From Precursor/Stem Cells to Cordocytic Phenotypes In The Skin
Viorel Pais1*, Emil Pais2
1* Independent Researcher, Bucharest, Romania.
2 Spectral Molecular Imaging, Los Angeles, California, United States.
*Corresponding Author
Viorel Pais,
6 Constantin Radulescu Motru Street,
Bloc 35, Scara B, Apt. 73, Sector 4,
Postal Code 40363, Bucharest, Romania.
Tel: (40)31-409-7790
E-mail: viorelpais@hotmail.com
Article Type: Research Article
Received: January 25, 2014; Accepted: January 23, 2014; Published: January 27, 2014
Citation: Pais V, Pais E. (2014). From Precursor/Stem Cells to Cordocytic Phenotypes In The Skin, Int J Stem Cell Res Transplant, 02(01), 52-58. doi: dx.doi.org/10.19070/2328-3548-140009
Copyright: Pais V© 2013. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
This cytohistopathological study was performed to have a better knowledge of the continuum of cellular events, from mesenchymal stem cells to mature cordocytic phenotypes with their morphological heterogeneity and multiple functions in the human skin. We used light microscopy, as well as transmission and scanning electron microscopy to study multiple tissue fragments obtained by skin biopsy from post-burn tissue, surrounding superficial temporal artery, and skin, in cases with urticaria pigmentosa, Fabry’s diffuse angiokeratoma, allergic vasculitis, hypodermitis, as well as basocellular nevomatosis and basocellular carcinoma. Our observations proved that the cordocytes appear from precursor/stem cells only outside the pericytes in the case of nascent dermal vessels, or from mesenchymal cells of the connective tissue surrounding superficial temporal artery within their niches, and they are responsible for multiple functions, especially protective for vessels and nerves in the reticular dermis. Their spatial and temporal interactions, either with each other or with another phenotype, undifferentiated or differentiated cells, and their implications in skin diseases, with changing phenotypes, position, and number, merit much more interest in further comprehensive studies. Our results showed that these protective interstitial cells promote useful interactions, influencing differentiation and fate of cells closely surrounded by them, as well as preventing or delaying some pathological processes.
2.Introduction
3.Materials and Methods
4.Results and Discussions
5.References
Key words
Human Skin; Stem Cells; Cordocytes; Skin Diseases; Ultrastructure.
Introduction
For basic science and modern medical care it is important to better understand the continuum of cellular events from genesis to death, and from precursor/ stem cells to well-differentiated cells with their changing phenotypes, multiple interactions, and functions. A promissing cell for both fundamental knowledge and clinical applications – as a good candidate for transplantation in regenerative medicine – is a special interstitial cell described in the last few years with the terms: “Cajal-like cell,” “telocyte,” or “cordocyte” in diverse extra-digestive organs. Still, little information is reported in literature about this controversial cell which proves close relationships with stem cells in many situations, and research work is now very intensive in this field, including the human skin and brain [1-9].
Materials and Methods
We used multiple tissue fragments obtained by skin biopsy from post-burn skin, superficial temporal artery, and in cases with urticaria pigmentosa, Fabry’s diffuse angiokeratoma, allergic vasculitis, hypodermitis, basocellular nevomatosis and basal cell carcinoma, to be examined by light microscopy (LM), transmission electron microscopy (TEM), and scanning electron microscopy (SEM). The samples observed under a light microscope were fixed with 2.5% buffered glutaraldehyde, postfixed with 1% buffered osmium tetroxide, dehydrated through graded alcohols and embedded in an epoxy resin (Epon 812). Sections with a thickness of 4-6 μm were cut using an ultramicrotome, mounted on glass slides, stained with 1% toluidine blue and examined by conventional light microscopy. Multiple ultrathin sections, 70 nm thick, were also cut with an ultramicrotome and contrasted with 2% uranyl acetate solution, as well as Reynolds lead citrate solution. The specimens were then observed under a Philips EM 300 transmission electron microscope (Philips, The Netherlands). Using a scanning electron microscope (JEOL, Japan) a segment from a superficial temporal artery was examined.
