Pharmacological Properties of Capparis spinosa Linn
Moufid A, Farid O, M. Eddouks
Faculty of Sciences and Techniques Errachidia, Moulay Ismail University, Morocco.
*Corresponding Author
M. Eddouks,
Professor,
Faculty of Sciences and Techniques Errachidia,
Moulay Ismail University,
BP 21, Errachidia, 52000, Morocco.
Tel: +212535574497
Fax: +212535574485
E-mail: mohamed.eddouks@laposte.net
Article Type: Review Article
Received: May 21, 2015; Accepted: June 16, 2015; Published: June 19, 2015
Citation: Moufid A, Farid O, M. Eddouks (2015) Pharmacological Properties of Capparis Spinosa Linn. Int J Diabetol Vasc Dis Res, 3(5) 99-104. doi: dx.doi.org/10.19070/2328-353X-1500020
Copyright: M. Eddouks© 2015. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Capparis spinosa Linn. (Capparaceae) was traditionally used for pharmacological purposes and has potential for use in modern cosmetics. This review aims to assess the current available knowledge of Capparis spinosa and its constituents for management of several diseases. Bibliographic investigation was carried out by scrutinizing classical text books and peer reviewed papers, consulting worldwide accepted scientific databases (SCOPUS, PUBMED, SCIENCE DIRECT, Google Scholar) to retrieve available published literature. Only articles on pharmacological and phytochemical studies of Capparis spinosa have been selected. The main pharmacological activities of caper are antidiabetic, antihypertensive, hypolipidemic, antioxidant, antimutagenic, anti-allergic, hepatoprotective, antibacterial, antiviral, antifungal, immunomodulatory, anti-apoptotic and anti-inflammatory activities. Phytochemicals studies of this plant revealed the existence of many bioactive components such as the saccharides and glycosides, flavonoids, alkaloids, terpenoids and volatile oils, fatty acids, vitamin C, vitamin Eand steroids. We conclude that Capparis spinosa is beneficial for the treatment of many pathologies supporting then its use in human nutrition and healthcare.
2.Introduction
3.Methods
4.Description and Ecology
5.Anti Diabetic and Hypolipidemic Activities
6.Cardiovascular Activty
7.Antioxidant, Anti-Allergic and Anti-Inflammatory Activities
8.Antimicrobial Acivity
9.Hepatoprotective Effect
10.Phytochemistry
11.Conclusion
12.Acknowledgment and Grants
13.References
Key Words
Capparis Spinosa; Ethnopharmacology; Phytochemistry; Pharmacological Activities.
Introduction
Traditional medicine is very important for treating many pathologies such as cancer, diabetes, hypertension, microbial and viral contamination, AIDS, allergy and others. The all genus of Capparis have been used extensively in folk medicine by many cultures since ancient times, especially in all countries of the Mediterranean basin (Morocco, Spain, Tunisia, Italy, Turkey) and in Western as well as Central Asia [1]. The relationship between capers and human beings can be traced back to the Stone Age. Remains of Capparis spinosa Linn. (Capparaceae family) were unearthed in archaeological sites as early as the lower Mesolithic (9500–9000b.p.) [2]. Recently plant remains of Capparis spinosa (C. spinosa) have been discovered for the first time in China and the eastern part of Central Asia supporting the use of caper for medicinal purposes from 2800 years b.p [3]. Floral buds of caper are commonly used in the Mediterranean cuisine as flavoring for meat and other foods. Caper has been used as a medicinal plant with many beneficial properties. This review aims to assess the current available knowledge of C. spinosa and its phytochemical compounds for management of several diseases.
Methods
Bibliographic investigation was carried out by scrutinizing classical text books and peer reviewed papers, consulting worldwide accepted scientific databases (SCOPUS, PUBMED, SCIENCE DIRECT, Google Scholar) to retrieve available published literature. Only articles on pharmacological and phytochemical studies of C. spinosa have been selected. Overall, 100 articles were reviewed and 63 articles were selected for the study.
