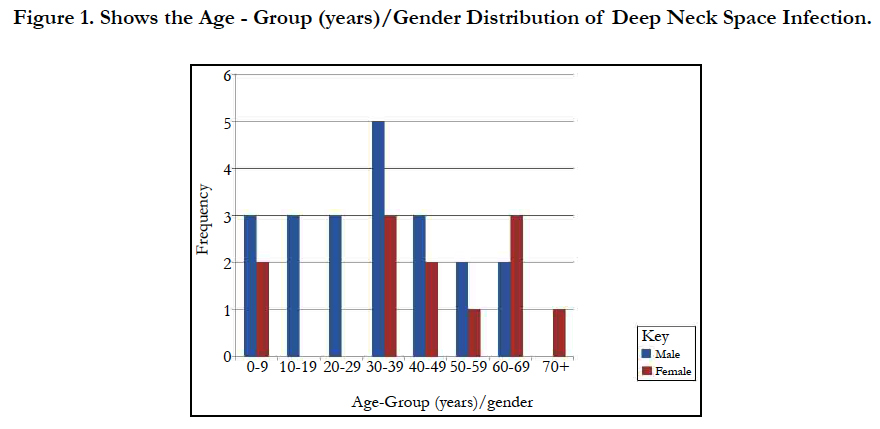
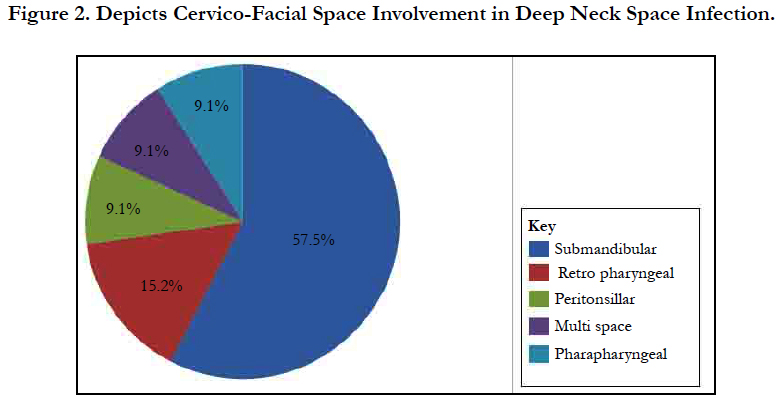
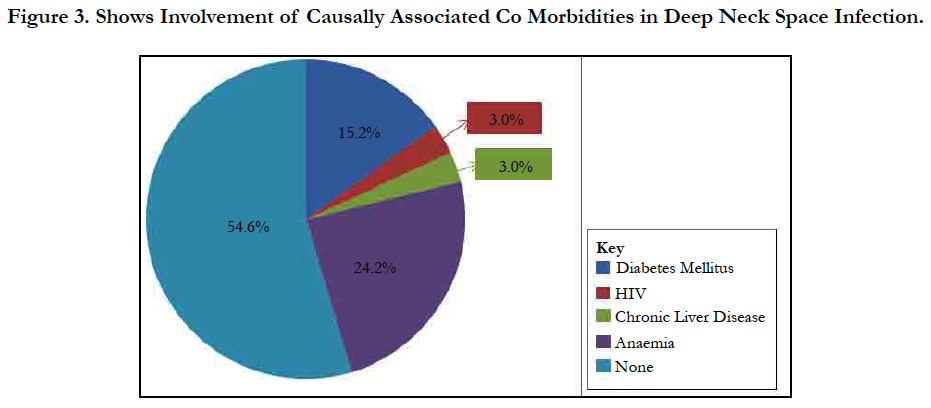
Results: Thirty-three cases of DNSI with a M:F=1.75:1 were studied. Age range was 6 months - 82years with mean = 35.3 years ± 26.0 Figure 1. In this, 60.6% presented early (≤ 7days) while 39.4% late ( that is after 7 days). The submandibular space predominated (n = 20, 60.6%) then the Space of Gilette (n = 5, 15.2%) Figure 2. Major source was odontogenic (n = 19, 57.6%) with 57.9%, 26.3% and 15.8% for the 1st, 2nd and 3rd mandibular molars, respectively, which tend to be extensive. We recorded a case that extended to the coccyx. Almost half had causally-associated co-morbidity (n = 15, 45.5%); predominantly anemia (n = 8, 24.2%) Figure 3.
Parenteral Ceftriazone/Augmentin with Metronidazole for 24-48 hours before surgical intervention of which 36.4% were delayed interventions. All had ICU management which improved outcome.
Morbidity and mortality rates were 24.2% and 15.2% respectively with the latter often following upper airway obstruction from Ludwig’s angina in 80% of which three-fifth expired before surgical intervention from late presentation.
Conclusion: Extensive deep neck space infections with their attendant morbidity and mortality remain frequent in our environment. Early diagnosis and early identification of predictors of outcome with prompt intervention are key to satisfactory results.
