Innovative Approaches to the Management of Acute Arterial Hypertension - Clevidipine Butyrate
Lakshmi N Kurnutala, M.D.1, Gurneet Sandhu, M.D.1, Rashmi Vandse, M.D.1, Suren Soghomonyan, M.D. Ph. D1*, Sergio D. Bergese, M.D.1,2
1* Department of Anesthesiology, Wexner Medical Center, Ohio State University, Columbus, OH.
2 Department of Neurological Surgery, Wexner Medical Center, Ohio State University, Columbus, OH.
*Corresponding Author
Suren Soghomonyan,
Department of Anesthesiology,
Wexner Medical Center,
Ohio State University, Columbus, OH.
Tel: (614)-293-0775;
Fax: (614)-366-1943
E-mail: suren.soghomonyan@osumc.edu
Article Type: Review Article
Received: November 18, 2013; Accepted: December 14, 2013; Published: December 16, 2013
Citation: Soghomonyan S et al., (2013) Innovative Approaches To The Management Of Acute Arterial Hypertension - Clevidipine Butyrate. Int J Anesth Res. 1(4), 18-24. doi: dx.doi.org/10.19070/2332-2780-130006
Copyright: Soghomonyan S© 2013. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited
Abstract
Acute arterial hypertension is one of the major concerns in many clinical settings including but not limited to operating room, intensive care and emergency care units. Perioperative hypertension is one of the major reasons for cancellation of elective surgeries, and also increases the perioperative morbidity. We would like to discuss pathophysiology, evaluation of the patients with acute hypertension, management of these patients and future considerations of the current intravenous antihypertensive medications.
2.Introduction
3.Pathophysiology of Acute Hypertension
4.Evaluation of Patients With AHE
5. Management of AHE
6. Clevidipine for The Management of Acute Hypertension
7. Disclosure
8. References
Keywords
Acute Hypertension; Intravenous; Anti-Hypertensive Medications; Calcium Channel Blockers; Dihydropyridine; Clevidipine.
Introduction
Acute arterial hypertension (AH) occurs in many clinical settings, some of which include the operating room, intensive care and emergency care units. The episodes of acute AH may occur in patients with pre-existing hypertension or as a de novo phenomena [1,2]. Early recognition and timely intervention in these patients are very important to prevent devastating complications.
In United States, the incidence of chronic AH among the adult population (>20 years age) reaches approximately 29% and the numbers are still growing. 72 million people in the country and 1 billion people worldwide are affected by chronic AH [3]. Among these 29% of people suffering from AH, only 58% undergo treatment, and target blood pressure (BP) values (< 140/90 mm Hg) are achieved in only 33% of Caucasians, 28% in African Americans, and 18% of Mexican Americans [4]. Up to 55,000 deaths each year are directly caused by AH, while it is considered a major contributing factor in at least another 300,000 deaths annually in United States [5].
High blood pressure affects most of the organ systems in human body and, constitutes the most important reason for office visits and prescription of medications. Chronically hypertensive patients are more prone to acute hypertensive episodes (AHE). The incidence of AHE (alternatively, named hypertensive crisis) in these patients approaches 1% [6,7]. Noteworthy that the AHE may occur in patients without any past history of AH. The Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC-7) classified blood pressure into four categories: normotensive, pre-hypertensive, and hypertensive stage I and stage II (Table 1). The hypertensive crisis is further divided into two types depending on the blood pressure and end organ damage – hypertensive emergency and hypertensive urgency. A hypertensive urgency is defined as an increase in systolic BP (SBP) above 180 mmHg or diastolic BP (DBP) above >110 mm Hg with no evidence of end organ damage to brain, heart, kidney, or eyes [3]. Hypertensive emergency is defined as an elevation of SBP >180 mmHg or DBP >120 mmHg associated with an end organ damage.
Hypertensive crises most frequently confronted by the emergency departments account for 27.5% of all nonsurgical emergencies and 3% of all emergency room visits. One of the most common reasons for postponing elective surgery is uncontrolled hypertension [8]. There are multiple anesthesia concerns related to hypertension in the perioperative period. Sympathetic overactivation occurring during anesthesia induction can increase the SBP in normotensive patients by up to 20-30mm Hg. In contrast, in hypertensive patients the SBP values could increase by up to 90 mmHg [9]. Because of anesthesia-induced sympathetic suppression and blunting of the baroreceptor reflex, these patients may experience episodes of sustained arterial hypotension following anesthesia induction and during anesthesia maintenance. In the postoperative period, the patients suffering from AH may present with rebound hypertension because of fading of the anesthetic drug effects, pain, excitement, hypercapnia and sympathetic activation triggered by hypothermia and shivering. The blood pressure fluctuations occur more frequently in patients with a history of preexisting AH, which makes them prone to postoperative complications [10,11].
