Early Stages of Chronic Kidney Disease and Physical Growth in Children - A Hospital Based Study
Sasi Vaithilingan*
Professor and HOD, Department of Pediatric Nursing, Vinayaka Mission’s College of Nursing, Puducherry, India.
Vinayaka Missions Research Foundation, Salem, India.
*Corresponding Author
Dr. Sasi Vaithilingan, Ph.D(N),
Professor and HOD, Department of Pediatric Nursing,
Vinayaka Mission’s College of Nursing, Puducherry, India.
Vinayaka Missions Research Foundation, Salem, India.
E-mail: sasicoumar@gmail.com
Received: April 16, 2019;Accepted: December 17, 2019;Published: December 24, 2019
Citation: Sasi Vaithilingan. Early Stages of Chronic Kidney Disease and Physical Growth in Children - A Hospital Based Study. Int J Translation Community Dis. 2019;7(2):112-117. doi : dx.doi.org/10.19070/2333-8385-1900018
Copyright: Sasi Vaithilingan© 2019. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Growth retardation is the common complication in children with chronic kidney disease. As it develop in early stages of the disease, growth monitoring is essential.
Methods: The observational study was carried out in Institute of Child Health, Chennai. A total of 125 children with stage 2 to stage 4 were included in the study. The growth parameters were measured and plotted in growth charts prescribed by Indian Academy of Pediatrics.
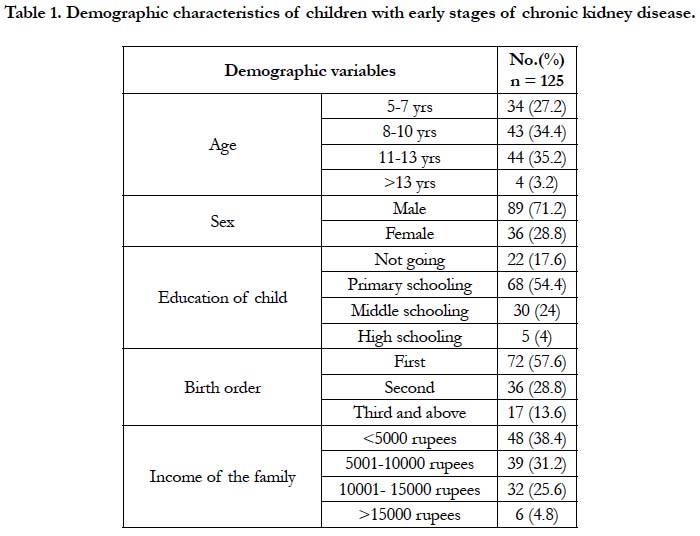
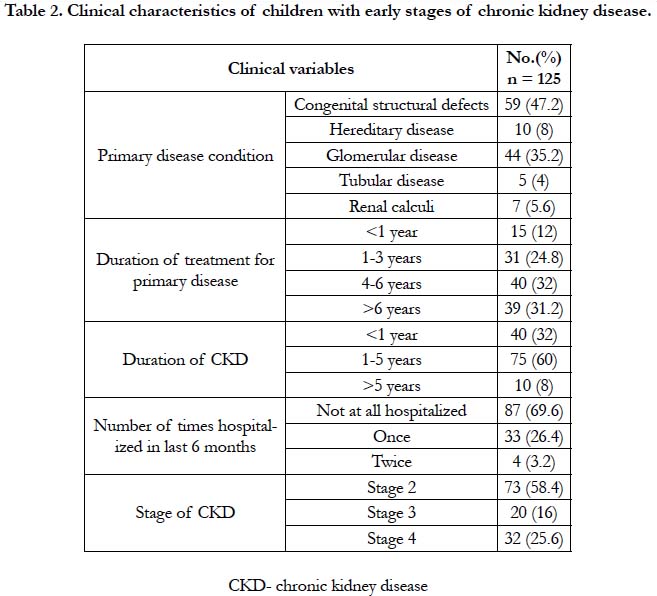
Results: Among 125 children with chronic kidney disease, 89(71.2%) of them were boys with ratio of 2.4:1. The mean age at presentation was 9.27 ± 2.57. Congenital structural defects found in 59(47.2%) and 44(35.2%) children with glomerular disease. Majority, 73(58.4%) were in stage 2, 20(16%) in stage 3 and 32(25.6%) in stage 4. The mean duration of diagnosis of Chronic Kidney Disease was 2.4 years. The mean height at presentation was 117.6 cm. Severely stunted growth among 59 (43.7%) children with height < 3rd percentile. Likewise, 55(44%) were underweight. The mean weight at presentation was 21.43kg. For Body Mass Index, 66(52.8%) children were <25th percentile (wasted), in which 49 (55.1%) were males. Height was associated with age and stage. Weight was associated with primary disease, duration of Chronic Kidney Disease, stage and birth order. Body Mass Index of these children was strongly associated with primary disease.