2.Introduction
3.Methods
4.Results
5.Discussion
6.Acknowledgement
6.Conclusion
8.References
Keywords
Deep Neck Space Infection; Scope; Dangers; Predictors of Outcome.
Introduction
Within the deep aspects of the neck are eleven spaces created by planes of greater and lesser resistance between the fascial layers. These spaces may be real or potential and may expand when pus separates these layers of fascia. These deep neck spaces communicate with each other, forming avenues by which infections may spread. These infections remain an important health problem with significant risks of morbidity and mortality [1].
These infections are challenging for the following reasons: (i). Complex anatomy: This makes precise localization of infections in this region difficult. (ii) Deep location: This makes clinical diagnosis of infections difficult because they are often covered by a substantial amount of unaffected superficial soft tissue. This may also make them difficult to palpate and impossible to visualize externally. (iii) Access: Normal superficial tissues must be crossed to gain surgical access to the deep neck spaces, placing all of the intervening neurovascular and soft tissue structures at risk of injury. (iv) Proximity: The deep neck spaces are surrounded by a network of vital structures that may become involved in the inflammatory process. Neural dysfunction, vascular erosion or thrombosis, and osteomyelitis with intracranial invasion are just a few of the potential sequelae that can occur. (v) Communication: The spaces are real/potential avenues of communication with each other. Infection in one space can spread to adjacent spaces, thus gaining access to increasingly larger portions of the neck [1].
The severity of infections of these anatomic spaces of the head and neck has been graded based on the level to which they threaten the airway or vital structures, such as the heart and mediastinum or cranial contents: the severity can either be low, moderate, or high [2]. A lower severity is noted when the infection does not threaten the airway. Infections of the spaces that can hinder access to the airway due to swelling or trismus are considered to be of moderate severity, while those that obstruct or deviate the airway or threaten vital structures are graded as severe [2].
The microbiology of deep neck infections usually reveals mixed aerobic and anaerobic organisms, often with a predominance of oral flora. Both gram-positive and gram-negative organisms may be cultured. A study by Asmar et al., [3] on the microbiology of retropharyngeal abscesses demonstrated polymicrobial culture results in almost 90% of patients. May et al., [4] found evidence that biofilm phenotypes may be associated with the pathogenesis of deep neck infections including the recalcitrance to treatment experienced with larger deep neck abscesses. Unfortunately, up to 20-50% of deep neck infections have no immediate identifiable source [5].
This study was therefore designed to share our experiences on the scope and dangers of this clinical entity, the import of early diagnosis, early identification of predictors of outcome with prompt intervention to the overall outcome.
Methods
This was a 4.5-year clinical analysis of all cases that met the inclusion criteria for deep neck space infections (DNSI). We utilised the standard codes contained in ICD-10 version 10 that met the inclusion criteria for DNSI for the retrieval of case files at the Health Record’s department for analysis from January 1, 2012- June 30, 2016. The study was conducted at the ORL-Head & Neck Surgery department of The Jos University Teaching Hospital, Plateau state, Nigeria with records from the Oral & Maxillofacial Surgery and General Surgery departments. Data were generated for analysis from demographic characteristics, clinical features, duration of symptoms before presentation, sources of infection, causally associated co-morbidity, imaging, diagnosis, treatment modality offered, time taken between hospital admission and surgical intervention, length of hospital stay and complications. Predictors of outcome were also identified in the cohort. Cases of superficial infections of the Head & Neck/dento-alveolar regions were excluded.
The data was subjected to statistical analysis using the SPSS version 16.0 Inc, Chicago, IL. Results were presented in simple descriptive, pictorial forms and figures.
The study observed the Helsinki Declaration.