Autoregulatory processes are abundant within many biological systems. They comprise inherent adaptive mechanisms that work to adjust that organ system response to stimuli to maintain the principal biological variables within the normal range. The efficacy of autoregulatory mechanisms depend on the extent of stimuli and varies among organ systems. The important organs responsible for the overall homeostasis, namely, the brain, heart and kidney require specific minimal amounts of perfusion and oxygen to maintain their optimal function. The cerebral autoregulation curve shifts to the right in patients with chronic hypertension [12,13]. Special care should be taken in selecting the optimal antihypertensive therapy for acute hypertensive episodes. Under normal conditions, the cerebral autoregulation is well maintained within the mean BP range of 60-160 mmHg. In cases of AHE, there is an acute failure of autoregulatory mechanisms which causes acute cerebral hyperemia, cerebral edema, blood-brain barrier dysfunction and irreversible damage of the nervous tissue [13,6].
Pathophysiology of Acute Hypertension
Even though AH is the most prevalent disease in the United States, the etiopathology of this disease is poorly understood till now and considered multifactorial [6,14,15,16]. AHE is precipitated by a sudden increase in systemic vascular resistance (SVR) resulting from mechanical stress and injury, endothelial damage, renin-angiotensin activation, oxidative stress and this causes failure in cerebral blood flow autoregulation [14,15]. Although, AHE is more characteristic for patients with a history of hypertension, it may still occur in normotensive patients. Vascular endothelial injury results from repeated episodes of acute hypertension which is associated with elevated SVR. As BP increases, vessel walls are subjected to increased stress, which leads to the release of vasoconstrictors resulting in further endothelial damage [6,14,17]. If not promptly treated, AHE activates the systemic hemocoagulation and other mechanisms including increased arteriolar permeability, upregulation of multiple inflammatory mechanisms, induction of oxidative stress, and overproduction of inflammatory cytokines [6]. Platelet aggregation and fibrinogen polymerization cause further vasoconstriction, and thrombosis aggravating vascular injury and decreasing blood flow to critical organs [6,14]. If this vicious cycle is not terminated, autoregulatory dysfunction becomes imminent [18].
Evaluation of Patients With AHE
Early detection and treatment of AHE is very important to prevent further damage to end organs. Proper history and physical examination gives very good idea about the current situation. A focused history should be obtained regarding AH, cardiovascular disorders, endocrine pathology (diabetes, adrenal tumors, pheochromocytoma), previous surgery, use of recreational drugs (cocaine, amphetamines, and phencyclidine), current medications (MAO inhibitors) and patient’s compliance to the medications, particularly antihypertensives.
In emergency situations, patients may present to the emergency department with nausea and vomiting, headache, chest pain, dyspnea, vertigo, and neurologic symptoms [19]. Hypertensive emergency cases are differentiated from the hypertensive urgency patients by thorough physical examination to identify the presence of end organ damage by referring cardiovascular, pulmonary, neurologic and fundoscopic examination [20]. The organs most susceptible to end-organ damage associated with hypertensive emergencies include the eye, kidney, heart, and brain [13,18]. Patients with a history of hypertension may tolerate SBP >200 mmHg or DBP of >150 mmHg without developing clinical signs and symptoms. There are no standard guidelines for laboratory investigations in these patients. Complete blood count, electrolytes, creatinine, and urine analysis along with EKG, and chest x-ray are used to evaluate the end organ damage. Further investigations can be ordered depending on the systems at risk during an AHE including echocardiogram, abdominal ultrasound, brain and thoraco-abdominal CT scan or MRI. Hypertensive emergencies associated with end organ damage are presented in Table.2
Management of AHE
According to JNC-7, the treatment of hypertensive emergencies include immediate intervention with a goal of reducing SBP by 10-15%, but no more than 25% within the first hour. Reduction of the absolute BP to 160/110 mmHg should be done gradually over the following two to six hours [6,14,15,18,21-25]. In cases of aortic dissection, the SBP should be reduced to less than 120 mmHg within twenty minutes. In hypertensive emergencies associated with ischemic stroke, BP must be decreased to less than 180/110 before thrombolytic therapy may be administered [15,18,22,24,25]. If BP reduction occurs too quickly, there may be a significant decrease in blood flow to tissues resulting in tissue damage and cell death [14,15,18,22]. Since overshooting a target BP (hypoperfusion) is associated with poor results, many treatment protocols require invasive arterial blood pressure monitoring [25,26].
