Predictors and Outcomes of No-reflow Phenomenon Following Primary Percutaneous Intervention for ST Elevation Myocardial Infarction
Rozza AA1*, Elzohery YZ2, Fahmy AM3, Abo Ahmed MA4, Elgizy ME5
1 Faculty of Medicine, The Department of Cardiology, Al Azhar, University, Cairo, Egypt.
2 National Heart Institute, Cairo, Egypt.
*Corresponding Author
Ahmed A. Rozza, MD,
Faculty of Medicine, The Department of Cardiology,
Al Azhar, University, Cairo, Egypt.
Tel: 201000495009
E-mail: ahmedelagiz@gmail.com
Received: August 21, 2017; Accepted: September 25, 2017; Published: September 28, 2017
Citation: Rozza AA, Elzohery YZ, Fahmy AM, Abo Ahmed MA, Elgizy ME. Predictors and Outcomes of No-reflow Phenomenon Following Primary Percutaneous Intervention for
ST Elevation Myocardial Infarction. Int J Cardiol Res. 4(2), 82-90. doi: http://dx.doi.org/10.19070/2470-4563-1700014
Copyright: Rozza AA© 2017. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: The term no-reflow was first used by Majno and colleagues in the setting of cerebral ischemia in 1967. This phenomenon was initially described by Krug et al. during induced myocardial infarction in the canine model in 1966 and again by Kloneret al.in1974 in which it occurred for 90 min after temporary epicardial coronary artery occlusion. Myocardial tracers, such as carbon black or thioflavin S were injected to document uniform flow distribution across the myocardial tissue after 40 min of occlusion. After 90 min, persistent subendocardial perfusion defects were seen with no-reflow.
Objectives: Detection, prevention, and treatment of no-refloware likely to have an important impact on the outcome of PPCI. Here we propose possible personalized forms of prevention and treatment, on the basis of the notion that no-reflow is a dynamic process characterized by multiple pathogenetic components.
Methods: We analyzed a total of 120 consecutive patients with STEMI and treated with PPCI between1st of November 2016 to July 2017 at our institution. The patients are divided into two groups according to the final TIMI flow after the primary PCI, and the incidence of in hospital MACE as follows: Group A: had a normal flow after the PPCI and did not have in hospital MACE; Group B:had either no reflow after the PPCI or experienced in hospital MACE.
Results: The incidence of no reflow was 13.2%, and in hospital MACE was 5%, with cardiac death as the predominant form of in hospital MACE. The group with no reflow or in hospital MACE showed significantly older age, longer door to balloon time, higher levels of admission RPG, N/L ratio, and MPV. Compared to the literature, Egyptian patients had more diabetes mellitus, more dyslipidaemia, longer door to balloon time.
Conclusions: Older patient age, longer door to balloon time, admission hyperglycemia, higher admission N/L ratio, MPV, longer reperfusion time, elevated level of high sensitive CRP on admission, and markedly elevated levels of CKMB, large thrombus burden LTB, are useful predictive factors for the occurrence of no reflow post PPCI, and/or in hospital MACE.
2.Abbreviations
3.Introduction
4.1 Statistical Analysis
5.Results
5.1 The Baseline Clinical Characteristics
6. Discussion
6.1 Baseline Clinical Characteristics
6.2 Admission characteristics
6.3 Timing Variables
6.4 Laboratory Results
6.5 Admission Hyperglycemia
6.6 Neutrophils/Lymphocytes Ratio (N/L)
6.7 Mean Platelet Volume (MPV)
6.8 Angiographic Findings and Procedural Aspects
6.9 Procedural Outcome
6.10 In-Hospital Course
6.11 Study Limitations
7. Conclusions
8. Perspectives
9.References
KeyWords
Acute Myocardial Infarction; No-Reflow Phenomenon; Percutaneous Coronary Intervention.
Abbreviations
ACC/AHA: American College of Cardiology/American Heart Association; ACS: Acute Coronary Syndrome; AMI: Acute Myocardial Infarction; BMS: Bare metal Stents; CK: Creatine Kinasel; CK-MB: Creatine Kinase M Band; CrCl: Creatinine Clearance; CRP: C-reactive Protein; DES: Drug Eluting Stents; DM: Diabetes Mellitus; ECG: Electrocardiogram; ESC: European Society of Cardiology; HCT: Haematocrit; Hgb: Haemoglobin; HR: Heart Rate; HTN: Hypertension; IDDM: Insulin-Dependent Diabetes Mellitus; IHD: Ischemic Heart Disease; IRA: Infarct-Related Artery; LAD: Left Anterior Descending Artery; LCX: Left Circumflex Artery; LDLc: Low Density Lipoprotein cholesterol; LMC: Left Main Coronary Artery; MACE: Major Adverse Cardiac Events; MI: Myocardial Infarction; MPV: Mean platelet volume; MVD: Multivessel Disease; NIDDM: Non- Insulin-Dependent Diabetes Mellitus; N/L Ratio: Neutrophils/Lymphocytes Ratio; NSTEMI: Non St elevation Myocardial Infarction; OM: Obtuse Marginal Artery; PAD: Peripheral Arterial Disease; PCI: Percutaneous Coronary Intervention; PDA: Posterior descending artery; POBA: Plain Old Balloon Angioplasty; PPCI: Primary percutaneous coronary intervention; RCA: Right Coronary Artery; RPG: Random plasma Glucose; ST: The Segment From S wave end To onset of T Wave In ECG; STEMI : ST Segment Elevation Myocardial Infarction; TIMI : Thrombolysis In Myocardial Infarction.