Results
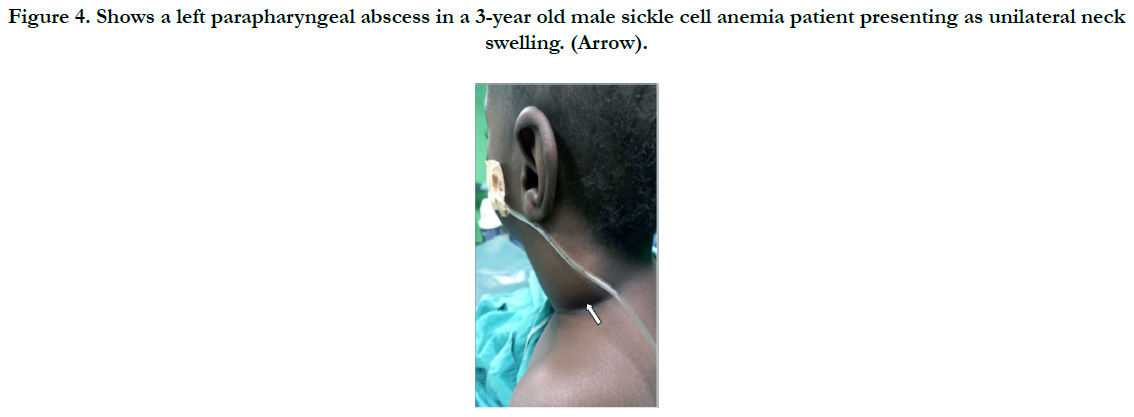
We analyzed 33 cases with deep neck space infections (DNSI) that met the inclusion criteria which constituted 1.1% of all institutional operations (2,899). There were 21 males and 12 females with a male to female ratio ( M:F ratio ) of 1.75:1. Their ages ranged from 6 months - 82 years, mean = 35.3 years ± 26.0, mode = 60 years and a median age of 37 years. DNSI was most prevalent in the age group 30-39 years (n = 8, 24.2%) Figure 1. Clinical presentation was majorly odynophagia, dysphagia, ‘hot potato’ speech, loss of voice in children, hoarseness, snoring, drooling, difficult breathing, neck stiffness, trismus, neck swellingcommonly unilateral with constitutional symptoms of fever, prostration and toxaemia in different combinations depending on the space(s) involved and severity. Analysis of duration of symptoms before presentation include: Twenty (60.6%) presented within ≤7 days of onset of symptoms, 6 (18.2%) between 1-2 weeks while 17 (21.2%) over two weeks. The range was 3-21 days, mean = 9.2 days while mean & mode coincided with 7days. The submandibular space (n = 20, 60.6%) as Ludwig’s angina or submandibular abscesses was the predominant space involved, trailed by the Space of Gilette-the retropharyngeal space/abscess(n=5, 15.2%) of which 60% were of the paediatric population, while parapharyngeal, the peritonsillar and multispace abscesses had a tie with 3(9.1%) each. Interestingly, all cases of multispace abscesses had causally associated co-morbidities Figure 2. The most frequent source was odontogenic (n = 19,57.6%) in the frequencies of 11( 57.9%), 5(26.3%) and 3(15.8%) for the 1st, 2nd and 3rd mandibular molars, respectively which tend to be extensive. The left to right (L:R) mandibular molar ratio was 13:6(2.2:1). We recorded two odontogenic sources that developed parapharyngeal abscesses that involved the upper chest in one while the second, to the coccyx. All were from carious mandibular 3rd molar. The second most common source was rhinosinusitis 5(15.2%) then pharyngotonsillitis with 3(9.1%) while 6(18.2%)) had no immediate identifiable source. Almost half- 15(45.5%) had causally associated co-morbidity 15(45.5%) with anemia 8(24.2%) predominating including a case of HbSS (Sickle cell anemia) Figure 4. Diabetes Mellitus came second with 5(15.2%) and the rest were isolated cases of HIV 1(3.0%) and chronic liver disease 1(3.0%) Figure 3.
Figure 4. Shows a left parapharyngeal abscess in a 3-year old male sickle cell anemia patient presenting as unilateral neck swelling. (Arrow).