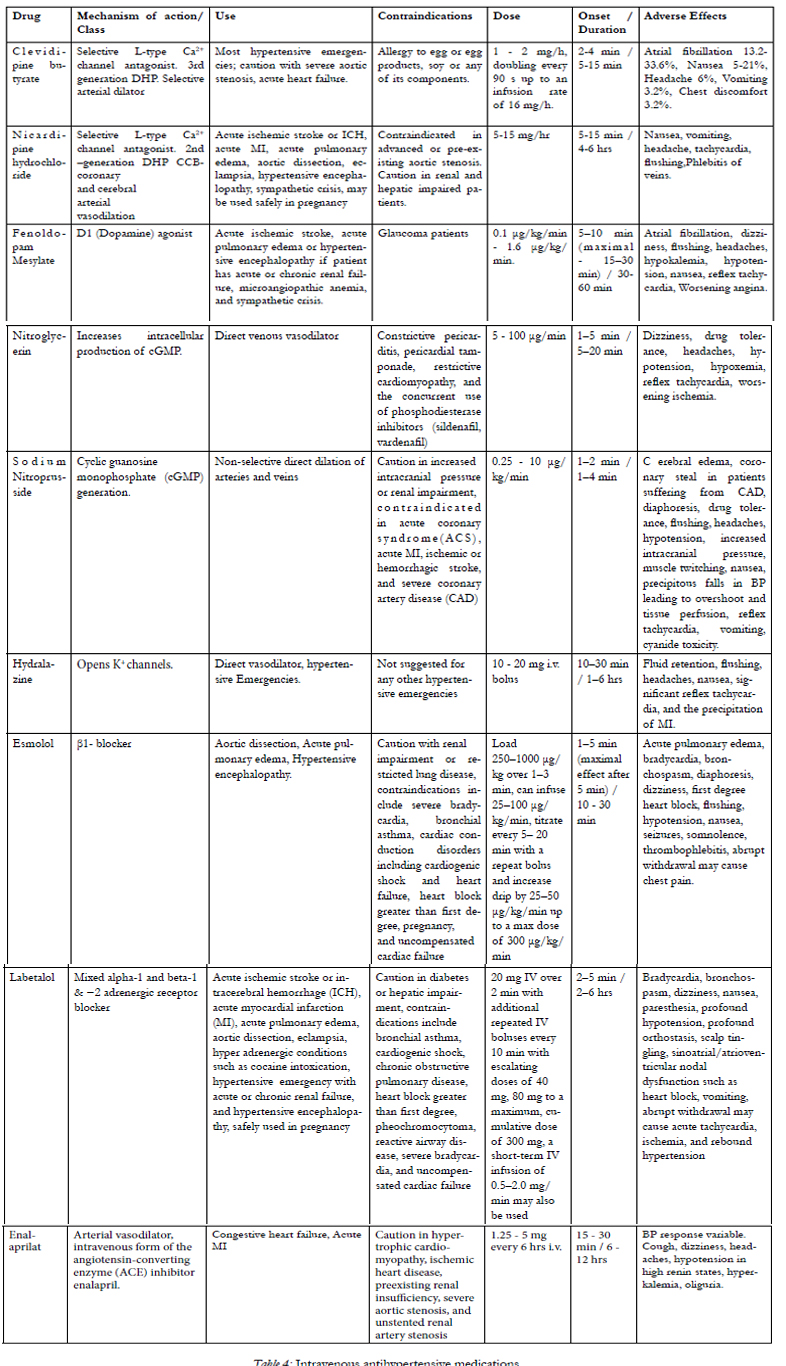
Hypertensive emergencies should be treated aggressively, using quick-onset intravenous medications, whereas hypertensive urgencies do not always require such aggressive treatment. Longer acting oral medications such as labetalol and clonidine may be more appropriate in situations of hypertensive urgency.However, caution should be exercised when using anti-hypertensive agents in the acute setting. An overly aggressive treatment approach may lead to organ hypoperfusion [27]. Once the immediate threat of organ damage is diminished, BP should be gradually controlled to the baseline within a period of 24–48 hours [28]. Characteristics of an ideal intravenous hypertensive agent are shown in Table 3 [29,30]. Currently there are multiple intravenous medications for treatment of acute hypertension in the emergency setting, and their main characteristics, advantages and disadvantages are shown in the Table 4.