Results and Discussions
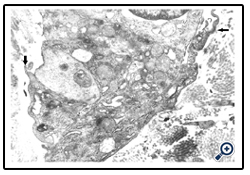
For spatial and temporal events which imply the participation of precursor/stem cells to cordocytic lineage in skin we observed stadial modifications, both in the perivascular zones of superficial temporal artery and in the reticular dermis post-burns (after 20 days following the critical event). By LM, in the adipose tissue around the superficial temporal artery we found large lipid droplets, coexisting with nascent adipocytes in the mass of precursor/stem cells, some in division, and long cordocytic phenotypes, i.e., well-differentiated cordocytes. All these cellular elements were embedded into an abundant collagenous extracellular matrix. Significantly, well-differentiated cordocytes were in contact with both undifferentiated cells and large adipous cells (Fig.1). In the vicinity of the vascular wall of superficial temporal artery, additionally to the adipocytes of different sizes, myelinated axons and small vessels were also seen, all of them surrounded by cordocytes. These interstitial cells, variable in length, were accompanied by an abundant collagenous extracellular matrix produced by themselves, like a thick band along their very thin and long processes. Thus, the collagen and other matriceal molecules appear to be produced by these cells, both protecting themselves and neighboring vascular and nervous structures (Fig.2). An abundant collagenous extracellular matrix was observed in the vascular wall of bearing small vessels in the dermis after burns. In this case of early vascular morphogenesis coexisting progenitor endothelial cells were seen occupying a central position, surrounded by hypertrophied pericytes with extensive rough endoplasmic reticulum (RER), and outside them, large fibroblasts also with dilated RER. In this stage of vessel formation it is remarkable the presence of rare smaller cells around the new vessel, embedded into the dense collagenous matrix. Occasionally, these outer cells establish direct contact with pericytes and they become cordocytes by differentiation, always positioned around the vessels, outside the pericytes. All these differentiating cells in the early vascular wall, before the lumen formation, are characterized by important modifications in cytoplasm organization and chromatin structure, indicating a specific cell lineage. Once differentiation is started on a specific lineage, the neighbor cells become different in their cytoplasmic specialization and their nuclei undergo more subtle modifications in chromatin geometry, remaining a precious indicator of the future cellular type (Fig.3). In mature, small vessels the pericytes may show a synthetic phenotype, with extensive RER in cytoplasm, or a contractile phenotype, with many filaments in cytoplasm, such as smooth muscle cells from arterioles or large vessels, arteries or veins. At the same time, fully-differentiated cordocytes, possessing characterisically long prolongations, can encircle the vascular wall, sometimes with overlapping processes. In some instances, cordocytes can be interconnected by peg and socket contacts, either themselves, or with pericytes, suggesting a role in vessel stabilization (Fig.4).
Figure 1: Adipose tissue around a superficial temporal artery showing many lipid droplets surrounded by adipocytes and precursor/stem cells, some of them in division (arrow). It is remarkable that interstitial cells (cordocytes) come in contact with both precursor/stem cells and adipocytes (arrowheads). Toluidine blue staining. x400
Figure 2: Privascular zone around a superficial temporal artery showing connective tissue, a nerve with myelinated axons, small vessels and adipocytes. It is remarkable that all these structures are intimately surrounded by long processes of cordocytes (arrowheads). Toluidine blue staining. x400
Figure 3: A nascent vessel in the post-burn dermis. One can see four cellular types in this image, namely, endothelial cells, pericytes, fibroblasts, and cordocytes, as well as numerous thin collagen fibrils. Note a contact area between cordocyte and pericyte (arrow). x20,000
Figure 4: A cordocyte interconnected with a pericyte (arrow) surrounding a vessel with hypertrophied pericytes and endothelial cells in mitosis, and visible chromosomes. x10,000
However, they are permissive for diapedesis of vascular cells from the lumen into perivascular space. In addition to their close relations with the vascular wall, these interstitial cells constantly show spatial relations with nerve endings in the dermis, more cordocytes monitoring even an unmyelinating axon with its Schwann cell. Characteristic thin and long cytoplasmic processes of the cordocytes can surround either a single Schwann cell, or a large area containing numerous unmyelinated axons embedded in their extracellular matrix. Moreover, an increased number of these interstitial cells around the dermal nerves were visualized in a case with histiocytic sarcoma, but no other proliferative cells which seem to be impeded to reach nerve endings Fig.5). Other significant relationships are established between resident dermal cells and well-differentiated cordocytes when the latter cells intimately surround more cell types by eans of their long and thin cytoplasmic processes facilitating different paracrine actions (Fig.6). In this respect, it is also significant the relation between cordocyte and fibroblast, hich can be seen in close contact until the fibroblast maturation occurs. Additionaly, interconnected and overlapped cordocytic prolongations surrounded large cell groups including lymphocytes and macrophages, themselves in close contact with each other (Fig.7). When mast cells are degranulated, some cordocytes can be closely seen (Fig.8). Interestingly, in these situations shed microvesicles can be visualized in the extracellular space, close to the cordocytic membrane. Perivascular resident cordocytes seem to be activated before mast cell degranulation, and large cordocytic processes separate vascular wall and mast cells under degranulation, as it is evidenced by ectoplasmic granules accumulation. Mast cells loaded with characteristic cytoplasmic granules and cordocytes in close contact can be seen around the vascular wall, either arteriolar or venular, in the case with urticaria pigmentosa. An accumulation of cellular elements including mast cells, macrophages, cordocytes, and pericytes were visualized around the vessels in a case with Fabry’s disease, when characteristic dense bodies in the cytoplasm of the endothelial cells and pericytes are seen, together with cordocytes, but not within Schwann cells (Fig.9). In some benign and malignant proliferations of the skin, such as basocellular nevomatosis, and basal cell carcinoma, reactive cordocytes could be visualized, like a barrier, around the proliferative cells, either isolated cells , or densely-packed cells (Fig.10). Interestingly, in a case with basal cell carcinoma some tumoral cells showed gradually proapoptotic features, i.e., indented nuclei with chromatin margination and condensation, clumps of chromatin of different size, and cytoplasm densification with separation from the other viable neighboring tumor cells.