We empirically utilised high dose parenteral Cefriazone/Augmentin (GlaxoSmithKline) with Metronidazole infusion for 24-48hours pre-operatively to subdue overwhelming sepsis before operation which sufficed for frank cases of Ludwig’s angina 3(9.1%). Update of antibiotics, which was infrequent, followed laboratory results of microscopy, culture and sensitivity.
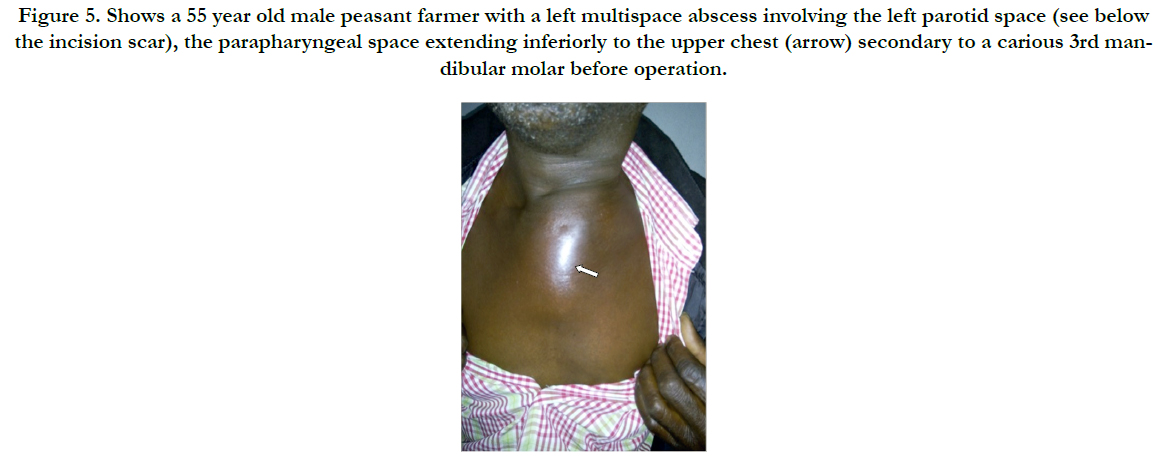
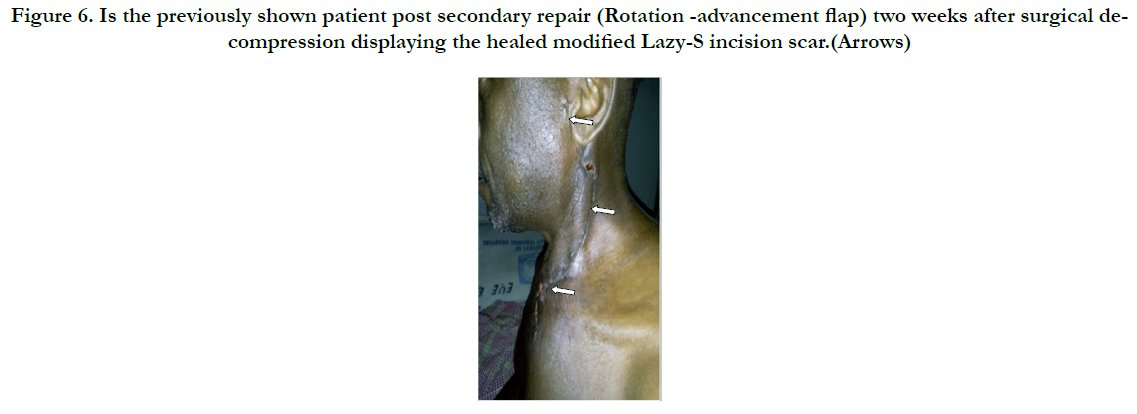
Surgical decompression was employed in 30(90.9%) after successfully securing the airway via endotracheal intubation. We utilised the Levitt approach for parapharyngeal abscesses whereas various modifications of the Lazy-S incision in scenarios of diverse space combinations yielded a satisfactory outcome as was observed in the patient with extension to the upper chest (Figure 5 & 6). He later had a secondary wound repair with a rotation-advancement flap. However, that with extension down to the coccyx had transcervical and posterior lumbo-sacral approaches (Figure 7 & 8) with an estimated 3L of pus drained. Internal drainage with cruciate mucosal incisions was reserved for retropharyngeal and peritonsillar abscesses (commonly under LA). All that were intubated were transferred to the Intensive Care Unit (ICU) with the cuffed endotracheal tube (CETT) in situ for ventilatory support and protection of the tracheo-bronchial tree for at least 24-48 hours. This enabled significant resolution of the effects of acute inflammatory changes from both the disease and the surgical manipulation before extubation. Tracheostomy was performed on two (6.1%).).
Figure 5. Shows a 55 year old male peasant farmer with a left multispace abscess involving the left parotid space (see below the incision scar), the parapharyngeal space extending inferiorly to the upper chest (arrow) secondary to a carious 3rd mandibular molar before operation.
Figure 6. Is the previously shown patient post secondary repair (Rotation -advancement flap) two weeks after surgical decompression displaying the healed modified Lazy-S incision scar.(Arrows)
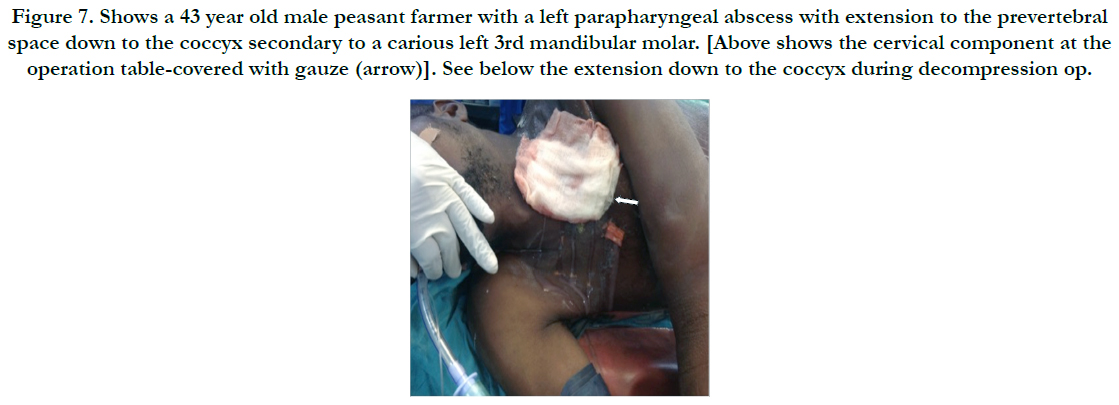
Figure 7. Shows a 43 year old male peasant farmer with a left parapharyngeal abscess with extension to the prevertebral space down to the coccyx secondary to a carious left 3rd mandibular molar. [Above shows the cervical component at the operation table-covered with gauze (arrow)]. See below the extension down to the coccyx during decompression op.