Choosing the optimal drug for therapy among multiple intravenous anti-hypertensive agents to treat the AHE depends on the patient’s medical conditions as well as the preference of individual prescribers and institutional guidelines [13,25]. Calcium-channel blockers inhibit the L-type calcium channels, and the subclass of dihydropyridines (Clevidipine, Nicardipine, Nifedipine etc.) are commonly considered as a first-line treatment of hypertensive emergencies because of their strong vasodilatory effects and fewer side effects on cardiac conduction and contractility when compared to classes such as beta blockers [31, 59, 60].
Clevidipine for The Management of Acute Hypertension
Clevidipine is a third-generation, intravenous, dihydropyridine (DHP) calcium-channel blocker (CCB). Clevidipine was approved by the Federal Food and Drug Administration (FDA) of the USA in 2008 for BP reduction when oral therapy is not feasible or desirable [32,33]. Clevidipine is the only intravenous antihypertensive approved by the FDA in the last decade. The drug is an ultrashort-acting vasoselective calcium antagonist for short-term intravenous BP control that acts on L-type calcium channels [34]. These channels regulate the influx of calcium ions into the arterial smooth muscle cells during depolarization, and target excitable tissues, such as cardiac and smooth muscle, blood cells and neurons [32,34]. Clevidipine has high vascular selectivity, reducing the blood pressure by selectively dilating arterioles and reducing peripheral resistance, thus increasing stroke volume and cardiac output. It is composed of the two enantiomers S- and R-clevidipine. Both components of this racemic mixture are equally potent and produce a similar antihypertensive effect [35,36]. Clevidipine is formulated as a sterile, milky-white, ready-to-use lipid intravenous emulsion that is almost insoluble in water and has a pH of 6.0 – 8.0 [35,36]. Clevidipine is formulated in an oil-in-water or 20% lipid emulsion of soybean oil (200 mg/ml), contains approximately 0.2 g of fat per milliliter (2.0 kcal/ml), glycerin (22.5 mg/ml), purified egg yolk phospholipids (12 mg/ml), and sodium hydroxide to adjust pH.35 Strict asepsis must be maintained during administration of clevidipine infusion, because it contains phospholipids and can support microbial growth. If any contamination is suspected, the medication must be discarded. Once the stopper is punctured, the drug must be used within 12 hours. Clevidipine is administered as an intravenous infusion and is rapidly metabolized by plasma and extravascular esterases to an inactive carboxylic acid metabolite and formaldehyde molecules. It reaches a steady-state arterial blood concentration (Vdss 0.17 l/kg) quickly during infusion, and the concentration declines rapidly post infusion, regardless of the infusion length, resulting in a rapid onset as well as rapid recovery of effect [37]. 99.7% of clevidipine is protein bound in plasma at body temperature, though no concentration dependent protein binding of clevidipine has been observed. Clevidipine is considered a high-clearance drug with a mean blood clearance (CLb), independent of body weight, of 0.142 1/min/kg, resulting in extremely short initial (1.6 min) and terminal (15.5 min) half-lives. The relatively short termination of therapeutic effect is primarily related to the elimination rate of the compound than its redistribution.The contribution of the metabolism in blood (as opposed to tissues) to the total elimination of clevidipine is considered to be less than 10%. This emphasizes the high esterase activity in the extravascular tissues. The elimination of clevidipine is not affected by hepatic or renal dysfunction. Clevidipine is excreted primarily in the urine (63 – 74%) and feces (7 – 22%) within the first 72 h after administration [38].
Clevidipine is administered as an intravenous infusion via a peripheral or central venous catheter. The medication is contraindicated in patients with allergies to soy products, eggs, and egg products, or defective lipid metabolism. The onset of action of intravenous clevidipine
is within 2-4 minutes,when it produces a 4-5% decrease in SBP. After initiation of clevidipine intravenous infusion, the clevidipine plasma concentration decreases in a biphasic pattern. During the first phase, 85-90% of clevidipine is eliminated with an elimination half-life of 1 minute and a terminal half-life of 15 minutes [32]. Clevidpine is a specific arteriolar dilator, causing a decrease in arterial resistance and leading to a decrease in mean BP [37]. Because of selective action on arteriols, clevidpine has minimal or no effects on heart rate, myocardial contractility, conduction, cardiac output and stroke volume [18,28].