Conclusion: Regular monitoring of physical growth in children with chronic kidney disease is essential to identify the progress of disease and initiate appropriate management in these children.
2.Patients and Methods
2.1 Statistical Analysis
3.Result
3.1 Growth Parameters
3.2 Association of Variables
4.Discussion
5.Acknowledgement & Funding Source
6.References
Introduction
Chronic kidney disease (CKD) is a continuum of disease with mild renal insufficiency which progress to End Stage Renal Disease [1]. The incidence of pediatric patients at pre dialysis CKD ranges from 5.7 to 12 cases per million of age related population [2]. Growth retardation is the most common problem seen in children with CKD [3]. It develops early in the course of CKD [1] and affects upto 35% of these children, significant proportion of children present with severe growth retardation by the time of renal transplantation [4].
Healthy children exhibit a maximal growth velocity in infants, followed by a period of flattened growth and thereafter a growth spurt during puberty. In children with CKD, the normal growth pattern gets altered. The adolescents with CKD showed pubertal delay with reduction of total pubertal gain [5, 6].
Multiple etiology is responsible for growth retardation includes modifiable factors like anorexia, caloric deficits, hyposthenuria, salt wasting, anemia, metabolic acidosis, mineral depletion and less modifiable factors such as growth hormone resistance, renal osteodystrophy, therapies and non-modifiable risk factor as age at the onset of CKD [7-9]. The technological and pharmacological advancements in the field of nephrology created positive impact on prolonging the survival of children with CKD. Despite these advances, the CKD children continue to grow poorly [10]. Altered growth hormone and organ resistance to growth hormone are the major contributors for the growth retardation in CKD [11]. Moreover, pediatric registry data showed increased mortality rate in children with poor growth [12, 13].
Data from NAPRTCS 2005 database revealed that 37% of children with CKD had statutory growth impairment. Even those with mild reduction of GFR, exhibited short stature [14]. Moreover, the growth velocity can be restored with intensive nutritional and medical intervention to achieve normal adult height [15]. The National Kidney Foundation’s Clinical Practice guideline recommended regular monitoring of growth in children with CKD [16]. However, many of the studies reported the growth pattern of children in dialysis, transplantation and with growth hormone therapy [17-19].
India has started the CKD registry for the adults and Indian CKD guidelines are available to treat adult population [20]. The database for Indian children with CKD is lacking and moreover, there is lack of published data on the growth of children with early stages of CKD. Thus, the present study aimed to identify the growth (height, weight and BMI) of children with early stages of CKD and to explore the associative factors for impaired growth in in these children.
Patients and Methods
The study was approved by institutional ethical committee of Rajiv Gandhi Government General Hospital, Chennai. The observational study was carried out in Institute of Child Health, Egmore, Chennai. Written informed consent from caregivers and Child’s assent from children >12 years of age was obtained for them to participate in the study. From November 2013 – February 2014, a total of 125 children were identified with CKD (stage 2 to stage 4) from their anthropometric measurements and GFR was determined by Schwartz formula [21]. The study included children between 5-15 years of age, in stage 2 to stage 4 CKD and seek services from pediatric nephrology OPD with minimal follow up of 6 months. Children with malignancy, dialysis and with multiple complications were excluded from the study.