Description and Ecology
Members of the Capparaceae are trees and shrubs. This family had a medium size with 40-45 genera and approximately 700-900 species and are distributed in warm and tropical, sometimes very arid, regions of both hemispheres of the world [4]. C. spinosa has a great economic, cosmetic and medicinal importance. This plant has developed special mechanisms in order to survive in the semiarid lands conditions and consequently its introduction may help to prevent the disruption of the equilibrium of those fragile ecosystems and the soil degradation. Caper grows spontaneously on the steppes of northern Africa and Mediterranean countries, but recently caper is cultivated and exported. Morocco is the main producer and the first exporter of the caper fruits in the world.
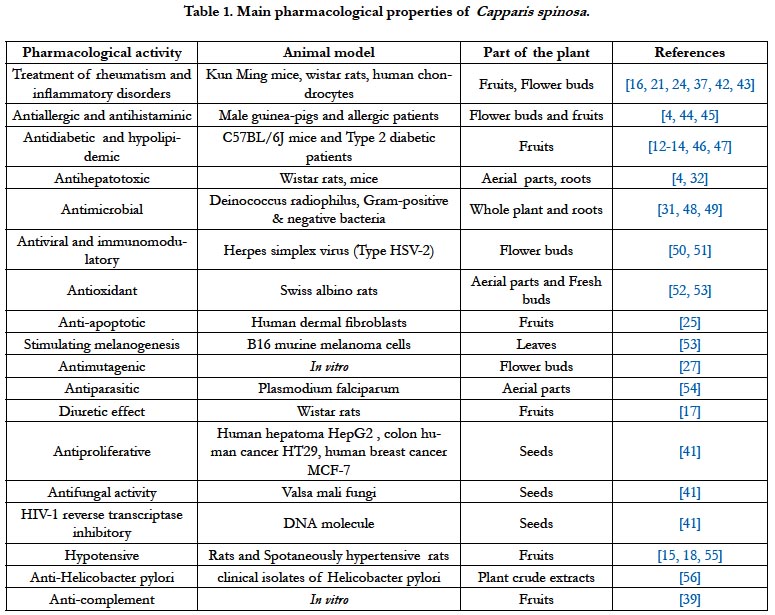
Several studies have reported that C. spinosa possess several pharmacological activities including anti-inflammatory, anti-allergic, antidiabetic, hypolipidemic, hepatoprotective, antimicrobial, antiviral, immunomodulatory, antioxidant, anti-apoptotic, stimulating melanogenesis, antimutagenic, antiparasitic, antihypertensive, antiproliferative, antifungal, anti-HIV, anti- Helicobacter pylori and anti-complement effects (Table 1).
Anti Diabetic and Hypolipidemic Activities
In the folk medicine many plants are used to control diabetes [5-7]. C. spinosa had a large importance for the treatment of some diseases such as diabetes. Although insulin treatment and other chemical therapies can control many aspects of diabetes, numerous complications and undesirable effects such vascular dysfunctions, nephropathy, neuropathy and retinopathy appear when the disease is not well controlled [8-10]. Some studies have reported the potent hypoglycaemic effect of C. spinosa [7]. In Morocco this plant is traditionally used in diabetes control and treatment and is considered as the most commonly aromatic in the Moroccan kitchen [6, 11].
We have previously demonstrated that the aqueous extract of C. spinosa decreased blood glucose, plasma triglycerides and plasma cholesterol levels in both normal and diabetic rats after oral and repeated administration [12]. The results showed that this plant extract had a clear preventive effect on the hypertriglyceridemia related to development of insulin resistance and glucose intolerance. In addition, we have demonstrated the potent hypolipidemic and anti-obesity effects of caper in high fat diet mice as well as the beneficial effect on insulin resistance [13]. Recently a controlled human study has determined the efficacy of C. spinosa in the treatment of hyperglycemia in Type 2 diabetic patients. Interestingly no liver, kidney and other side effects were observed in these patients when treated by 400 mg caper fruit extract three times a dayfor two months [14].