Owing to the drug’s pharmacological profile, which includes rapid onset of action, small volume of distribution, and high clearance, clevidipine can be considered an ideal agent in patients with AHE. Clinical studies with clevidipine have consistently demonstrated its safety and efficacy in patients with acute hypertensive episodes requiring treatment with parental antihypertensive medications.
Pharmacodynamic and pharmacokinetic properties of clevidipine had been evaluated in healthy volunteers in clinical trials. Intravenous clevidipine was shown to have a short duration of action, short half-life, high clearance rate and fast elimination. Clevidipine was rapidly titratable, safe and well tolerated [37,38,39].
In Phase II trials, racemic formulation of clevidipine was preferred over the individual enantiomer formulations to achieve clinical efficacy in patients with essential hypertension. The trials also demonstrated the dose dependent relationship between clevidipine and BP (SBP & DBP) reduction; which allowed for predictable dose responses and a more efficient control of hypertension to desired BP ranges [40,41].
During the Phase III trials with clevidipine, the medication
was evaluated in patients undergoing elective cardiac surgery. In these trials, clevidipine efficiently reduced the MAP, and systemic vascular resistance (27% at maximal infusion rate) in a dose-dependent fashion. Clevidipine infusion was associated with hemodynamic stability (heart rate, central venous pressure and cardiac index). No changes in cardiac lactate metabolism were revealed in doses used to treat the systemic hypertension [42,43,44].
Levy et al., and Singla et al., studied clevidipine in two randomized double blind, placebo controlled trials in preoperative and postoperative cardiac surgery patients respectively (ESCAPE1 and ESCAPE2). In both studies the target SBP was achieved within 4-7 minutes, the success rate of controlling the SBP was 92.5% and 91.8% respectively [45,46]. Aronson S et al., in their large randomized controlled trial (ECLIPSE trial) evaluated the efficacy of clevidipine compared with sodium nitroprusside, nitroglycerin (perioperatively), or nicardipine (postoperatively) in elective cardiac surgery patients. In this study, the clevidipine group achieved superior BP control compared with other drugs. Clevidipinetherapy was also associated with a significantly lower 30-day postoperative mortality than sodium nitroprusside [47]. In the VELOCITY trial (Pollack et al.), clevidipine’s safety and efficacy were evaluated in patients with severe hypertension in the emergency room or intensive care unit. In these patients, the clevidipine infusion continued for up to 96 hours. In 88.9% of patients the target SBP achieved within 30minutes (median time - 10.9 minutes). Only 1.6% of patients experienced decrease in SBP below the target range. At least one adverse effect was observed in 39.7% of patients, with 8.7% of them considered serious. Adverse effects (in descending order) included headache, nausea, chest discomfort, and vomiting [48].
The ACCELERATE trial (Graffagnio et al.), was designed as a multicenter, single arm study. It evaluated the use of clevidipine for the management of severe hypertension in patients with intracerebral hemorrhage.Clevidipine was effective in reducing the SBP to a target range of less than 160 mm Hg in 97% patients within 3-10 minutes [49]. Bekker et al., evaluated the use of clevidipine in controlling the perioperative hypertension in elective neurosurgical patients. They found that SBP values were reduced to target levels within 15minutes in 78.6% of times after initiation of infusion. The researchers concluded that clevidipine was an effective and safe drug that could be effectively used to control the perioperative hypertension in patients undergoing intracranial procedures [50]. Varelas PN et al., evaluated the role of clevidipine in their open-label pilot study in patients undergoing clipping or coiling of aneurysm for acute hypertension in patients with subarachnoid hemorrhage. They concluded that clevidipine controlled the SBP in < 22 minutes and kept the values within the elective range 70% of the time without major complications [51].
Clevidipine can also be used for intraoperative BP control in patients with suspected diagnosis of pheochromocytoma or during elective pheochomocytoma resection. In both case reports the BP was well controlled with clevidipine infusion [52,53]. Further studies with larger numbers of patients will be required to assess clevidipine efficacy in this patient group relative to the traditionally used medications.
It is not clear whether, clevidipine is safe during pregnancy,labor and breast feeding. It is known that the drug is being excreted in milk. In animals, clevidipine increases fetal and maternal mortality and the gestational duration. Until better evidence becomes available, clevidipine during pregnancy may be recommended for use only in those cases when treatment benefits will clearly outweigh the potential risks [32].