The demography of the child like age, sex, education of the child, birth order were collected from the parent’s and details of clinical variables like primary diagnosis, duration of the treatment, duration of CKD, hospitalization in past 6 months were obtained from child’s medical record. The staging of CKD was done according to CKD classification described by the clinical practice guidelines of NKF/KDOQI [16]. The growth parameters were measured as per the recommended procedure by KDOQI guidelines. Standing height was considered as the average of three measurements to the nearest millimeter using wall mounted stadiometer. Weight was measured to the nearest 0.1 kg on calibrated digital scale. BMI was determined by dividing weight in Kilogram by height in meter square. The observed values of height and weight were plotted in the age and sex specific growth charts. Where as, the BMI was plotted in height, age and sex specific BMI charts prescribed by Indian Academy of Pediatrics 2007 [22] to interpret the observed measurements in percentiles.
Database in the statistical package SPSS version 17.0 was developed. The categorical variables were presented in number and percentage and continuous variable in mean and standard deviation. The Chi square test was applied to identify the factors associated with impaired growth. Values of P<0.05 were considered as statistically significant.
Results
In table 1, among 125 children identified, majority 89 (71.2%) of them were boys and 36 (28.8%) were girls. The male and female ratio is 2.4:1. The mean age of the children at presentation was 9.27 ± 2.57, in which maximum of children were in the age group of 8-13 yrs. Half of these children were in primary schooling whereas, 22 (17.6%) of them were dropped out from school. About one third of these children were from poor economic status with monthly family income <5000 rupees.
In table 2, Congenital structural defect was the main cause for CKD in 59(47.2%) children and 44(35.2%) of them presented with glomerular disease. These children were classified for the CKD stage based on KDOQI criteria. It revealed majority of them 73 (58.4%) were in stage 2, 20 (16%) with stage 3 and 32 (25.6%) with stage 4 CKD. Since the early stages of the CKD are asymptomatic, most of the children 87 (69.6%) were not hospitalized from the time of their primary diagnosis. The mean duration of diagnosis of CKD is 2.4 years. All the 125 children received conservative treatment.
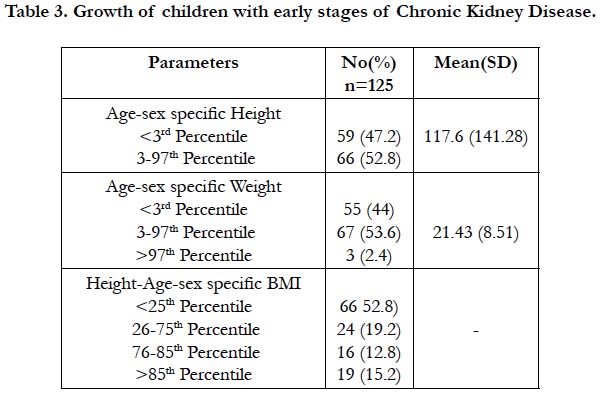
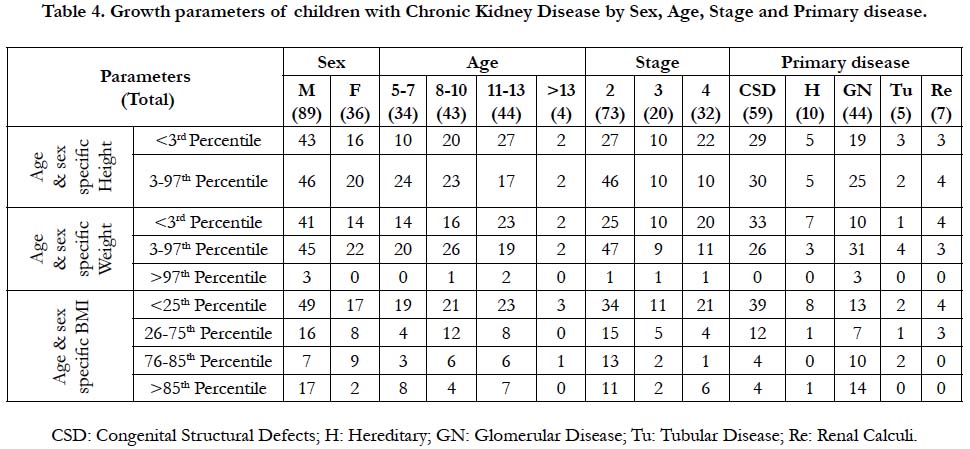
The age and sex specific height, weight and height - age and sex specific BMI of 125 children with CKD were identified and listed in Table 3. The mean height at presentation was 117.6 cm. Nearly, half of these children 59 (43.7%) showed severely stunted growth (< 3rd percentile). It was commonly noticed (table 4) among male children 43(48.3%), also in congenital structural defects 29(49.2%), and in glomerular disease 19(43.2%). The stunting was also observed in children with stage 2 CKD 27(37%) and stage 4 CKD 22 (68.8%). Most of the stunted children with CKD belong to the age group of 8-13 years.