Cardiovascular Activty
Hypertension is a serious public health problem worldwide; more than 20% of the world’s population suffers from high blood pressure. Among the new choices for preventing or treating these disorders the natural products isolated from medicinal plants are taking an important place. C. spinosa is widely used in the folk medicine for its diuretic and antihypertensive effects [6, 15, 16] and vasorelaxant effect [15]. These studies have demonstrated the beneficial effect of C. spinosa causing a significant reduction in arterial blood pressure by acting on heart rate, the Nitric oxide synthesis pathway as well as the angiotensine converting enzyme inhibition. In addition, we have reported the furosemide-like effect of C. spinosa in normal and anaesthetized rats and a significant increase of glomerular filtration rate, urinary volume and electrolyte output without modifications in the plasma angiotensin converting enzyme and renin activities [17, 18]. These studies support the beneficial role of caper in the management of hypertension.
Antioxidant, Anti-Allergic and Anti-Inflammatory Activities
Numerous investigations support the relationship between the Mediterranean diet and low incidence of several diseases, such as cardiovascular diseases and cancer; and they recommended an increase of the consumption of fruits and vegetables. Some plants are known in phytomedicine around the world as anti-oxidative and contain often wide variety of antioxidant molecules, such as phenolic acids, flavonoids and other natural antioxidants distributed in different parts of the plants. Several compounds have been identified in C. spinosa including flavonoids, saponins, tannins and tocopherol. These flavonoids display a remarkable role in various pharmacological activities including anti-allergic, antiinflammatory and antioxidant effects. The important presence of alpha tocopherol in the caper parts makes a new choice for protecting cell membranes from oxidative damage [19, 20]. Cappariside is one of new antioxidant isolated from the C. spinosa fruit [19]; this cappariside (4-hydroxy-5-methylfuran-3-carboxylic acid) can be used against multiple types of cancer cells [21]. Similarly more than eleven organic acids isolated from C. spinosa possess similar beneficial effects. Recently, a study has revealed that an N-butanol extract of caper induced apoptosis through mitochondrial pathway [22]. In addition, hydro-ethanolic extract obtained from C. spinosa leaves has been demonstrated to exhibit antioxidant activity in both chemical and biological tests [23]. In Male Chinese Kun Ming mice, an acute inflammation in mice hind paws by subcutaneously injection of Carrageenan has been induced, and the anti-inflammatory effects of C. spinosa aqueous extract was evaluated with calculating of the paw edema “or inhibition rate” [23]. After oral administration C. spinosa extract induced a significant anti-inflammatory activity against carrageenan-induced edema. Additionally, the methanolic extract of the flowering buds of C. spinosa possess a chondroprotective effect on human articular chondrocytes. These results could be explained by the high density of flavonoids in methanolic extract of the flowering buds of C. spinosa which play an anti-apoptotic or cytoprotective action and prevented cell death caused by reactive oxidative substances (ROS). These flavonoids not only play a direct role as ROS scavengers but also inhibit the enzymes involved in the production of these oxidative substances protecting the DNA against the toxic effects of these compounds [16]. In a recent study the significant inhibitory effect of stachydrine isolated from C. spinosa has been demonstrated to be similar to a standard analgesic and antiinflammatory drug; Diclofenac sodium [24]. This study has demonstrated experimentally that C. spinosa extract possess an antirheumatoid and anti-arthritic effect, reduced tissue swelling and decreased the edema in both Carrageenan induced paw oedema and Xylene induced ears edema in Male wistar rats. Moreover the ethanolic extract of C. spinosa has been demonstrated to protect against apoptosis and cell death induced by oxygenic water H2O2 in normal and systemic sclerosis fibroblasts and reduced the levels of reactive oxygen species in systemic sclerosis fibroblasts [25]. Furthermore C. spinosa extract has been shown to stimulate melanogenesis in B16 murine melanoma cells, and the increase in the melanin was accompanied by an upregulated expression of tyrosinase [26]. In another study, the genotoxic and antimutagenic effects of caper flower buds aqueous extract on the Allium cepa L root tip meristem cells have been demonstrated [27]. The results of this study suggest that C. spinosa buds aqueous extract is nongenotoxic and reveals that this extract has antimutagenic potential against ethyl methane sulfonate induced chromosomial and mitotic aberrations in terms of lagging chromosomes. Moreover, the determination of the protective effects was evaluated against oleaceae antigen challenge induced and histamine-induced bronchospasm in male guinea pigs, and the histamine skin prick test was performed on human volunteers. The methanolic fraction of C. spinosa has been demonstrated to exert an inhibitory effect against histamine-induced skin erythema on humans and a good protective effect against the bronchospasm induced by antigenchallenge in sensitized guinea-pigs [28].