Figure 5: Long and thin processes of the cordocytes which separate groups of unmyelinated axons in the dermis (arrows) and other overlapped cordocytic processes outside the nerve endings (arrowheads). x10,000
Figure 6: Cordocytic processes intimately surrounding a group of densely-packed cells in the reticular dermis (arrows).x10,000
Figure 7: A group of immune cells in contact with each other and surrounded by interconnecting cordocytic processes with shed microvesicles close to the cell membrane (arrows). x20,000
Figure 8: Cordocytic long prolongation separating the vascular wall from a degranulating mast cell in a case with urticaria pigmentosa (arrow). Note the presence of shed microvesicles in the extracellular space, close to the cordocytic membrane. x16,000
Figure 9: Numerous dense bodies in the long cordocytic processes encircling unmyelinated axons and a Schwann cell. x20,000
Figure 10: Long cellular processes probably belonging to the cordocytic phenotype applied on the proliferated cells in a case with basocellular nevomatosis (arrow). x8,000
Beneath these dying cells many stratified cell prolongations belonging probably to the fibroblasts, rather than to the cordocytic elements were observed (Fig.11). However, the cordocytes themselves may undergo phenotypic changes, in both nuclear and cytoplasmic organization (Fig.12), or they clearly show the mortal phenotype (as general rule, apoptotic/necrotic one), when nucleus and cytoplasm become hyperdense and compact in aspect (Fig.13). This continuum of events, from mesenchymal stem cells to mature and functional phenotype, and finally, to death, programmed or accidentally, lead to the normal homeostasis and/or repairing processes of the human skin. Therefore, on the vascular walls, for example at the superficial temporal artery surface, a dense cordocytic network can be visualized by SEM (Fig.14).
Figure 11: Multiple thin and long cellular processes of the multilayered fibrocytic phenotype on the proliferated cells in a case with basal cell carcinoma. Note two apoptotic tumor cells displaying incisures of nuclear envelope, clumps of chromatin, and cytoplasmic condensation. x8,000
Figure 12: A cordocyte showing proapoptotic changes of nucleus with envelope incisures and clumps of chromatin, and a dense cytoplasm. x25,000
Figure 13: Typical apoptotic cell of cordocytic phenotype showing nucleus shrinkage and cytoplasmic densification, and nearby apoptotic bodies (arrow). x12,000
Figure 14: Long and sinuous multilayered cordocytic processes, visualized by SEM at the vascular surface of a superficial temporal artery and intermingled with adherent erythrocytes. Stratification of these cells around the large arteries is an important cell mechanism for vessel protection as physical barriers. x16,000
In this work we analyzed by light and electron microscopy some morphological and ultrastructural aspectsconcerning the special interstitial cells named by us “cordocytes,” from their origin as precursor/stem cells until death, in a variety of normal and clinical conditions in the human skin. We used the term “cordocyte” to define a mesenchymal cell for its main morphofunctional characteristics, like a cord and encircling the target protected cells or tissue, as general rule, nervous or vascular tissue, acting locally or at any distance. Similar cells in the skin were described using the term “Cajal- like cells” [10] or “telocytes” [11]. These cells have been localized in close proximity of connecting to fibroblasts, mast cells, adipocytes, and connective tissue fibers [12]. Also, telocytes were identified between the bulge areas of hair follicles and the adjoining nerve fibres in reticular dermis. Their presence and actions either by direct contact or indirect, via shed microvesicles, increase the efficiency of resident local stem cells in process of repair/regeneration. So far, telocytes are involved in skin homeostasis, remodeling, regeneration, and repair [13]. Telocytes were analyzed in normal and scleroderma skin [14], and their progressive reduction and loss was evidenced in dermal cellular network of systemic sclerosis [15].