Introduction
Primary percutaneous coronary intervention (PCI) is the reperfusion strategy of choice in restoring blood flow to the occluded coronary artery in patients with ST elevation myocardial infarction (STEMI) [2]. Impaired coronary flow (Thrombolysis in Myocardial infarction grade < 3) despite restoration of epicardial coronary artery patency in the absence of any spasm or dissection is known as no-reflow [3].
It is thought to be caused by a combination of ischemic endothelial injury that obstructs the capillary lumen, neutrophil accumulation, reactive oxygen species and distal embolization of atherothrombotic debris. No-reflow occurs in 11-41% of STEMI patients treated by primary PCI and is associated with poor left ventricular function, adverse clinical events and death [4].
A number of clinical, serologic and angiographic parameters have been shown to be associated with no-reflow. The results of clinical trials testing a number of treatment strategies for no-reflow have been conflicting and there is no definitive treatment of no-reflow once it has occurred [5, 6].
In the absence of an effective treatment strategy, it is crucial to prevent no-reflow by knowing the predictors or risk factors of no-reflow. Previous studies have identified various predictors of no-reflow, which are different between studies, likely due to the differences in the populations being studied [7].
Methods
This study is a prospective study, comprised 120 patients with STEMI presenting to Al-Azhar Main University Hospital and National heart institute (NHI) from 1st of November 2016 to July 2017.
Patients with STEMI eligible for PPCI according to European Society of Cardiology (ESC) guidelines were included [2]. while the following were excluded: (patients performed percutaneous coronary interventions for stable angina pectoris, unstable angina or non-ST elevation myocardial infarction (NSTEMI), Patients with malignancies, coagulation disorders, Advanced liver or renal disease, Patients with valvular, congenital heart diseases and those with cardiomyopathies).
Every patient's record included: Informed consent taken from patients. In case of incompetent patients, the informed consent will be taken from the guardians. Thorough history taking with special emphasis on: Risk factors (Age, gender, diabetes, hypertension, smoking, dyslipidemia, family history). History of acute coronary syndromes (ACS) and revascularization. Door to balloon time (in hours). Presence of pre-infarction angina (defined as presence anginal pains within the past 48 hrs. preceding the incidence of STEMI. Complete clinical examination, with demonstration of admission blood pressure, pulse, and killip class. Laboratory investigation (on admission): Complete blood count (CBC) (including mean platelet volume (MPV) and neutrophils/ lymphocytes (N/L) ratio). Random plasma glucose (RPG) level (in mg/dl). Standard 12 lead electrocardiogram (ECG). Conventional coronary angiography indicating initial TIMI flow in the IRA. The patients were studied according to the presence of various clinical and laboratory variables (age, gender, absence of pre-infarction angina, door to balloon time, location of the infarction, admission plasma glucose level and CBC including N/L ratio and MPV, and initial TIMI flow in the IRA), the final TIMI flow after the PPCI, and the incidence of in hospital MACE.
Data were fed to the computer and analyzed using IBM SPSS software package version 20.0. Qualitative data were described using number and percent. Quantitative data were described using Range (minimum and maximum), mean, standard deviation and median. Comparison between different groups regarding categorical variables was tested using Chi-square test. When more than 20% of the cells have expected count less than 5, correction for chi-square was conducted using Fisher’s Exact test or Monte Carlo correction. The distributions of quantitative variables were tested for normality using Kolmogorov-Smirnov test, Shapiro- Wilk test and D'Agstino test, also Histogram and QQ plot were used for vision test. If it reveals normal data distribution, parametric tests were applied. If the data were abnormally distributed, non-parametric tests were used. For normally distributed data, comparison between two independent populations were done using independent t-test, while abnormally distributed data was assessed using Mann Whitney test. Significance test results are quoted as two-tailed probabilities. Significance of the obtained results was judged at the 5% level.
Results
The study was a two-center, prospective, observational study consisted of 120 consecutive patients admitted at Al-Azhar main university hospital and the national heart institute (NHI) for primary PCI from the 1st of November 2016 to 31st of July 2017. The patients are divided into two groups according to the final TIMI flow after the primary PCI, and the incidence of in hospital MACE as follows:
Group A: had a normal flow after the PPCI and did not have in hospital MACE.
Group B: had either no reflow after the PPCI or experienced in hospital MACE.