Though 67(53.6%) were with normal weight, 55(44%) were underweight with <3rd percentile for age and sex specific weight. The mean weight at presentation was 21.43 kg. The underweight was seen in male 41 (46.1%) than female children 14 (38.9%). In Table 4 among 55 underweight children, half of them 23/44 (52.3%) were in 10-13 years of age. Moreover, under-weight was remarkably seen in children with stage 2 CKD 25/73(34.5%) and with stage 4 CKD 20/32 (62.5%). Even children with congenital structural defects 33/59 (55.9%) found to have under-weight. Among 125 children with CKD, 3(2.4%) of them were obese and presented with glomerular disease.
Table 4. Growth parameters of children with Chronic Kidney Disease by Sex, Age, Stage and Primary disease.
The findings of height, age and sex specific Body Mass Index (BMI) revealed that 66 (52.8%) children with CKD were <25th percentile (wasted), in which 49/66 (55.1%) were males. Whereas, overweight was observed among 19 (15.2%) children with male predominance. In table 4, wasting was noted in children with stage 2 CKD 34/73 (46.6%) and stage 4 CKD 21/32 (65.6%). Moreover, CKD children with congenital structural defects showed wasting 39/59 (66.1%). Only 24(19.2%) of children with CKD had normal BMI.
Association of height with demographic variables noticed statistically significant with age at p<0.05 which infers that earlier the onset of disease condition resulted in retarded growth. With the clinical variables, the stage of CKD was strongly associated with height at p<0.001 which means that later the stages more the height impairment. Weight of children was associated with birth order at p<0.01, primary disease at p<0.05, duration of CKD at p<0.05, stage of CKD at p<0.05, which infers that congenital structural defects with prolonged duration of CKD and progress in CKD stage affect the weight of these children. BMI of CKD children was strongly associated with primary disease at p<0.001 and with sex at p<0.05.
Discussion
The true prevalence of growth of children with early stages of CKD (stage 2 - stage 4) in our population remains grey due to lack of central reporting system [1]. In India, there is no specific CKD screening programme for children to identify the cases early. Moreover, lack of health care facilities, trained health care professionals, and knowledge about CKD, socio economic factors, high illiteracy and long distance to travel for treatment contribute to the failure of timely diagnosis and referral to the tertiary hospitals and leads to large number of attrition during follow up [23]. Thus failed to identify and monitor the growth of children with CKD regularly. This report from a single tertiary center provides the best possible available data on growth of children in early stages of chronic kidney disease (stage 2 – stage 4) that was observed in specific time period.
On screening the children in the Pediatric Nephrology OPD, 125 children were presented with CKD under various stages (stage 2-stage 4). In the present study, children with GFR <90 ml/min/1.73m2 were considered to have CKD and classified based on KDOQI guidelines [16]. Though many studies [24, 25] considered GFR <75 ml/min/1.73m2 as cutoff for CKD, Bhimma and Salevic used the GFR level similar to our study [26, 27].
This study observed that CKD was predominant among male children and the mean age at presentation was 9.27 ± 2.57. It was consistent with findings of Thomas[3]. Also, Pstrusinka in their study documented 82.23% of children with CKD were males [28]. Wong identified the mean age of children with CKD as 9.9 ± 5.1 which supported the present findings [12].
Present study noticed that 17.6% of children with CKD were drop out from school due to their ill health and frequent visit to hospital. Supportively Shitza and Shkruti demonstrated 13% of dropout in their study [29]. Despite this, our children were from poor economic status and a similar finding wasreported by Marciano [30]. Congenital structural defect was the leading cause of CKD in the present study. Whereas, recent study31demonstrated more number of children with glomerulonephritis than structural defects. A retrospective study findings by Soares [32] documented decline in median rate of GFR (10ml/min/1.73m2 per year) was more in children with glomerular nephritis in comparison with congenital nephro-uropathies. The NAPRTCS report [33] revealed structural defect predominance in younger children along with increased incidence of Glomerulonephritis in older children which is in par with present findings.