The safety of clevidipine in pediatric population (< 18 years) is unknown. The drug’s use in pediatric patients has been reported in case reports and retrospective reviews, however, no prospective studies have been conducted. Clevidipine-based therapy of hypertension in pediatric population was first reported in a 16-year old patient to control AH during the urgent placement of a peritoneal dialysis catheter [54]. Taking into account the pharmacokinetic and pharmacodynamic properties of clevidipine in adult population, it has been used in pediatric spine and cardiac surgery to provide controlled hypotension and control perioperative AH respectively. The lowest age of the pediatric patient treated with clevidipine in these retrospective analyses was 11 months. This drug has proved its efficacy in adequately controlling BP with minimal side effects [55,56,57]. Clevidipine’s pharmacokinetic profile in children is similar to that for adults showing rapid metabolism by non-specific blood and tissue esterase’s with a short half-life (< 1min) [58]. Nevertheless, properlydesigned prospective clinical trials are needed to evaluate the efficacy, safety and side effects of clevidipine in the pediatric population.
Disclosure
The authors have not received any funding and have no conflict of interests related to this publication.
References
- Vuylsteke A, Feneck RO, Jolin-Mellgård A, Latimer RD, Levy JH, et al. Perioperative blood pressure control: a prospective survey of patient management in cardiac surgery. J Cardiothorac Vasc Anesth 2000;14:269-73.
- Varon J, Marik PE. Perioperative hypertension management. Vasc Health Risk Manag 2008;4:615-27
- Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension.2003; 42(6):1206– 1252
- Hajjar I, Kotchen TA. Trends and prevalence, awareness, treatment, and control of hypertension in the United States, 1998 – 2000. JAMA 2003;290:199-206
- Rosamond W, Flegal K, Furie K, Go A, Greenlund K, et al. American Heart Association Statistics Committee and Stroke Statistics Subcommittee: A Report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2008;117;e25-e146
- Kuppasani K, Reddi AS. Emergency or urgency? Effective management of hypertensive crises. JAAPA. 2010; 23:44–49. [PubMed: 20821913]
- Owens WB. Blood pressure control in acute cerebrovascular disease. J Clin Hypertens (Greenwich). 2011; 13(3):205–211
- Handler J. Hypertensive urgency. J Clin Hypertens 2006;8:61-4
- Ahuja K, Charap MH. Management of perioperative hypertensive urgencies with parenteral medications. J Hosp Med. 2010; 5(2):E11–E16.
- Goldman L, Caldera DL. Risks of general anesthesia and elective operation in the hypertensive patient. Anesthesiology 1979;50:285- 92
- Wolfsthal SD. Is blood pressure control necessary before surgery? Med Clin North Am 1993;77:349-63
- Kessler CS, Joudeh Y. Evaluation and treatment of severe asymptomatic hypertension. Am Fam Physician. 2010; 81(4):470–476
- Belsha, CW. Management of hypertensive emergencies. In: J.T.Flynn et al. (eds.), Pediatric Hypertension, Clinical Hypertension and Vascular Diseases: Pediatric Hypertension. New York: Springer Science+Business Media; 2013. p. 557-571
- Smithburger PL, Kane-Gill SL, Nestor BL, Seybert AL. Recent advances in the treatment of hypertensive emergencies. Crit Care Nurse. 2010; 30(5):24–30
- Varon J. Treatment of acute severe hypertension: current and newer agents. Drugs 2008;68:283-97
- Varon J, Marik PE. Clinical review: the management of hypertensive crises. Crit Care. 2003; 7(5):374–384
- Vaughan CJ, Delanty N. Hypertensive emergencies. Lancet. 2000 Jul 29;356(9227):411-7.
- Polly DM, Paciullo CA, Hatfield CJ. Management of hypertensive emergency and urgency. Adv Emerg Nurs J. 2011; 33(2):127–136
- Katz JN, Gore JM, Amin A, Anderson FA, Dasta JF, et al. Practice patterns, outcomes, and end-organ dysfunction for patients with acute severe hypertension: the Studying the Treatment of Acute hyperTension (STAT) registry. Am Heart J. 2009; 158(4):599–606
- Vaughan CJ, Delanty N. Hypertensive emergencies. Lancet. 2000; 356(9227):411–417
- Aggarwal M, Khan IA. Hypertensive crisis: hypertensive emergencies and urgencies. Cardiol Clin. 2006; 24(1):135–146
- De Gaudio AR, Chelazzi C, Villa G, Cavaliere F. Acute severe arterial hypertension: therapeutic options. Curr Drug Targets. 2009; 10(8):788–798
- Flanigan JS, Vitberg D. Hypertensive emergency and severe hypertension: what to treat, who to treat, and how to treat. Med Clin North Am. 2006; 90(3):439–451
- Hays AJ, Wilkerson TD. Management of hypertensive emergencies: a drug therapy perspective for nurses. AACN Adv Crit Care. 2010; 21(1):5–14
- Pollack CV, Varon J. Hypertensive emergencies: acute care evaluation and management. Emergency Medicine Cardiac Research and Education Group International. 2008; 3:1–9.