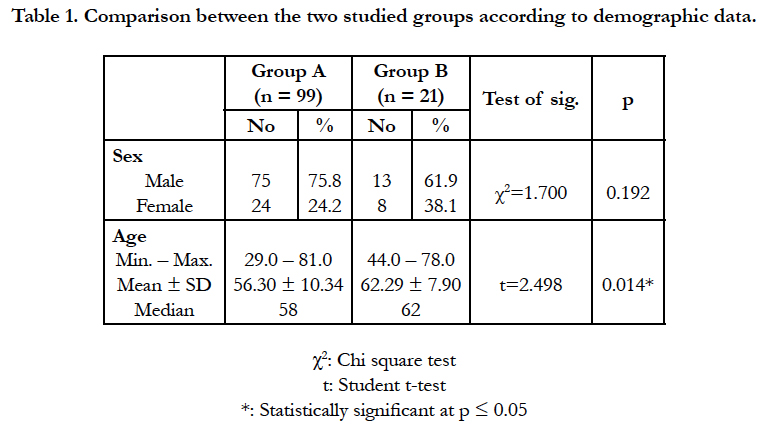
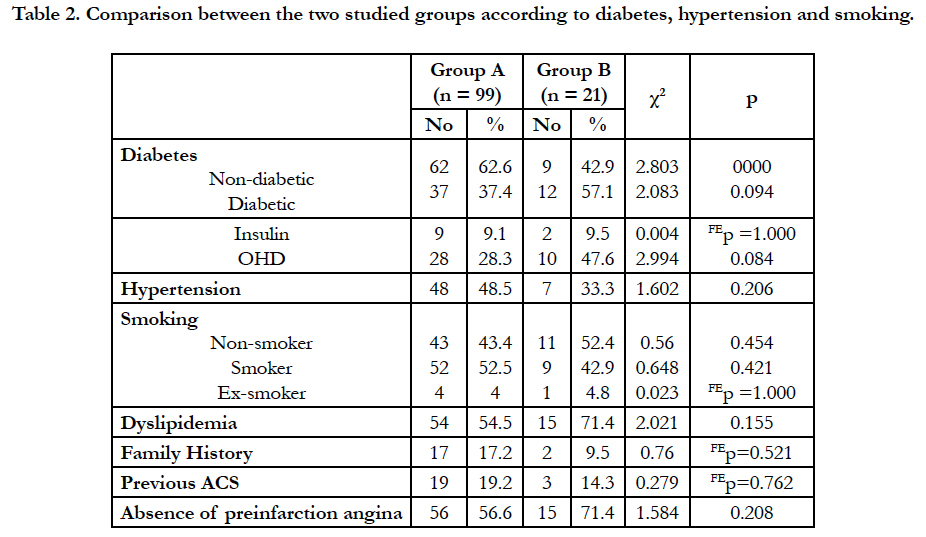
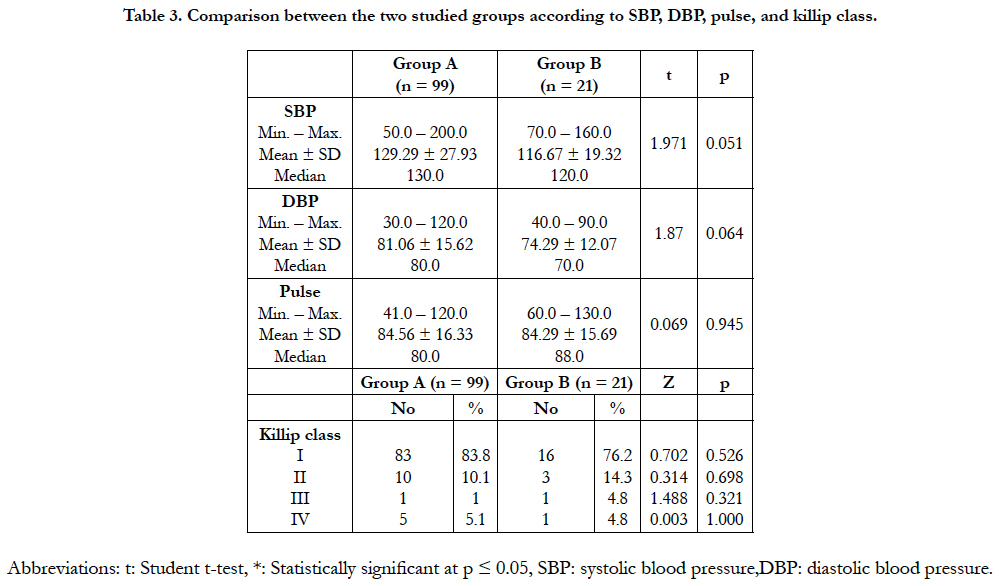
The demographic criteria as demonstrated in (Table1). The patients in the group B were significantly older, there is no significant difference between the two groups according to gender according to the presence of risk factors as demonstrated in (Table 2), patients in group B had more prevalence of NIDDM and dyslipidemia more than group A, according to Admission characteristics as shown in (Table 3) with demonstration of admission blood pressure, pulse, and killip class there is no significant difference between the two groups.
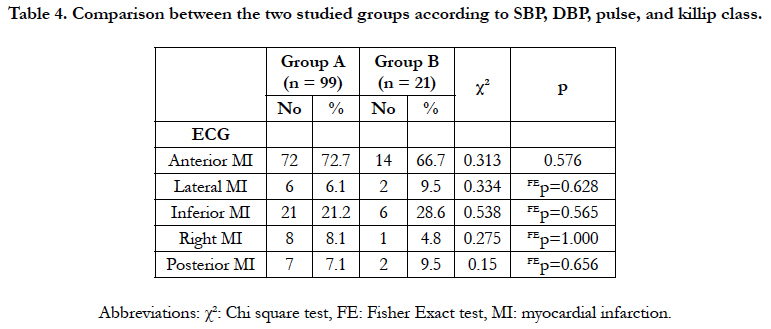
As regard ECG diagnosis; (Table 4) 72 (72.7%) patients presented with anterior STEMI in group A, and 14 (66.7%) patients in group B. 21 (21.2%) patients presented with inferior STEMI in group A, and 6 (28.6%) patients in group B. 6 (6.1%) patients presented with lateral STEMI in group A, and 2 (9.5%) patients in group B. 8 (8.1%) patients presented with right STEMI in group A, and 1 (4.8%) patient in group B. 7 (7.1%) patients presented with posterior STEMI in group A, and 2 (9.5%) patients in group B.