Antimicrobial Acivity
The antimicrobial activity of C. spinosa aqueous extract has been tested against some dermatophytes, this extract prevented completely the growth of two species of fungi: Misrosporum canis and Trichophyton violaceum [29]. On the other hand the petroleum ether and hexanic extracts of C. spinosa aerial parts showed a similar activity of inhibition against both gram-positive and gram-negative bacterial species tested such as Helicobacter pylori, Escherichia coli and Bacillus cereus [30, 31].
Hepatoprotective Effect
An important compound named p-Methoxy benzoic acid has been isolated from an aqueous extract of C. spinosa and its antihepatotoxic activity has been evaluated against hepatotoxicities in wistar rats after intraperitonial injection of the carbontetrachloride CCl4 and paracetamol successively [32]. This compound prevented toxic effects of both CCl4 and paracetamol in hepatocytesboth in vivo and in vitro tests. These results are comparable to the known antihepatotoxic agent named Silymarin [32, 4].
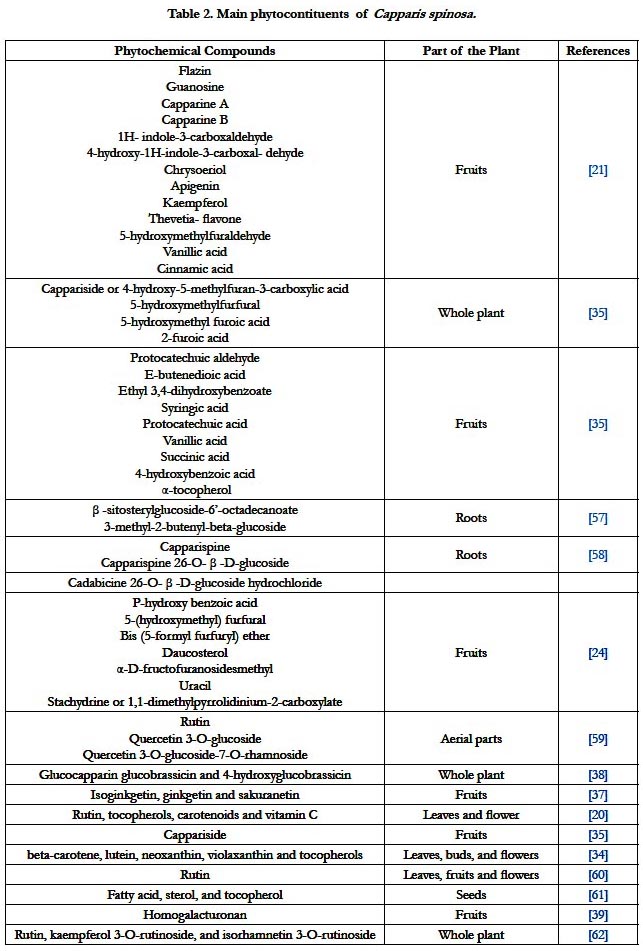
Phytochemistry
Several phytochemical studies have tried to elucidate the phytocompounds of C. spinosa. The constituents of C. spinosa include the saccharides and glycosides, flavonoids, alkaloids, terpenoids and volatile oils, fatty acids and steroids [33]. The major compounds found in C. spinosa are flavonoids, indoles, and phenolic acids [21]. Interestingly, a study has revealed the presence of both α- and γ-tocopherol in buds of C. spinosa as well as an appreciable level of vitamin C [20]. Lutein and Violaxanthin are the main carotenoids and the principal form of tocopherol detected in leaves was alpha-tocopherol [34]. Other investigations have demonstrated that C. spinosa contains in large amounts many secondary metabolites like flavonoids (rutin, quercetin, quercetin-3- rutinoside, kaempferol-3-rutinoside) phenloic components, different alkaloids (capparispine, capparispine 26-O-β-d-glucoside and cadabicine 26-O-β-d- glucoside hydrochloride) and organic acids [21, 35]. Caper infusion has identified rutin, kaempferol 3-O-rutinoside, and isorhamnetin 3-O-rutinoside as dominant flavonoids in addition to isoginkgetin, ginkgetin and sakuranetin which have demonstrated to exert an anti-inflammatory activity by inhibiting the nuclear factor-kappa B [36, 37]. Moreover, the different glucosinolates found in C. spinosa were glucocapparin, mercaptoglucocapparin, 4-hydroxyglucobrassicin and glucobrassicin and glycinyl-glucocapparin [38]. In addition, a homogalacturonan polysaccharide was obtained from the fruits of C. spinosa and had a strong inhibitory effect on the activation of complement system through the classic pathway than that of heparin [39]. A recent study has revealed the presence of high levels of oleic acid and α-Tocopherol in C. spinosa seeds [40]. Finally, a lectin has been isolated from C. spinosa and potently inhibited HIV-1 reverse transcriptase and proliferation of both hepatoma HepG2 and breast cancer MCF-7 cells [41]. Table 2 summerizes the known phytochemial components of C. spinosa.