- Rhoney D, Peacock WF. Intravenous therapy for hypertensive emergencies, part 1. Am J Health Syst Pharm 2009;66:1343-52
- Rodriguez MA, Kumar SK, De Caro M. Hypertensive crisis. Cardiol Rev. 2010; 18(2):102–107
- Peacock WF, Angeles JE, Soto KM, et al. Parenteral clevidipine for the acute control of blood pressure in the critically ill patient: a review. Ther Clin Risk Manag 2009;5:627-34
- Levy JH. Treatment of perioperative hypertension. Anesthesiol Clin North Am 1999;17:567-79
- Oparil S, Aronson S, Deeb GM, et al. Fenoldopam: a new parenteral antihypertensive: consensus roundtable on the management of perioperative hypertension and hypertensive crises. Am J Hypertens 1999;12:653-64
- Eisenberg MJ, Brox A, Bestawros AN. Calcium channel blockers: an update. Am J Med. 2004; 116:35–43
- The Medicines Company Cleviprex® (Clevidipine Butyrate) injectable emulsion for intravenous use: US prescribing information [online] Available from: www.cleviprex.com/pdf [Last accessed 8th December 2011]
- CDER New Molecular Entity (NME) Drug and New Biologic Approvals for Calendar Year 2008 FDA Public Health Advisory. Washington, DC: FDA/Center for Drug Evaluation and Research, 2008. Available from: UCM081805.pdf[Last accessed 16 September 2009]
- Nordlander M, Sjöquist PO, Ericsson H, Rydén L. Pharmacodynamic,pharmacokinetic and clinical effects of clevidipine, an ultrashort-acting calcium antagonist for rapid blood pressure control.Cardiovasc Drug Rev 2004;22:227-50
- Noviawaty I, Uzun G, Qureshi AI. Drug evaluation of clevidipine for acute hypertension. Expert Opin Pharmacother 2008;9:2519- 29. Characteristics of clevidipine.
- Sorbera LA, Castanera J. Clevidipine. Drugs Future 2004;29:105-11
- Ericsson H, Fakt C, Jolin-Mellgård A, Nordlander M, Sohtell L, et al. Clinical and pharmacokinetic results with a new ultrashortacting calcium antagonist, clevidipine, following gradually increasing intravenous doses to healthy volunteers. Br J Clin Pharmacol 1999;47:531-8
- Ericsson H, Fakt C, Höglund L, Jolin-Mellgård A, Nordlander M, et al. Pharmacokinetics and pharmacodynamics of clevidipine in healthy volunteers after intravenous infusion. Eur J Clin Pharmacol 1999;55:61-7
- Prlesi L, Cheng-Lai A. Clevidipine: a novel ultra-short-acting calciumantagonist. Cardiol Rev 2009;17:147-52
- Schwieler JH, Ericsson H, Löfdahl P, Thulin T, Kahan T. Circulatory effects and pharmacology of clevidipine, a novel ultra short acting and vascular selective calcium antagonist, in hypertensive humans.J Cardiovasc Pharmacol 1999;34:268-74
- Ericsson H, Bredberg U, Eriksson U, Jolin-Mellgård A, Nordlander M, et al. Pharmacokinetics and arteriovenous differences in clevidipine concentration following a short- and a long-term intravenous infusion in healthy volunteers. Anesthesiology 2000;92:993-1001
- Kieler-Jensen N, Jolin-Mellgård A, Nordlander M, Ricksten SE. Coronary and systemic hemodynamic effects of clevidipine, an ultra-short-acting calcium antagonist, for treatment of hypertension after coronary artery surgery. Acta Anaesthesiol Scand 2000;44:186-93
- Ericsson H, Schwieler J, Lindmark BO, Löfdahl P, Thulin T, et al. Enantioselective pharmacokinetics of the enantiomers of clevidipine following intravenous infusion of the racemate in essential hypertensive patients. Chirality 2001;13:130-4
- Bailey JM, Lu W, Levy JH, Ramsay JG, Shore-Lesserson L, et al.Clevidipine in adult cardiac surgical patients: a dose-finding study.Anesthesiology. 2002 May;96(5):1086-94.