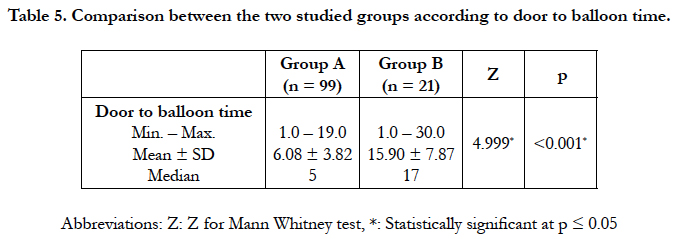
Timing variables: as shown in (Table 5)
The longer mean time from onset of symptoms to balloon inflation in 1ry PCI was more in group B 15.9 ± 7.8 hours, versus 6 ± 3.8 hours in group A. with statistically significant p value <0.001.
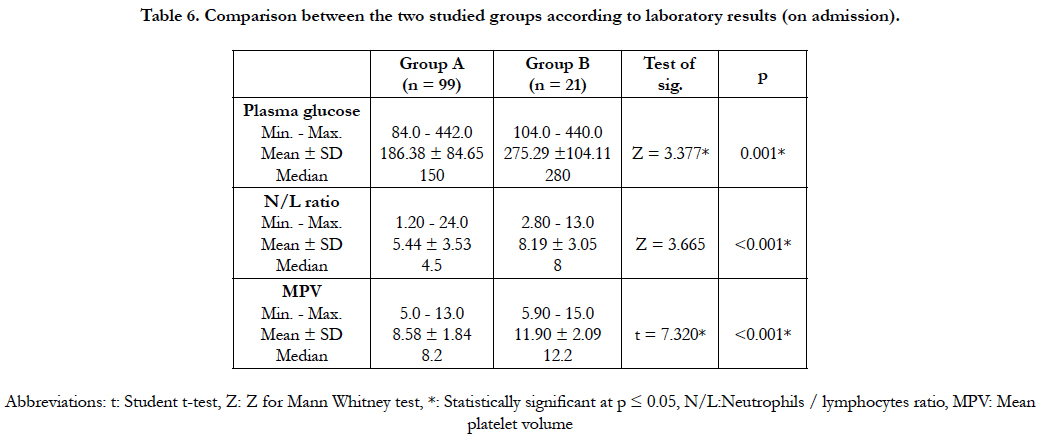
Laboratory results (Tables 6) shows that: The median of admission random plasma glucose was higher in group B the median was 280 mg/dl (range=336 mg/dl), while in group A was 150 mg/dl (range=358 mg/dl). P value was statistically significant <0.001.
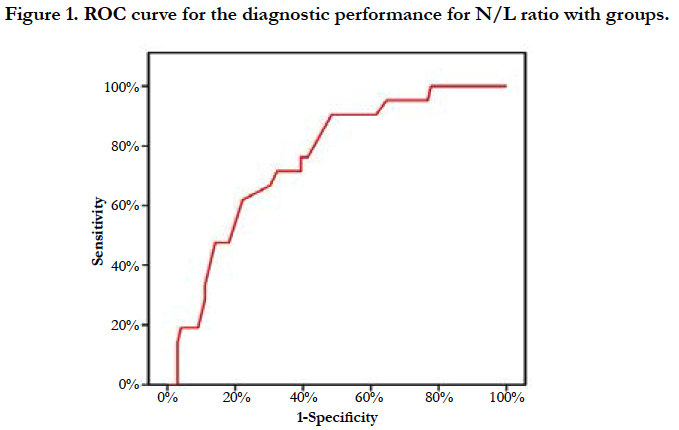
The Mean neutrophils/lymphocytes ratio in higher in group B8.19 ± 3.05, compared to group A which was 5.44 ± 3.53 with significant p value <0.001. ROC curve analysis (Figure 1) of results revealed that N/L ratio > 4.6 predicts no reflow or in hospital MACE with sensitivity 90.4%, and specificity 51.5%.
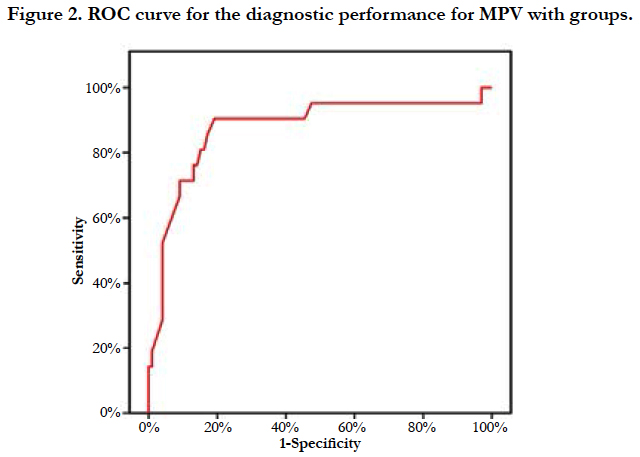
The mean of mean platelet volume (MPV) in group B was higher 11.9 ± 2.09 FL, compared to group A it was 8.58 ± 1.84 FL, WITH statistically significant P value < 0.001. ROC curve analysis of results (Figure 2) revealed that MPV > 9.9 FL predicts no reflow or in hospital MACE with sensitivity 90.4%, and specificity 80.8%.