Conclusion
This review summarizes the main pharmacological activities of C. spinosa including antidiabetic, antihypertensive, hypolipidemic, antioxidant, antimutagenic, anti-allergic, hepatoprotective antibacterial, antiviral, antifungal, immunomodulatory, anti-apoptotic and anti-inflammatory effects. These beneficial effects support the use of this plant for human healthcare from ancient time. However, the underlying mechanisms of these pharmacological activities are not totally elucidated and few clinical trials have been performed until now. In addition, few data are available about the safety of C. spinosa. Moreover, the phytochemical studies revealed clearly that the main constituents of this plant are the saccharides nd glycosides, flavonoids, alkaloids, terpenoids and volatile oils, fatty acids, vitamin C, vitamin E and steroids. More studies dealing with the relationship between the active principle and pharmacologicalactivity are still needed to develop new complementary active principles able to enrich the therapeutical arsenal especially for treatment of chronic diseases like diabetes and cardiovascular disorders.
Acknowledgment and Grants
This work was supported by the CNRST under the grant number PPR/2015/35.
References
- Rivera D, Alcaraz F (2003) Review of food and medicinal uses of Capparis L. Subgenus Capparis (Capparidaceae). Economic Botany 57(4): 515-534.
- Hansen JM (1991) The Palaeoethnobotany of Franchthi Cave. Indiana University Press, Bloomington. 119: 38–39.
- Jiang HE, Li X, Ferguson DK, Wang YF, Liu CJ, et al. (2007) The discovery of Capparis spinosa L. (Capparidaceae) in the Yanghai Tombs (2800 years b.p.), NW China, and its medicinal implications. J Ethnopharmacol 113: 409–420.
- Aghel N, Rashidi I, Mombeini A (2007) Hepatoprotective activity of Capparis spinosa root bark against CCl4 induced hepatic damage in Mice. Iranian Journal of Pharmaceutical Research 6(4): 285-290.
- Ouahidi ML, Khalidi A, Lemhadri A, El-Hessni A, El-Bouhali B, et al. (2009) Medicinal plants used for the treatment of diabetes mellitus in North-Western region of Morocco (Gharb): An ethnopharmacological approach. Advances in Phytotherapy Research, Research Signpost. 129-138.
- Eddouks M, Maghrani M, Lemhadri A, Ouahidi ML, Jouad H (2002) Ethnopharmacological survey of medicinal plants used for the treatment of diabetes mellitus, hypertension and cardiac diseases in the south-east region of Morocco (Tafilalet). J Ethnopharmacol 82(2-3): 97-103.
- Farooq Z, Iqbal Z, Mushtaq S, Muhammad G, Iqbal MZ, Arshad M (2008) Ethnoveterinary practices for the treatment of parasitic diseases in livestock in Cholistan desert (Pakistan). J Ethnopharmacol 118(2): 213-219.
- De Souza CR, Zanetti ML (2001) The use of disposable syringes in the administration of insulin at home. Rev Lat Am Enfermagem 9(1): 39-45.