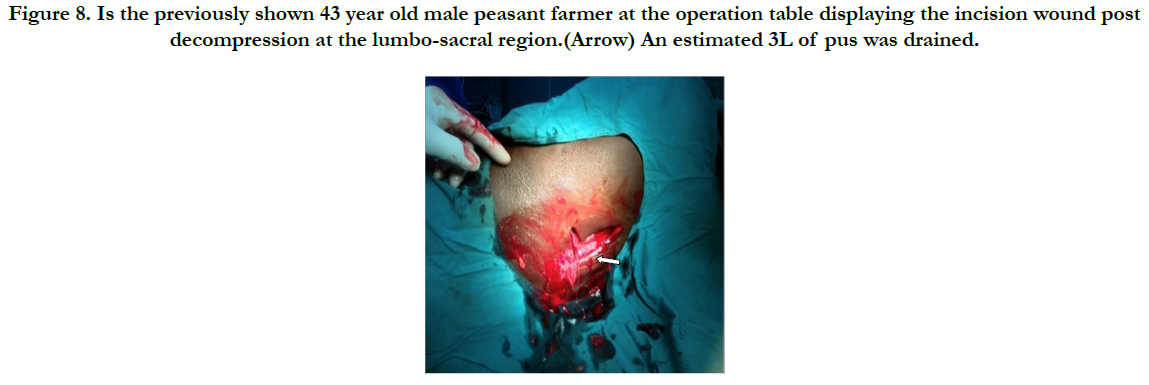
Figure 8. Is the previously shown 43 year old male peasant farmer at the operation table displaying the incision wound post decompression at the lumbo-sacral region.(Arrow) An estimated 3L of pus was drained.
Analysis of the time taken between hospital admission and intervention revealed that; 22(66.7%) had immediate intervention, 8(24.2%) within 1-2 days, while 2(6.1%) in 3-7days while another 2(6.1%) had surgical intervention after 7days. The range is from immediate-21days, with a mean = 1.5 days, mode/ median coincided at immediate intervention. The delays were a result of inter-departmental bureaucracy in referrals between the originating department and the ENT-Head & Neck surgery departments in situations where patients had not presented primarily to the ENT dept and inability of patient care givers to raise funds for investigations and surgery.
Length of hospital stay ranged from 1week-3months based largely on multiplicity of space involved, complications and presence of causally-associated co-morbid conditions which had multispecialty care.
This cohort recorded morbidity and mortality rates of 24.2% and 15.2% respectively with the latter often following upper airway obstruction from Ludwig’s angina in 80% save one, that was secondary to retropharyngeal abscess that succumbed during emergency tracheostomy. It is worthy to note that 60% expired before any form of intervention from delayed presentations.
Discussion
No accurate estimate of the overall prevalence of deep neck space infections worldwide presently exists. A study by Adil et al., [6] using information from the 2009 KIDS’ In patient database, estimated that there were more than 3,400 US hospital admissions annually for paediatric deep neck space infections. Our study recorded a prevalence of 1.1% amongst institutional operations in the general population which may be an underestimation owing to fact that a significant number of DNSI being deeply seated require capital intensive investigative tools such as CT scan for diagnosis; a large proportion of which are small that resolve spontaneously on systemic antibiotics. In a study by Aarthi et al., [7] on the role of Colour Doppler USG in the diagnosis of fascial space infections in a cross-sectional study that compared clinical evaluation on 25 patients recorded a sensitivity and specificity of 81.8% and 36.4% respectively with positive predictive value of 72.0% and Accuracy of 64.7%. In another study by Misurya et al., [8] who compared clinical examination versus ultrasonographic (USS) results, they concluded that the addition of USS increased the sensitivity and the specificity for the detection of abscesses; thus the diagnostic accuracy and proper treatment planning. In our series, most of our diagnoses were made clinically with occasional inputs from plain radiographs and USS which accounted for 24.2% despite the fact that it is among the cheapest imaging modality in our environment. This further confirms the degree of economic backwardness of Africa while the prohibitive cost of a CT scan compounds the situation. High resolution contrast CT scan has a superior tissue separation, vascular identification and abscess delineation [9, 10]. There was the predominance of the male gender in our study in ratio 1.75:1, this has similarly been observed by various researchers globally [11-13]. This has been attributed to several factors including cigarette smoking, betel nut chewing, tobacco chewing and poor oral hygiene amongst others [14, 15].