- Levy JH, Mancao MY, Gitter R, Kereiakes DJ, Grigore AM, et al. Clevidipine effectively and rapidly controls blood pressure preoperatively in cardiac surgery patients: the results of the randomized, placebo-controlled efficacy study of clevidipine assessing its preoperative antihypertensive effect in cardiac surgery-1. Anesth Analg 2007;105:918-25
- Singla N, Warltier DC, Gandhi SD, Lumb PD, Sladen RN, et al. ESCAPE- 2 Study Group. Treatment of acute postoperative hypertension in cardiac surgery patients; an efficacy study of clevidipine assessing its postoperative antihypertensive effect in cardiac surgery-2 (ESCAPE-2), a randomized double-blind, placebo-controlled trial.Anesth Analg 2008;107:59-67
- Aronson S, Dyke CM, Stierer KA, Levy JH, Cheung AT, et al. The ECLIPSE trials: comparative studies of clevidipine to nitroglycerin, sodium nitroprusside, and nicardipine for acute hypertension treatment in cardiac surgery patients. Anesth Analg. 2008; 107(4):1110–1121.
- Pollack CV, Varon J, Garrison NA, Ebrahimi R, Dunbar L, et al.Clevidipine, an intravenous dihydropyridine calcium channel blocker,is safe and effective for the treatment of patients with acute severe hypertension. Ann Emerg Med 2009;53:329-38
- Graffagnino C, Bergese SD, Love J, Schneider D, Lazaridis C, et al. Clevidipine rapidly and safely reduces blood pressure in patients with acute intracerebral hemorrhage: The ‘ACCELERATE’ Trial.Cerebrovasc Dis. 2013;36(3):173-80.
- Bekker A, Didehvar S, Kim S, Golfinos JG, Parker E, et al. Efficacy of clevidipine in controlling perioperative hypertension in neurosurgicalpatients: initial single-center experience. J Neurosurg Anesthesiol.2010 Oct;22(4):330-5
- Varelas PN, Abdelhak T, Corry JJ, James E, Rehman MF, et al.Clevidipine for Acute Hypertension in Patients with Subarachnoid Hemorrhage: a Pilot Study. Int J Neurosci. 2013 Sep 6.
- Kline JP. Use of clevidipine for intraoperative hypertension caused by an undiagnosed pheochromocytoma: a case report. AANA J. 2010 Aug;78(4):288-90
- Bettesworth JG, Martin DP, Tobias JD. Intraoperative use of clevidipine in a patient with von Hippel-Lindau disease with associated pheochromocytoma. J Cardiothorac Vasc Anesth. 2013 Aug;27(4):749-51
- Tobias JD, Allee J, Ramachandran V, Groshong T. Clevidipine controls intraoperative blood pressure in an adolescent with renal failure. J. Pediatr. Pharmacol. Ther. 2009, 14, 144–147v
- Towe E, Tobias JD, Preliminary experience with clevidipine in the pediatric population. J. Intensive Care Med. 2010, 25, 349–352
- Tobias JD, Schechter WS, Phillips A, Weinstein S, Michler R, et al. Clevidipine for perioperative blood pressure control in infants and children undergoing cardiac surgery for congenital heart disease. J. Pediatr. Pharmacol. Ther. 2011, 16, 55–60
- Tobias JD, Hoernschemeyer DG. Clevidipine for controlled hypotension during spinal surgery in adolescents. J. Neurosurg. Anesth. 2011, 23, 347–351
- Joseph D. Tobias, David B. Tulman, and Sergio D. Bergese. Clevidipine for Perioperative Blood Pressure Control in Infants and Children. Pharmaceuticals (Basel). 2013 January; 6(1): 70–84.
- Bergese SD, Puente EG. Clevidipine butyrate: a promising new drug for the management of acute hypertension. Expert Opin Pharmacother. 2010 Feb;11(2):281-95.
- Tulman DB, Stawicki SP, Papadimos TJ, Murphy CV, Bergese SD.Advances in management of acute hypertension: a concise review.Discov Med. 2012 May;13(72):375-83. Review.