- Baynes C, Anyaoku V, Johnson DG, Elkeles RS (1991) Feedback inhibition of insulin secretion in type 2 diabetes. Clin Sci 81(5): 685-690.
- Bodmer M, Meier C, Stephan K, Jick S, Meier CR (2008) Metformin, Sulfonylureas, or other Antidiabetes Drugs and the Risk of Lactic Acidosis or Hypoglycemia: a nested case-control analysis. Diabetes Care 31(11): 2086–2091.
- Jouad H, Haloui M, Rhiouani H, EI-Hilaly J, Eddouks M (2001) Ethnobotanical survey of medicinal plants used for the treatment of diabetes, cardiac and renal diseases in the North centre region of Morocco (Fez–Boulemane). J Ethnopharmacol 77(2-3): 175–182.
- Eddouks M, Lemhadri A, Michel JB (2004) Caraway and caper: potential anti-hyperglycaemic plants in diabetic rats. J Ethnopharmacol 94(1): 143-148.
- Lemhadri A, Eddouks M, Sulpice T, Burcelin R (2007) Anti-hyperglycaemic and Anti-obesity Effects of Capparis spinosa and Chamaemelum nobile Aqueous Extracts in HFD Mice. American Journal of Pharmacology and Toxicology 2(3): 106-110.
- Huseini HF, Hasani-Rnjbar S, Nayebi N, Heshmat R, Sigaroodi FK, et al. (2013) Capparis spinosa L. (Caper) fruit extract in treatment of type 2 diabetic patients: a randomized double-blind placebo-controlled clinical trial. Complement Ther Med 21(5): 447-452.
- Zaggwagh NA, Michel JB, Eddouks M (2007) Cardiovascular Effect of Capparis spinosa Aqueous Extract. Part VI: in vitro Vasorelaxant Effect. American Journal of Pharmacology and Toxicology 2(3): 135-139.
- Panico AM, Cardile V, Garufi F, Puglia C, Bonina F, et al. (2005) Protective effect of Capparis spinosa on chondrocytes. Life Sci 77(20): 2479–2488.
- Moufid A, Khalidi A, Zeggwagh NA, Michel JB, Eddouks M (2009) Furosemide-like effect of Capparis spinosa. Advances in Phytotherapy Research, Research Signpost. 213-221.
- Khaldi A, Zeggwagh NA, Michel JB, Eddouks M (2009) Effect of Capparis spinosa in spontaneously hypertensive rats (SHR). Advances in Phytotherapy Research, Research Signpost. 201-211.
- Wiese S, Wubshet SG, Nielsen J, Staerk D (2013) Coupling HPLC-SPE-NMR with a microplate-based high-resolution antioxidant assay for efficient analysis of antioxidants in food--validation and proof-of-concept study with caper buds. Food Chem 141(4): 4010-4018.
- Tlili N, Khaldi A, Triki S, Munné-Bosch S (2010) Phenolic compounds and vitamin antioxidants of caper (Capparis spinosa). Plant Foods Hum Nutr 65(3): 260-265.
- Zhou H, Jian R, Kang J, Huang X, Li Y, et al. (2010) Anti-inflammatory effects of Caper (Capparis spinosa L.) fruit aqueous extract and the isolation of main phytochemicals. J Agric Food Chem 58(24): 12717-12721.
- Ji YB, Yu L (2014) N-butanol extract of Capparis spinosa L. induces apoptosis primarily through a mitochondrial pathway involving mPTP open, cytochrome C release and caspase activation. Asian Pac J Cancer Prev 15(21): 9153-9157.
- Mansour RB, Jilani IB, Bouaziz M, Gargouri B, Elloumi N, et al. (2014) Phenolic contents and antioxidant activity of ethanolic extract of Capparis spinosa. Cytotechnology 1-8.
- Feng X, Lu J, Xin H, Zhang L, Wang Y, et al. (2011) Anti-arthritic Active of Capparis spinosa L. Fruits and its Chemical Constituents. Yakugaku Zasshi 131(3): 423-429.
- Cao YL, Li X, Zheng M (2010) Capparis spinosa protects against oxidative stress in systemic sclerosis dermal fibroblasts. Arch Dermatol Res 302(5): 349–355.