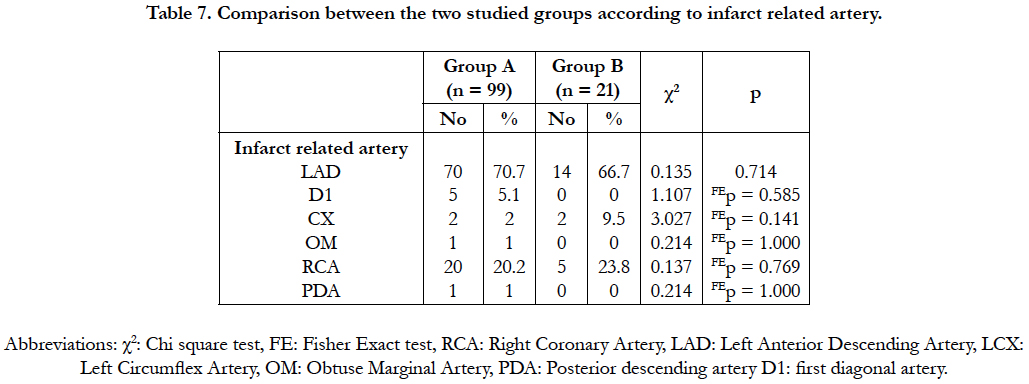
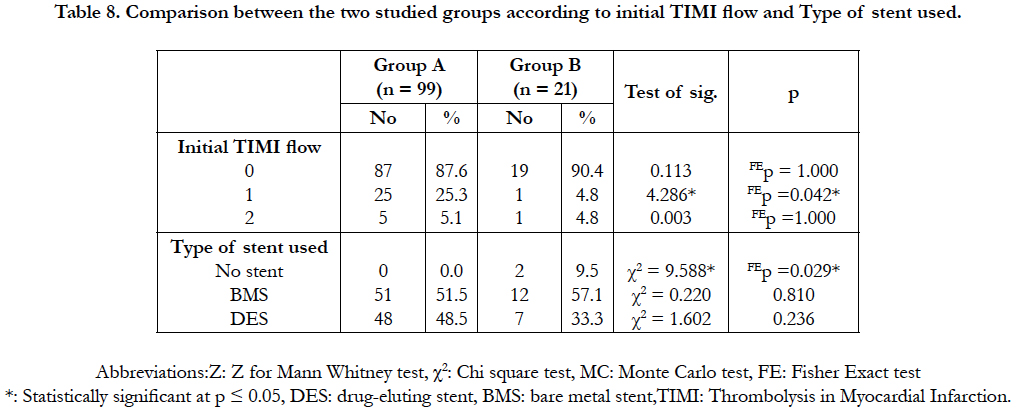
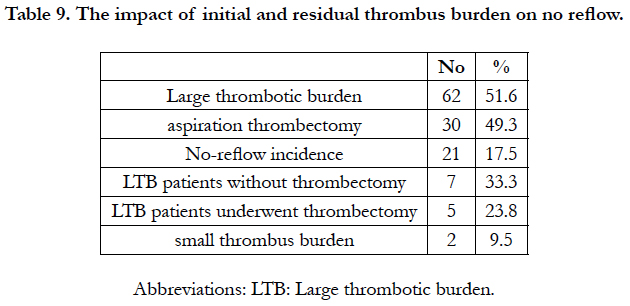
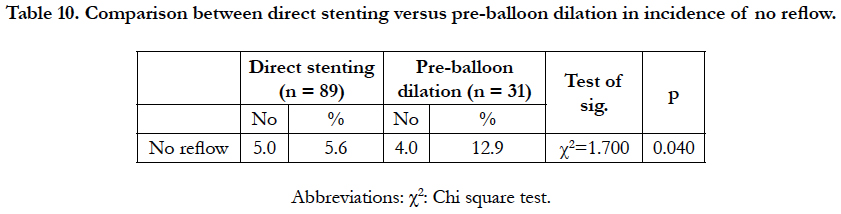
Angiographic findings and procedural aspects: As regarding (Infarct related artery (IRA): (Table 7), Initial TIMI flow (before 1ry PCI): (Table 8), Type of stent used: (Table 8)), there is no significant difference between the two groups. The impact of initial and residual thrombus burden on no reflow as shown in (Table 9) Large thrombotic burden (LTB) was observed in 62 patients among them 30 patients underwent aspiration thrombectomy, the no-reflow phenomenon occurred most frequently in LTB patients without thrombectomy, followed by those who underwent thrombectomy and the small thrombus burden group (33.8 vs. 23.8 vs. 9.5%, respectively. In comparison between direct stenting versus pre-balloon dilation in incidence of no reflow (Table 10) Patients who underwent direct stenting (n = 89) had a better risk profile compared with the use of pre-balloon dilation (n=31). The incidence of angiographic no-reflow was 12.9% in the preballoon dilation group and 5.6% in the direct stenting group (P = .040).
Table 8. Comparison between the two studied groups according to initial TIMI flow and Type of stent used.
We found in our study that the mean age was close to the mean age in other studies, with significantly higher in no reflow group than in reflow group (62.29 ± 7.9 years vs 56.3 ± 10.3 years respectively, p=0.014), with prevalence of DM and dyslipidemia higher than that published in the literature, because of the pandemic of DM in our country which may in part be associated with the metabolic syndrome and stressing the urgent need for a national policy for primary and secondary prevention of diabetes and dyslipidemia. The prevalence of smoking in our country is still high inspite of aggressive public health efforts to limit tobacco use. Control of hypertension is of utmost importance, as this is one of the major risk factors, with comparison with above mentioned studies, the ratio of hypertensive patients was not largely different to our study.