In a retrospective study, Bhimma [26], demonstrated that out of 71, 17 were in stage 2, 20 were in stage 3 and 21 were in stage 4 CKD. Odentunde and Wong identified similar results and consistent to present study findings [34, 35]. As the early stages of CKD are asymptomatic many of the cases failed to get reported and hospitalized. Here also 87 children with CKD were not hospitalized from the time of diagnosis of primary disease. Furth [36] reported higher hospitalization rates among children with severe and moderate CKD than those with normal growth. McKenna documented 10% of children with CKD were hospitalized and the remaining were managed conservatively [37]. Moreover, the mean duration of diagnosis of CKD in the present study was 2.4 years, where as, it was 6.91 years in Pstrusinka’s study [28].
Here, the growth parameters of the children with CKD were interpreted based on Indian growth reference standard percentile charts. The Age and Sex specific height of children with CKD of the present study identified 59 stunted children (<3rd percentile) with male predominance and congenital structural defects. Also observed more number of stunted children in stage 2 CKD. Zivienjak, in their seven year prospective study among 190 boys with congenital or hereditary CKD, documented stage 2 CKD by 10 years of their age [6]. Likewise, Rodig reported severe short stature in 12% of children with CKD particularly with non- glomerular CKD [9]. Where as, Wong observed increased prevalence of growth failure with increasing stage of CKD (P=0.0001) [12]. However, Salevic reported that the mean Ht SDS of children with stunting was – 3.00 [27] and supportive to the present study.
This study children not only presented with height deficit, even for their age and sex specific weight, 55 of them were underweight (<3rd percentile). The underweight was markedly noticed among male children, with congenital structural defects and with stage 2 CKD and stage 4 CKD. The dip in the weight of the CKD children was due to muscle wasting. Overweight was observed in 3 children with glomerular disease which may be the manifestations of the condition and treatment they receive for it. Rodig, demonstrated that shorter the duration of CKD and high prevalence of edema were the factors associated with increased weight in glomerular diseased CKD children [9]. The Height-age-sex specific BMI of the CKD children in this study identified wasting in 66 children with male predominance and also in children with stage 2 CKD.
It was evident from the present study that the variables age and stage of CKD were associated with height percentile. The primary diseases, duration of CKD, stage of CKD and birth order were associated with weight percentile. The Height-age-sex specific BMI was strongly associated with primary disease. Rodig reported that a 10ml/min/1.73 m2 decrease in GFR was associated with an average decrease in height SDS of 0.14 in male children and 0.12 SDS in female children, decrease in weight SDS of 0.09 in males and 0.15 SDS in females with glomerular CKD as well as in males 0.13 SDS and females 0.13 SDS with non-glomerular CKD [9]. Even, Salevic found significant difference in growth with different CKD stages (P<0.001) and identified primary diagnosis, CKD stages and therapy modality are predictors of CKD [27]. The above findings were more consistent with present study findings.
Though it was a single tertiary center study, stage 2 CKD was predominant with growth deficit which lies as an iceberg yet. As parental height has its effect on children height, it is not possible to determine the physical growth deficit with the impact of disease alone. Thus further research is needed to correlate the parental height with the children height. However, this finding gives alarm to have regular screening of children with CKD and monitoring the growth parameters from the time of identification and early initiation of treatment to prevent further complications.
Acknowledgement & Funding Source
I would like to acknowledge Dr. R. Padmaraj, Senior Nephrologist, ICH, Chennai and Dr. S. Kamala, Professor, Rani Meyyammai College of Nursing for their guidance towards the study. Financial support was not obtained.
References
- Marks DS, Sinha R. Pediatric Chronic Kidney Disease in Principles and Practice of Pediatric Nephrology. Second Edition, New Delhi. Jaypee Brothers Medical Publishers (P) Ltd. 643-669.
- Harambat J, van Stralen KJ, Kim JJ, Tizard J. Epidemiology of chronic kidney disease in children. PediatrNephrol, 2012; 27:363-373. PMID: 21713524.