- Matsuyama K, Villareal MO, Kchouk ME, El Omri A, Han J, et al. (2009) Effect of Tunisian Capparis spinosa L. extract on melanogenesis in B16 murine melanoma cells. J Nat Med 63(4): 468–472.
- Sultan AÖ, Çelik TA (2009) Genotoxic and Antimutagenic effects of Capparis spinosa L. on the Allium cepa L. Root tip meristem cells. Caryologia 62(2): 114-123.
- Trombetta D, Occhiuto F, Perri D, Puglia C, Santagati NA, et al. (2005) Antiallergic and antihistaminic effect of two extracts of Capparis spinosa L. flowering buds. Phytother Res 19(1): 29-33.
- Ali-Shtayeh MS, Abu Ghdeib SI (1999) Antifungal activity of plant extracts against dermatophytes. Mycoses 42(11-12): 665–672.
- Masadeh MM, Alkofahi AS, Alzoubi KH, Tumah HN, Bani-Hani K (2014) Anti-Helicobactor pylori activity of some Jordanian medicinal plants. Pharm Biol 52(5): 566-569.
- Boga C, Forlani L, Calienni R, Hindley T, Hochkoeppler A, et al. (2011) On the antibacterial activity of roots of Capparis spinosa L. Nat Prod Res 25(4): 417-421.
- Gadgoli C, Mishra SH (1999) Antihepatotoxic activity of p-methoxy benzoic acid from Capparis spinosa. J Ethnopharmacol 66(2): 187–192.
- Yang T, Liu YQ, Wang CH, Wang ZT (2008) Advances on investigation of chemical constituents, pharmacological activities and clinical applications of Capparis spinosa. Zhongguo Zhong Yao Za Zhi 33(21): 2453-2458.
- Tlili N, Nasri N, Saadaoui E, Khaldi A, Triki S (2009) Carotenoid and tocopherol composition of leaves, buds, and flowers of Capparis spinosa grown wild in Tunisia. J Agric Food Chem 57(12): 5381-5385.
- Yang T, Wang C, Liu H, Chou G, Cheng X, et al. (2010) A new antioxidant compound from Capparis spinosa. Pharm Biol 48(5): 589–594.
- Bonina F, Puglia C, Ventura D, Aquino R, Tortora S, et al. (2002) In vitro antioxidant and in vivo photoprotective effects of a lyophilized extract of Capparis spinosa L buds. J Cosmet Sci 53(6): 321-335.
- Tlili N, Elfalleh W, Saadaoui E, Khaldi A, Triki S, et al. (2011) Phenolic compounds, tocopherols, carotenoids and Vitamin c of commercial caper. J Food Bio Chem 35: 472–483.
- Bianco G, Lelario F, Battista FG, Bufo SA, Cataldi TR (2012) Identification of glucosinolates in capers by LC-ESI-hybrid linear ion trap with Fourier transform ion cyclotron resonance mass spectrometry (LC-ESI-LTQ-FTICR MS) and infrared multiphoton dissociation. J Mass Spectrom 47(9): 1160-1169.
- Wang H, Wang H, Shi S, Duan J, Wang S (2012) Structural characterization of a homogalacturonan from Capparis spinosa L. fruits and anti-complement activity of its sulfated derivative. Glycoconj J 29(5-6): 379-387.
- Duman E, Özcan MM (2014) Physicochemical properties of caper species seed oils collected from two different harvest years. J Sci Food Agric 1-7.
- Lam SK, Ng TB (2009) A protein with antiproliferative, antifungal and HIV-1 reverse transcriptase inhibitory activities from caper (Capparis spinosa) seeds. Phytomedicine 16(5): 444–450.
- Kala CP (2005) Ethnomedicinal botany of the Apatani in the Eastern Himalayan region of India. J Ethnobiol Ethnomed 1: 11.
- Al-Said MS, Abdelsattar EA, Khalifa SL, EL-Feraly FS (1988) Isolation and identification of an anti- inflammatory principle from Capparis spinosa. Pharmazie 43(9): 640-641.
- Alcántara M, Morales M, Carnés J (2013) Food allergy to caper (Capparis spinosa). J Investig Allergol Clin Immunol 23(1): 67-69.