Ndrepepa G et al., [9] studied the clinical factors related to the development of no-reflow phenomenon after successful coronary reperfusion in patients with AMI. Between January 1998 and December 2007, 1518 patients with STEMI presenting within 24 hours from the symptom onset were treated with PPCI in the Deutsches Herzzentrum Munich. Mean age of the no reflow group patients was significantly higher than the reflow group (65.8 vs 61.4 years, p=0.001), and history of previous MI was significantly higher in no reflow group than reflow group (18.5% vs 11.7% respectively, p=0.041), with non-significant difference in sex (71.3% vs 75% respectively), presence of DM (14.8% vs 20.3%, respectively), hypertension (66.7% vs 67.3% respectively), current smoking (30.6% vs 40.5% respectively) & dyslipidemia (57.4% vs 58.1% respectively).
In our study, we did not find the significant difference between group A and B (the normal flow and no reflow groups respectively) regarding killip class (more patients with killip class ≥ II were found in the no reflow group in some studies in the literature), pulse rate (pulse rate was significantly higher in no reflow group in some studies in the literature), location of MI (anterior MI was significantly higher in the no reflow group in some studies in the literature).
Ndrepepa G et al., [9] reported that there was significant difference between the no reflow and reflow groups as regards killip class (class I 63% vs 70.9%, class ≥ II 34% vs 29.1%, p=0.019), with no significant difference between the study groups with respect to median SBP (125 vs 130 mmHg), median DBP (70 mmHg in both groups), median of pulse rate (78 bpm in both groups), and location of MI (anterior 41.7% vs 43%, inferior 41.7% vs 38.1%, lateral 16.6% vs 18.9%).
Ito M., et al., [10] there was significant difference between the no reflow and reflow groups as regards killip class (class I % 83.3% vs 72.1%, class ≥ II 16.7% vs 27.9%, p=0.03).
In our study, the door to balloon time in the normal flow group was near to that published in the literature, but the door to balloon time in the no reflow group was much longer than that published in the literature. Longer door to balloon time is associated with more ischemic injury to tissues, hence the occurrence of no reflow and in hospital MACE.
Ndrepepa G., et al., [9] reported that door to balloon time was significantly longer in the no reflow group than reflow group (the median was 10.7 vs 6.5 hours, p=0.001).
Akpek M., et al., [8] reported that door to balloon time was significantly longer in the no reflow group than reflow group (the mean was 4.8 ±1.3 hours’ vs 4.2 ± 1.4 hours, p<0.001).
In our study, the admission RPG was significantly higher in group B than in group A (the mean was 275.3 ± 104.1 mg/dl vs 186.4 ± 84.7 mg/dl, p=0.001). The N/L ratio was significantly higher in group B than in group A (the mean was 8.19 ± 3.05 vs 5.44 ± 3.53, p<0.001). ROC curve analysis of results revealed that N/L ratio >4.6 predicts no reflow or in hospital MACE with sensitivity 90.4%, and specificity 51.5%. The MPV was significantly higher in group B than in group A (the mean value was 11.9 ± 2.09 fl vs 8.58 ± 1.84 fl, p<0.001). ROC curve analysis of results revealed that MPV > 9.9 fl predicts no reflow or in hospital MACE with sensitivity 90.4%, and specificity 80.8%. In this study, the admission RPG, MPV, and N/L ratio were of near values to that mentioned in literature.
This study. Moreover, patients with hyperglycemia had a lower contrast enhancement score and lowerΔWMS than did those without it, even after adjusting for differences in the peak CK value. These results indicate that the effects of hyperglycemia on microvascular integrity and WMS could be independent from the infarct size [12]. Still, we could not definitely determine whether hyperglycemia was a cause or consequence of a large infarct size that could be related to the no-reflow phenomenon. Further prospective studies in which the blood glucose level was controlled before coronary reperfusion would be required to clarify these associations.
N/L ratio were found to be significantly higher in patients with thrombus formation than in patients without thrombus formation. LI-Dongbao et al., found that N/L ratio was independently predictive of thrombus formation in the IRA, and thrombus formation in the IRA was the only predictor of no-reflow/slow flow during PCI [13].
Recent studies with animal models have shown direct visualization of neutrophilic invasion of atherosclerotic plaque. Neutrophils may make plaques rupture more easily through the release of proteolytic enzymes, arachidonic acid derivatives, and superoxide radicals [14]. Therefore, the higher neutrophil count may not only mirror the exacerbated inflammatory condition found in patients with atherosclerotic disease, but also may be associated with the role of those cells in the instability of atherosclerotic plaque.
In this study we assume that the presence of larger, more reactive platelets or platelet aggregates may be associated with intravascular plugging on both epicardial and tissue level of the IRA, thus resulting in no-reflow and after PPCI. Higher MPV may correspond with the increased number of both plateletleukocyte and platelet-platelet aggregates [15].
Huczek Z., et al., [16] found that Administration of abciximab during PPCI resulted in significant reduction of six-month mortality in patients with high MPV values. The results of their study suggest that patients with high MPV on admission represent the group with higher risk for thrombosis [16].
Akpek M., et al., [8] found that admission RPG was significantly higher in the no reflow group than in reflow group (the mean was 196.6 ± 89.6 mg/dl vs 152.7 ± 62 mg/dl, p < 0.001). They also reported that the N/L ratio was significantly higher in no reflow group compared to that of normal flow group (4.6 ± 1.7 vs. 3.1 ± 1.9, p<0.001), and that N/L ratio >3.3 predicted no reflow with 74% sensitivity and 83% specificity.
Huczek Z.,et al., [16] reported that the mean admission MPV was significantly higher in the no reflow patients compared with those with reflow post PPCI (10.8 ± 0.95 FL vs 9.9 ± 0.85 FL, p<0.0001), and that a value of MPV ≥ 10.3 FL predicted no reflow with a sensitivity 61.9%, and specificity 74.3%.