- Thomas R, Kamath N, Iyenkar A. Assessment of growth and nutritional status in children with Chronic Kidney Disease(CKD): A single centre experience. Karnataka Pediatr J. 2012; 27(3-4): 155-158.
- Fletcher J, McDonald S, Alexander SI. Prevalence of genetic renal disease in children. PediatrNephrol, 2013; 28: 251-256. PMID: 23052649.
- Stefanidis CJ. Growth and Nutrition of children with chronic kidney failure. The Turkish J Pediatr. 2005; 47(S): 9-12.
- Zivienjak M, Franke D, Filler G, Haffner D, Froede K, Nissel R, et al. Growth impairement shows an age-dependent pattern in boys with chronic kidney disease. PediatrNephrol. 2007 Mar; 22(3): 420-9. PMID: 17131161.
- Bacchetta J, Harambat J, Cochat P, Salusky IB, Wesseling-Perry K. The consequences of chronic kidney disease on bone metabolism and growth in children. Nephrol Dial Transplant. 2012; 27: 3063- 3071. PMID: 22851629.
- Greenbaum LA, Warady BA, Furth SL. Current advances in chronic kidney disease in children: Growth, Cardiovascular and Neurocognitive Risk factors. SeminNephrol. 2009; 29(4): 425-434. PMID: 19615563.
- Rodig NM, McDermott KC, Schneider MF, Hotchikiss HM, Yadin O, Seikaly MG, et al. Growth in children with chronic kidney disease: A report from the chronic kidney disease in children study. PediatrNephrol. 2014; 29(10): 1987-1995. PMID: 24728472.
- Lewis M, Shaw J, Reid C, Evans J, Webb N, Verrier-Jones k. growth in children with established renal failure- a registry analysis. Nephrol Dial Transplant. 2007; 22(Suppl 7): vii176-vii180. PMID: 17724045.
- Pirojsakul K, Mathews N, Seikaly MG. Chronic kidney disease in children: recent update. The Open Urology & Nephrology Journal. 2015 Nov 26; 8(1).
- Wong CJ, Moxey-Mims M, Jerry-Fluker J, Warady BA, Furth S. CKiD (CKD in Children) Prospective Cohort Study: A review of current findings. Am J Kidney Dis, 2012; 60(6): 1002-1011. PMID: 23022429.
- Singhal R, Hux JE, Alibhai SMH, Oliver MJ. Inadequate predialysis care and mortality after initiation of renal replacement therapy. Kidney International. 2014; 86: 399-406. PMID: 24552848.
- Seikaly MG, Salhab N, Gipson D, Yiu V, Stablein D. Stature in children with chronic kidney disease: Analysis of NAPRTCS database. Pediatr Nephrol. 2006; 21: 793-799. PMID: 16583244.
- Kim H, Lim H, Choue R. Compromised diet quality is associated with decreased renal function in children with chronic kidney disease. ClinNutr Res. 2014; 3 :142-149. PMID: 25136542.
- Hogg RJ, Furth S, Lemley KV, Portman R, Schwartz GJ, Coresh J, et al. National Kidney Foundation’s Kidney Disease Outcome Initiative Clinical Practice Guidelines for chronic kidney disease in children and adolescents: evaluation, classification and stratification. Pediatrics. 2003; 111: 1416-1421. PMID: 12777562.
- Vachvanichsanong P, Dissaneewate P, McNeil E. Childhood chronic kidney disease in a developing country. PediatrNephrol. 2008; 23:1143-1147. PMID: 18389284.
- Sozeri B, Mir S, Kara OD, Dincel N. Growth impairement and nutritional status in children with chronic kidney disease. Iran J Pediatr. 2011; 21(3): 271-277. PMID: 23056801.
- Ingulli EG, Mak RH. Growth in children with chronic kidney disease: role of nutrition , growth hormone, dialysis and steroids. CurrOpinPediatr. 2014; 26(2):187-192. PMID: 24535500.
- Rajapurkar MM, John GT, Kirpalani AL, Abraham G, Agarwal SK, Almeida AF, Gang S, Gupta A, Modi G, Pahari D, Pisharody R. What do we know about chronic kidney disease in India: first report of the Indian CKD registry. BMC nephrology. 2012 Dec; 13(1): 10.