We found the involvement of all age groups in DNSI with an age-range of 6months-82years which was most prevalent in the 4th decade of life. This is in agreement with findings reported by various scholars [1,16, 17] which was attributed to diverse etiological factors from congenital, such as infected branchial cysts; to acquired conditions such as upper respiratory tract or dental infections. The presentations were mainly swellings of the cervico-facial region, grunting respiration, loss of voice in paediatric population, hoarseness, difficult breathing, snoring, sleep disturbance, odynophagia, dysphagia, trismus, torticolis, neck rigidity, in addition to constitutional symptoms of fever, cough and prostration in varying combinations which have also been reported [14, 18]. In this series, 60.6% presented within a week of onset of symptoms which was in agreement with a study [17] of which snoring and sleep disturbance were reported to be common features in early presenters [17]. However, these features are also seen in patients with obstructive adenotonsillar disease which is less acute; thus might have been responsible for the delayed presentation or referral to the specialist surgeon. This situation would require awareness campaigns to the general medical practitioners and the public on the dangers of delay with patients having these symptoms. We recorded 60.6% of the DNSI having involved the submandibular space as Ludwig’s angina/ submandibular abscesses which was in agreement with various reports [19, 20] but contrasts sharply with other scholars [11, 16] who recorded the parapharyngeal space as the predominant space involved. The involvement of the retropharyngeal space trailed a distant second with 15.2% in which 60% were of the paediatric population which had similarly been observed in a study [21]. However, a few researchers reported multispace abscess the most common [16, 17] which was commonly a feature observed in the adult population associated with causally-associated comorbidities which often carry a poor prognosis as observed in our series as well. The association of advanced age and diabetes mellitus with impaired immune response as a result of lazy leucocyte syndrome has been observed [11, 22].
The major source of DNSI in our cohort was odontogenic with 57.6%, which had been reported by various scholars [12-14] but at variance with a study [16]. Studies have incriminated the 3rd, 2nd and the 1st mandibular molars [23] in that order of frequencies, responsible, because the 3rd & 2nd molar roots get beyond the mylohyoid ridge. Odontogenic DNSI tends to rapidly spread to the submandibular space with subsequent communication with the parapharyngeal and the prevertebral spaces which extend from the skull base to the level of the hyoid and the coccyx, respectively. This further explains the extent of the two cases in our series which were all from carious 3rd molars. Coccygeal extension is an uncommon phenomenon as this serves the first reported case in world literature in the 21st century. In a separate study, the frequency of involvement of the mandibular molars was reversed [24] as observed in our series. For unknown reasons, the right mandibular molars were more affected than the left in a study [17] which was also reversed in our cohort with a left to right ratio of 2.2:1. For yet to be identified reasons, maxillary molars tend to be less frequent and less extensive as this study had a similar observation. However, gravity, in addition to the presence of less of the loose areolar tissues have been speculated to be responsible [17]. The frequent involvement of molars was attributed to the large uneven surfaces that promote lodgement of food debris, stagnation; coupled with their location far more posteriorly which tend to be more difficult to remove during brushing thus promoting cariogenesis [17]. Furthermore, odontogenic DNSI are able to spread rapidly because these spaces being real/potential or contain loose areolar tissue make the organisms commonly implicated in pus formation such as Staphylococcus aureus which produces the coagulase enzyme; while the Streptococcal bacteria that produces the streptodornases, streptokinases and hyaluronidases promote the rapid breakdown of tissues barriers [25, 26]. We are of the opinion that there may be more to the extensive effect of enzymatic elaboration of staphylococcus aureus and streptococcus spp in odontogenic deep neck space abscesses that make them more aggressive/extensive than other sources. There is the need for more research in this field to unravel the origin of this phenomenon.
We found in this cohort 18.2% had no immediate identifiable sources, which has also been reported [5] though was a major culprit in a study [16]. We speculate that the cases with no immediate identifiable source may likely follow rhinosinusitis which every individual suffer several episodes yearly [16] which might either be covert or overt. The lymphatics of the nose and the sinuses drain majorly into the upper deep cervical group of lymph nodes and the retropharyngeal (Space of Gilette) group of lymph nodes as the 1st echelon nodes in which exists a prominent node termed, the Node of Rouviere which has been found the major culprit in retropharyngeal abscesses and early cancer spread in Sinonasal and Middle ear cleft malignancies [27] taking into account that all these spaces communicate with each other.
Almost half of the cases (45.5%) in this series had causally associated co-morbidity with anemia predominating in 24.2% including a case of sickle cell anemia in a 3-year old male child. Sickle cell anemia patients have been found to be incapable of mounting immune resistance against capsulated organisms such as the pneumococcal organisms due to defective opsonin [28]. Anemia, which eventually influences outcome has been reported the most frequent causally associated comorbidity in a study with 75.5% [19]. A study in China [20] identified a leucocytosis of ≥15 x 109 and a neutrophilia of ≥ 85% being among important predictors of outcome in DNSI. Furthermore, other causally associated co-morbid situations such as Diabetes Mellitus (DM) have been reported by various researchers as the most frequent related with poor outcome [19, 22, 23] but trailed behind anemia in our study with 15.2%.
In this study, we utilised high dose parenteral Augmentin/Cefriaxone and Metronidazole infusion empirically which were updated after Culture and Sensitivity results. This combination has been reported by various scholars with good results [17-19] with no need for switch over. However, a study reported a satisfactory outcome with Moxifloxacin [29] compared with Clindamycin/ Ceftriaxone combination in a randomised clinical trial. The ability of the managing clinician to distinguish between a phlegmon/ cellulitis on one hand, whose treatment is basically antibiotics and on the other; abscess collection which requires both medical and surgical decompression is critical for a successful outcome [1].