In this study, there was no significant difference between groups A and B regarding IRA (LAD 70.7% vs 66.7%, LCX 2% vs 9.5%, RCA 20.2% vs 23.8%, D1 5%vs 0%, OM 1% vs 0%, PDA 1% vs 0%, respectively). No significant difference was present between the two groups regarding the initial (pre-intervention) TIMI flow grade (TIMI 0 in 87.6% vs 90.4%, TIMI 1 in 25.3% vs 4.8%, TIMI 2 in 5.1% vs 4.8%, respectively). Two patients (9.5%) in group B had POBA, while none in group A had POBA. No significant difference between the two groups regarding the type of stent used (BMS in 51.5% vs 57.1%, and DES in 48.5% vs 33.3%, respectively). In our study, more stent implantation, and more patients with initial TIMI 0 flow were found in the two studied groups than in the comparing studies, with the distribution of IRA near to that of other studies.
Ndrepepa G et al., [9] found no significant difference between the reflow and no reflow groups regarding IRA (LAD 43.3% vs 42.6%, LCX 20.7% vs 15.7%, RCA 33.6% vs 37.1%), and the type of intervention (POBA in 18.3% vs 18.5%, stenting in 81.7% vs 81.5%). They found significantly more patients with preintervention TIMI flow grade 0 in the no reflow group (54.6% vs 83.3%, p<0.001).
In our study, 13% of patients had no reflow (i.e.; TIMI flow <3, and/or MBG<2) after PPCI, and 87% of patients had reflow. There is a great variety in the literature regarding the incidence of no reflow after PPCI due to difference in the methods of diagnosis of no reflow, such as: post PPCI TIMI flow grade and MBG, radionuclide scintigraphy, and MCE, with difference in sensitivity between different methods.
Ndrepepa G et al., [9] reported that TIMI 3 flow post PCI was achieved in 89% of patients and 9% suffered no reflow detected using radionuclide scintigraphy examination. They found that previous MI (18.5% in no reflow group vs 11.7% in reflow group, p=0.041), baseline TIMI flow grade (TIMI 0-1 in 88% of no reflow group vs 64% of reflow group, p<0.001) were significant independent predictors of no reflow after PPCI.
In this study in-hospital MACE; cardiac death occurred in 5% of patients (6 cases), and they were cases of three vessel disease. Five of these cases were cases of incessant ventricular fibrillation that not responded to cardiopulmonary resuscitation according to European council of resuscitation guidelines, with inability to know whether they developed re-infarction or stent thrombosis. One case was presented with long standing cardiogenic shock, and died of persistent cardiogenic shock, that not responded to complete revascularization of all affected coronaries. No welldefined cases of reinfarction, stent thrombosis, and target vessel revascularization (TVR).
Akpek M., et al., [8] reported that in-hospital MACE; cardiac death 7%, reinfarction 5%, and in stent thrombosis 5%, with in hospital MACE significantly higher in the no reflow group (8.6% vs. 4.3%, P < 0.001), and N/L ratio was found to be independent predictor of in hospital MACE.
• The sample size is relatively small compared to large studies published in the literature, and larger studies are needed to validate these results.
• They do not represent all-comers who presented with acute STEMI because there are still many patients in our country treated with fibrinolysis only without further PCI because of
financial aspect. That is to say, the presumed lower mortality rate of affluent patients and the higher mortality rate of the sicker patients may balance each other out.
• There is a proportion of the delay to PCI comprises the time taken by patients to decide whether they can proceed with the procedure, based on financial constraints.
• Our data represent a two-centre experience where the operators are experienced and the hospital has good medical and paramedical team and good ambulance system. Whether these results can be generalized to other hospitals in our country is unclear.
• Our study was not designed to evaluate safety and efficacy of DES compared to BMS in patients with STEMI and thrombus aspiration compared to direct PCI without thrombus aspiration.
• Cases of cardiac death were not thoroughly investigated, for example by autopsy, to define well and help to further prevent the causes of in hospital cardiac death post PPCI.
• We did not follow the no reflow patients after hospital discharge.
Conclusions
• Older patient age, longer pain to balloon time, admission hyperglycemia, higher admission N/L ratio, MPV, longer reperfusion time, elevated level of high sensitive CRP on
admission, and markedly elevated levels of CKMB, are useful predictive factors for the occurrence of no reflow post PPCI, and/or in hospital MACE.
• It was noted that no-reflow phenomenon was less in patients had past history of ischemia (pre-conditioning).
• Reversal of the no-reflow was achieved equally by the use of IC verapamil as SNP (81.81% vs. 88.88%).
• No-reflow patients showed higher post procedural myocardial ischemia, LV dysfunction and mortality.
• Manual thrombus aspiration reduced no-reflow incidence in STEMI patients with large thrombus burden (LTB). However,
even after thrombectomy, presenting residual thrombi and decreased coronary flow were related to a higher occurrence of the no-reflow phenomenon.
• Direct stenting in patients with AMI may reduce the incidence of angiographic no-reflow, thereby increasing ultimate effective myocardial reperfusion.
Perspectives
The use of admission random plasma glucose level, N/L ratio & MPV, to predict cases of STEMI liable for occurrence of No reflow, who may benefit more from thrombus aspiration, GpIIb/ IIIa inhibitors administration, and equipping the Cath lab with adenosine and verapamil to manage no reflow.
• Routine assessment of TIMI flow grade, MBG, and STR, obtained from the routine management of STEMI patients, as they are inexpensive tools that provide additional prognostic information in their management.