- Schwartz GJ, Haycock GB, Edelmann CM, Spitzer A. A simple estimate of glomerular filteration rate in children derived from body length and plasma creatinine. Pediatrics. 1976; 58: 259-263. PMID: 951142.
- Khadilkar VV, KhadilkarAV, Choudhury P, Agarwal KN, Ugra D, Shah NK. IAP Growth Monitoring Guidelines for children from birth to 18 years. Indian Pediatr. 2007; 44(3): 187-197. PMID: 17413194.
- Prabakar MR, Chandrasekaran V, Soundarajan P. Epidemic of chronic kidney disease in India- what can be done?. Saudi J Kidney Dis Transpl. 2008; 19(5): 847-853. PMID: 18711313.
- MongHiep TT, Ismaili K, Collart F, Damme-Lombaerts RV, Godefroid N, Ghuysen M, et al. Clinical charactereistics and outcome of children with stage 3-5 chronic kidney disease. PediatrNephrol. 2010; 25: 935-940. PMID: 20148340.
- Rizzoni G, Basso T, Setari M. Growth in children with chronic renal failure on conservative treatment. Kidney International. 1984; 26:52-58. PMID: 6482179.
- Bhimma R, Adhikari M, Asharam K, Connolly C. The spectrum of Chronic kidney disease(stage 2-5) in KwaZulu-Natal, South Africa. Pediatr Nephrol. 2008; 23: 1841-1846. PMID: 18548285.
- Salevic CP, Radovic P, Milic N, Bogdanovic R, Paripovic D, Paripovic A, et al. Growth in children with chronic kidney disease: 13 year follow-up study. J Nephrol. 2014; 27: 537-544. PMID: 24756972.
- Pstrusinska KK, Medynka A, Chmielewska IB, Grenda R, Jozwiak AK, Keszczynska B, et al. Perception of Health related quality of life in children with chronic kidney disease by the patients and caregivers: multicenter Nation Study results. Quality life Res. 2013; 22: 2889-2897. PMID: 23595412.
- Shtiza D, Shkurti E. Performance disorders and quality of life of Albanian children and adolescents with Chronic Kidney Disease. European Scientific Journal. 2013 Nov 1; 9(33): 462-469.
- Mariciano RC, Soares BC, Diniz JS, Lima EM, Silva JMP, Canhestro, et al. Behavioral disorders and quality of life in children and adolescents with chronic kidney disease. Pediatr Nephrol Dial Transplant. 2011; 21: 1899-1905. PMID: 21110044.
- Tan SY, Naing L, Han A, Khalil MAM, Chong VH, Tan J. Chronic kidney disease in children and adolescents in Brunei Darussalam. World J Nephrol. 2016; 5(2): 213-219. PMID: 26981447.
- Soares CMB, Diniz JSS, Lima EM, Oliveira GR, Canhesro MR, Colosimo EA, et al. Predictive factors of progression to chronic kidney disease stage 5 in a pre-dialysis interdisciplinary programme. Nephrol Dial Transplant. 2009; 24: 845-855. PMID: 18840891.
- NAPRTCS. Annual Report. Rockville, MD: EMMES; 2005.
- Odetunde OI, Okafor HU, Uwaezuoke SN, Ezeonwn BU, Adiele KD, Ukaha OM. Chronic kidney disease in children as seen in a tertiary hospital in Enugu, South East Nigeria. Nigerian J Clinic Pediatr. 2014; 17(2): 196-200. PMID: 24553031.
- Wong H, MYIrea K, Feker J, Drukker A, Filler G. Prevalence of complication in children with chronic kidney disease according to KDOQI. Inter SocNephrol. 2009; 70: 585-590.
- Furth SL, Hwang W, Yang C, Neu AM, Fivush BA, Powe NR. Growth failure , risk of hospitalization and death for children with end-stage renal disease. PediatrNephrol, 2002; 17: 450-455. PMID: 12107811.
- McKennaAM, Keating LE, Vigneux A, Stevens S, Williams A, Geary DF. Quality of life in children with chronic kidney disease- Patient and caregivers assessments. Nephrol Dial Transplant. 2006; 21(7): 1899-1905. PMID: 16611686.