Our study employed surgical intervention in 90.9% under GA via oro-tracheal intubation along with extraction of the offending tooth/treatment of the primary source which was in agreement with a study [17] which was at variance with 42.1%% in a study [19]. Drainage under LA was left for submandibular and peritonsillar abscesses that did not threaten the airway. The Levitt approach was utilised for external open drainage of parapharyngeal abscesses. However, various modifications of the lazy-S incision was applied in scenarios of different space combinations in addition as observed in that that had extension to the upper chest who subsequently had a rotation-advancement flap repair for secondary wound closure while with extension to the coccyx had multiple approaches with a satisfactory outcome. We employed the cruciate incision for internal drainage for cases of peritonsillar and retropharyngeal abscesses with good outcome. Emergency tracheostomy was performed on 2 (6.1%) from upper airway obstruction in which one, a case of retropharyngeal abscess succumbed during the procedure. High mortality from emergency tracheostomy secondary to precarious airway has been reported [30]. In our cohort, we were able to successfully secure the airway via intubation in 90.6%. All had ventilatory support in the intensive care unit (ICU) for 24- 48 hours to allow for significant resolution of airway edema from inflammatory changes from both the disease and surgical manipulation including the protection of the tracheobronchial tree. Upper airway shutdown is the commonest cause of mortality in DNSI. Studies have shown that strict airway management from a well equipped/staffed ICU improved prognosis [19].
The overall hospital stay in this cohort was 7-90 days which is in agreement with a study [20] that had 2-90 days, this was attributed to the high level of those with co-morbidity and complications as was recorded in this study. However, studies from some researchers recorded shorter hospital stay [14-16] which was largely because their study population was paediatric who tend to have less causally associated co-morbidity thus less complications [16].
We recorded a morbidity of 24.2% which is significant taking into account the percentage of those who presented after 7 days (39.4%) vis a vis the 33.3% that had delayed surgical intervention for various reasons. This explains the presence in our cohort cases with extensive disease which may have been responsible for the high mortality of 15.2% in which 80% were secondary to upper airway obstruction from Ludwig’s angina. Furthermore, it is worth noting that 60% of the mortalities occurred before any form of surgical intervention from delayed presentation. This would require more community awareness on the need for early hospital presentation in cases of dental caries, unexplained sudden neck swellings, snoring or noisy breathing. There is also the need for improved interdepartmental referral system for effective transfer of patient care. This mortality is in contrast with reports by other scholars [14, 16, 29]. High mortality is a common occurrence in DNSI because of airway compromise that makes both tracheostomy and endotracheal intubation a precarious adventure. Ludwig’s angina, the term coined in 1836 by the German Physician, Wilhelm Friedrich von Ludwig, has been implicated because of the rapidity in progression which often leads to upper airway shutdown with lethal consequences especially with late presentation and identification [31].
Acknowledgement
We appreciate staff of the Health record’s department for the painstaking effort in case file retrievals which made this study possible. Our sincere thanks also go to Mr Innocent Amanum, The Office Assistant, Dept of Oral and Maxillofacial surgery for the wonderful secretarial work and diagram design.
Conclusion
Extensive deep neck space infections; with their attendant morbidity and mortality remain frequent in our environment. Early diagnosis and early identification of predictors of outcome with prompt intervention are key to satisfactory results.
References
- Murray AD, Talavera F, Tewfik TL, Meyers AD, Marcincuk MC, et al., (2016) Deep Neck Infections. Medscape.
- Flynn Thomas R (2004) Principles of management of odontogenic infections. Peterson's principle of oral and maxillofacial surgery. (2nd edn), BC decker Inc, Hamilton, london. 277–293.
- Asmar BI (1990) Bacteriology of retropharyngeal abscess in children. Pediatr Infect Dis J. 9(8): 595-7.
- May JG, Shah P, Sachdeva L, Micale M, Kruper GJ, et al., (2014) Potential role of biofilms in deep cervical abscess. Int J Pediatr Otorhinolaryngol. 78(1): 10-3.
- Adekeye EO, Adekeye JO (1982) The pathogenesis and microbiology of idiopathic cervicofacial abscesses. J Oral Maxillofac Surg. 40(2): 100–106.
- Adil E, Tarshish Y, Roberson D, Jang J, Licameli G, et al., (2015) The Public Health Impact of Pediatric Deep Neck Space Infections. Otolaryngol Head Neck Surg. 153(6): 1036-41.
- Aarthi NV, Parthiban J, Santana N, Giridhar AG, Yashoda DBK, et al., (2013) The Role of Colour Doppler USG in the Diagnosis of Fascial Space infections: A Cross-Sectional Study. J Clin Diagn Res. 7(5): 962-967.
- Misurya RVK, Lange AH, Dev S (2007) Role of ultrasonography in diagnosis and treatment planning of fascial space infections of head and neck region. Int J Oral Maxillofac Surg. 36(11): 1040–1041.
- Mayor GP, Millan JMS, Martinez VA (2001) Is conservative treatment of deep neck space infections appropriate? J Head Neck. 23(2): 126–33.