• Adequate knowledge of all the drugs that can be used safely for the reversal of the no-reflow phenomenon (including the indications, contraindications and side effects) must be known for all PCI operators.
• Direct stenting in patients with AMI may reduce the incidence of angiographic no-reflow, thereby increasing ultimate effective myocardial reperfusion.
• Adequate knowledge about pathophysiology of no-reflow must be kept in mind of all PCI operators as it is importantin the selection of the most appropriate therapeutic approach.
• The prevention of no-reflow phenomenon is the most important step by one of the following method:
A. Decrease of ischemia-related injury by reducing pain-onset-toballoon time (reperfusion time).
B. Decrease reperfusion-related injury by using glycoprotein IIb/IIIa antagonists.
C. Optimal and prompt treatment of hyperglycemiais likely to be an important target in the prevention of no-reflow.
References
- Morishima I, Sone T, Okumura K, Tsuboi H, Kondo J, Mukawa H, et al. Angiographic no-reflow phenomenon as a predictor of adverse long-term outcome in patients treated with percutaneous transluminal coronary angioplasty for first acute myocardial infarction. J Am Coll Cardiol. 2000 Oct;36(4):1202-9. PMID:11028471.
- Steg PG, James SK, Atar D, Badano LP, Blömstrom-Lundqvist C, Borger MA, et al. ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2012 Oct;33(20):2569-619. PMID:22922416.
- Harrison RW, Aggarwal A, Ou FS, Klein LW, Rumsfeld JS, Roe MT, et al. Incidence and outcomes of no-reflow phenomenon during percutaneous coronary intervention among patients with acute myocardial infarction. Am J Cardiol. 2013 Jan15;111(2):178-84. PMID:23111142.
- Schwartz BG, Kloner RA. Coronary no reflow, J Mol Cell Cardiol. 2012 April;52(4):873-882.
- Gick M, Jander N, Bestehorn HP, Kienzle RP, Ferenc M, Werner K, et al. Randomized evaluation of the effects of filter-based distal protection on myocardial perfusion and infarct size after primary percutaneous catheter intervention in myocardial infarction with and without ST-segment elevation. Circulation. 2005 Sep 6;112(10):1462-9. PMID:16129793.
- Kelbaek H, Terkelsen CJ, Helqvist S, Lassen JF, Clemmensen P, Kløvgaard L, et al. Randomized comparison of distal protection versus conventional treatment in primary percutaneous coronary intervention: the drug elution and distal protection in ST-elevation myocardial infarction (DEDICATION) trial. J Am Coll Cardiol. 2008 Mar 4;51(9):899-905. PMID:18308157.
- Choo EH, Kim PJ, Chang K, Ahn Y, Jeon DS, Lee JM, et al. The impact of no-reflow phenomena after primary percutaneous coronary intervention: a time-dependent analysis of mortality. Coron Artery Dis. 2014 Aug;25(5):392-8. PMID:24625688.
- Akpek M, Sahin O, Elick D, et al. association of neutrophils/lymphocytes ratio with coronary flow and in hospital MACE in patients with STEMI undergoing primary PCI. Eur Heart J 2013; 27:534–9.
- Ndrepepa G, Tiroch K, Keta D, Fusaro M, Seyfarth M, Pache J, et al. Predictive Factors and Impact of No Reflow After Primary Percutaneous Coronary Intervention in Patients with Acute Myocardial Infarction. Circ Cardiovasc Interv. 2010 Feb 1;3(1):27-33. PMID:20118156.
- Ito H, Iwakura K, Kawano S , Shintani Y, Yamamoto K, Kato A, et al. Predictive factors for development of the no-reflow phenomenon in patients with reperfused anterior wall acute myocardial infarction. J Am Coll Cardiol. 2001 Aug;38(2):472-7. PMID:11499740.
- Iwakura K, Ito H, Ikushima M, Kawano S, Okamura A, Asano K, et al. Association between hyperglycemia and the no-reflow phenomenon in patients with acute myocardial infarction. J Am Coll Cardiol. 2003 Jan 1;41(1):1-7. PMID:12570936.
- Capes SE, Hunt D, Malmberg K, Gerstein HC. Stress hyperglycemia and increased risk of death after myocardial infarction in patients with and without diabetes: a systematic overview. Lancet. 2000 Mar 4;355(9206):773-8. PMID:10711923.
- Zazula AD, Précoma-Neto D, Gomes AM, Kruklis H, Barbieri GF, Forte RY, et al. An assessment of neutrophils/ lymphocytes ratio in patients suspected of acute coronary syndrome. Arq Bras Cardiol. 2008 Jan;90(1):31-6. PMID:18317638.
- Naruko T, Ueda M, Haze K, van der Wal AC, van der Loos CM, Itoh A, et al. Neutrophil infiltration of culprit lesions in acute coronary syndromes. Circulation. 2002 Dec 3;106(23):2894-900. PMID:12460868.
- Campo G, Valgimigli M, Gemmati D, Percoco G, Tognazzo S, Cicchitelli G, et al. Value of platelet reactivity in predicting response to treatment and clinical outcome in patients undergoing primary coronary intervention: insights into the STRATEGY study. J Am Coll Cardiol. 2006 Dec 5;48(11):2178-85. PMID:17161242.
- Huczek Z, Kochman J, Filipiak KJ, Horszczaruk GJ, Grabowski M, Piatkowski R, et al. Mean platelet volume on admission predicts impaired reperfusion and long-term mortality in acute myocardial infarction treated with primary percutaneous coronary intervention. J Am Coll Cardiol. 2005 Jul 19;46(2):284-90. PMID:16022956.