- Caruso M, Frasca G, Di Giuseppe PL, Pennisi A, Tringali G, et al. (2008) Effects of a new nutraceutical ingredient on allergen-induced sulphidoleukotrienes production and CD63 expression in allergic subjects. Int Immunopharmacol 8(13-14): 1781-1786.
- Eddouks M, Lemhadri A, Michel JB (2005) Hypolipidemic activity of aqueous extract of Capparis spinosa L. in normal and diabetic rats. J Ethnopharmacol 98(3): 345-350.
- Eddouks M, Ouahidi ML, Lemhadri A, Burcelin R (2009) Pharmacological study of Capparis spinosa and Chamaemelum nobile in HFD mice. Advances in Phytotherapy Research, Research Signpost. 151-159.
- Mahasneh AM, Abbas JA, Al-Oqalah A (1996) Antimicrobial activity of extracts of herbal plants used in the traditional medicine of Bahrain. Phytotherapy Research 10: 251-253.
- Mahasneh AM (2002) Screening of some indigenous Qatari medicinal plants for antimicrobial activity. Phytotherapy Research 16: 751–775.
- Arena A, Pavone B, Tomaino A, Bonina FP, Saija A, et al. (2008) Antiviral and immunomodulatory effect of a lyophilized extract of Capparis spinosa L. buds. Phytotherapy Research 22: 313–317.
- Daoudi A, Aarab L, Abdel-Sattar E (2013) Screening of immunomodulatory activity of total and protein extracts of some Moroccan medicinal plants. Toxicol Ind Health 29: 245-53.
- Al-Soqeer A (2011) Antioxidant activity and biological evaluation of hot-water extract of Artemisia monosperma and Capparis spinosa against lead contamination. Research Journal of Botany 6: 11-20.
- Germano MP, Pasquale RD, D’angelo V, Catania S, Silvari V, et al. (2002) Evaluation of extracts and isolated fraction from Capparis spinosa L. buds as an antioxidant source. J Agric Food Chem 50(5): 1168-1171.
- Abdel-Sattar V, Maes L, Salama MM (2010) In Vitro activities of plant extracts from Saudi Arabia against Malaria, Leishmaniasis, Sleeping Sickness and Chagas Disease. Phytotherapy Research 24: 1322–1328.
- Eddouks M, Moufid A, Michel JB, Zeggwagh NA (2009) Cardiovascular effect of Capparis spinosa in anesthetized normal rats. Advances in Phytotherapy Research, Research Signpost. 175-185.
- Shad AA, Ahmad S, Ullah R, AbdEl-Salam NM, Fouad H, et al. (2014) Phytochemical and biological activities of four wild medicinal plants. Scientific World Journal 1-7.
- Khanfar MA, Sabri S, Zarga MH, Zeller KP (2003) The chemical constituents of Capparis spinosa of Jordanian origin. Nat Prod Res 17(1): 9-14.
- Sharaf M, El-Ansari MA, Saleh NA (2000) Quercetin triglycoside from Capparis spinosa. Fitoterapia 71(1): 46-49.
- Pu Fu X, Aisa HA, Abdurahim M, Yili A, Aripova SF, et al. (2008) New spermidine alkaloids from Capparis spinosa roots. Phytochemistry Letters 1: 59–62.
- Ramezani Z, Aghel N, Keyghobadi H (2008) Rutin from different parts of Capparis spinosa growing wild in Khuzestan/Iran. Pak J Biol Sci 11: 768-772.
- Matthäus B, Ozcan M (2005) Glucosinolates and fatty acid, sterol, and tocopherol composition of seed oils from Capparis spinosa Var. spinosa and Capparis ovata Desf. Var. canescens (Coss.) Heywood. J Agric Food Chem 53(18): 7136-7141.
- Siracusa L, Kulisic-Bilusic T, Politeo O, Krause I, Dejanovic B, et al. (2011) Phenolic composition and antioxidant activity of aqueous infusions from Capparis spinosa L. and Crithmum maritimum L. before and after submission to a two-step in vitro digestion model. J Agric Food Chem 59: 12453-12459.