- Smith JL, Hsu JM, Chang J (2006) Predicting deep neck space abscess using computed tomography. Am J Otolaryngology. 27(4): 244–7.
- Lee YQ, Kanaqalinqam J (2011) Deep neck abscesses: the Singapore experience. Eur Arch Ortorhinolaryngol. 268(4): 609-14.
- Parhiscar A, Harel G (2001) Deep neck abscess: A retrospective review of 210 cases. Ann Otol Rhinol Laryngol. 110(11): 1051–54.
- Sethi DS, Stanley RE (1994) Deep neck abscesses: Challenging trends. J Laryngol Otol. 108(2): 138-43.
- Katara G, Saxena A, Bhagat S, Singh B, Kaur M, et al., (2015) Deep Neck Space Infections: A Study of 76 Cases. Iran J Otorhinolaryngol. 27(81): 293–299.
- Mathew GC, Ranganathan LK, Gandhi S, Jacob ME, Singh I, et al., (2012) Odontogenic maxillofacial space infections at a tertiary care center in North India: a five-year retrospective study. Int J Infect Dis. 16(4): e296–e302.
- Yang W, Hu L, Wang Z, Nie G, Li X, et al., (2015) Deep Neck Infection: A Review of 130 Cases in Southern China. Medicine(Baltimore). 94(27): e994.
- Ibiyemi ST, Okojie-Adesomoju VN, Dada-Adegbola HO, Arotiba JT (2014) Pattern of Orofacial Bacterial Infections in a Tertiary Hospital in SouthWest Nigeria. J West Afr Coll Surg. 4(4): 112-141.
- Cmejrek RC, Coticchia JM, Arnold JE (2002) Presentation, Diagnosis and Management of Deep Neck Space abscesses in infants. Arch Otolaryngol Head Neck Surg. 128(12): 1361-1364.
- Fomete B, Agbara R,Osunde DO, Ononiwu CN (2015) Cervicofacial Infection in a Nigerian tertiary health institution: A retrospective Analysis of 77 cases. J Korean Assoc Oral Maxillofac Surg. 41(6): 293-298.
- Han X, An J, Zang Y, Gong X, He Y.(2016) Risk Factors For Life-Threatening Complications of Maxillofacial Infections. J Craniofac Surg. 27(2): 385-390.
- Coticchia JM, Getnick GS, Yun RD, Arnold JE (2004) Age-, site-, and timespecific differences in pediatric deep neck abscesses. Arch Otolaryngol Head Neck Surg. 130(2): 201-7.
- Huang TT, Tseng HY, Liu TC, Hsu CJ, Chen YS (2005) Deep Neck Infection in Diabetic patients: Comparison of clinical picture and outcome with Non-Diabetic patients. Otolaryngol Head Neck Surg. 132(6): 943-7.
- Fasting NS, Saikrishna D, Kumar GSV, Shetty SK, Rao MR (2014) Detection of bacterial flora in orofacial space infections and their antibiotic sensitivity profile. J Maxillofac Oral Surg. 13(4): 525-532.
- Marion G, Rinaldi R, Marchese-Raqone R, Saia G Bertolin A, Dalborqo R, et al., (2008) Deep Neck Infection with dental origin: Analysis of 85 consecutive cases (2000-2006). Acta Otolaryngol. 128(2): 201-6.
- Walia IS, Borle RM, Mehendiratta D, Yadav OA (2014) Microbiology and Antibiotic sensitivity of Head & Neck space infections of odontogenic origin. J Maxillofac Oral Surg. 13(1): 16-21.
- Kariyama T, Karasawa T, Nakagawa K, Saiki Y, Yamamoto E, et al., (2000) Bacteriologic features and Antimicrobial susceptibility in isolates from orofacial odontogenic infections. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 90(5): 600-8.
- John Groves (1985) A synopsis of Otolaryngology, (4th Edn), Wright Publ, Bristol. Chapter 21: 111, 60: 297.
- Makani J, Ofori-Acquah SF, Nnodu O, Wonkam A, Ohene-Frempong K (2013) Sickle cell disease: New Opportunities & Challenges in Africa. Scientific World J. 2013: 193252.
- Gomez-Arambula H, Hidalgo-Hurtado A, Rodriguez-Flores R, Ana-Maria, Gonzalez Amaro, et al., (2015) Moxifloxacin Vs Clindamycin/Ceftriaxone combination in the management of odontogenic maxillofacial infectious processes: A preliminary intrahospital controlled clinical trial. J Clin Exp Dent. 7(5): e634-e639.
- Kokong DD, Yaro PJ, Embu HY, Nimkur LT, Iduh AA, et al., (2016) Tracheostomy: The dynamics of Indications in an Old procedure. J Otolaryngol Rhinol. 2(2): 1-5.
- Candamourthy R, Venkatachalam S, Babu MRR, Kumar GS (2012) Ludwig Angina-An emergency: A Case Report with Literature Review. J Nat Sci Biol Med. 3(2): 206-208.