In present, the ways for an unifying theory on special interstitial cells from the human body according to the similarities in main characteristics are open despite the local differences in aspect and functions due to specific organ requirements and microenvironment. Their protective roles, especially around the vascular wall or nerves is firstly, a physical one, like a barrier, as the cellular membrane acts as a barrier to keep harmful materials outside the cell. Secondly, these cells such as stromal cells in the dermis (like the fibroblasts, immune cells, pericytes, and inflammatory cells) may release growth factors that promote cell divisions, keeping the epidermis regenerating from the bottom. Certain types of skin cancers (basal cell carcinomas) cannot spread throughout the body because the cancer cells require nearby stromal cells to continue their divisions. Thus, from the physical barriers to growth factors is a long route, and therefore, multiple phenotypes and molecules must be identified and characterized in their complex interactions, from stem cells to specific well-differentiated cells. Additionally, the relationships between stem cells and cordocytes have to be better documented in future studies.
Here, we have illustrated the presence of both precursor/stem cells in vascular wall morphogenesis in postburn dermis, to the cordocytic lineage, and different cordocytic phenotypes and their few morphological roles in the skin. At the same time, it is important to underline the importance of the extracellular matrix in the early vascular wall. It is known that stem cells are typically located in the microenvironment formed by the specific extracellular matrix, including complex extracellular stress signal and chemical signals, guide stem cell migration, self-renewal and differentiation. Cordocytic differentiation always takes place at the vascular periphery, outside the pericytes and then some well-differentiated cordocytes may migrate in the reticular dermis, only a few cells remaining around the newly-formed vessels, with protective roles. The maintenance of their networks is made by proliferation of preexistent cells or by proliferation and differentiation from precursor/stem cells. Now, it is clear the origin of these cells from mesenchymal precursor cells with differentiation toward Cajal-like phenotype. However, the factors which influence the changing phenotypes and their number in any tissue remain to be determined. It is known that bacteria can affect the chromatin structure and transcriptional program of host cells by influencing diverse epigenetic factors (i.e., histone modification, DNA methylation, chromatinassociated complexes, noncoding RNAs, and RNA splicing factors). The number of cordocytes surrounding dermal nerves was increased around the vascular wall in a case with urticaria pigmentosa, when mast cell degranulation has occured. Finally, the mortal phenotype for these interstitial cells in the skin was identified as necroapoptotic one, in a case with hypodermitis. However, we need to know better this field of life, i.e., death, because chromatin structure, transcriptional factors, transcripts, and other small molecules must be still identified in future efforts. It is known that small molecular changes can induce significant phenotypical changes. So far, there is little knowledge of ultrastructural patterns of cell death in a large variety of cell types, in health and disease [16,17].
In conclusion, this study demonstrates that the cordocytes appear from precursor/stem cells only outside the pericytes in the case of nascent dermal vessels, or from mesenchymal cells of the connective tissue surrounding superficial temporal artery within their niches, and they are responsible for multiple functions, especially protective for vessels and nerves in the reticular dermis. Finally, a continuum of cellular events, from mesenchymal stem cells to mature cordocytic phenotypes with their morphological heterogeneity and multiple functions must be demonstrated ultrastructurally in the human skin and other organs.
References
- Pais V (1983) Ultrastructura pielii umane. Editura Medicala, Bucuresti.
- Pais V (2002) Atlas de patologie ultrastructurala cutanata. Editura Ager, Bucuresti.
- Kapinas K, Grandy R, Ghule P, Medina R, Becker K, et al. (2013) The abbreviated pluripotent cell cycle. Journal of Cellular Physiology 228: 9-20.
- Pais V, Danaila L, Pais E (2012) From pluripotent stem cells to multifunctional cordocytic phenotypes in the human brain: an ultrastructural study. Ultrastructural Pathology 36: 252-259.
- Pais V, Danaila L, Pais E (2013) Ultrastructural characterization of a developing pericytic microtumor in the white matter postlaceration. International Journal of Stem Cell Research and Transplantation 1: 1-7.
- Pais V, Danaila L (2010) The interstitial cells of the human brain. An atlas of light and electron microscopy. (3rdedn). Ars Academica, Bucharest.
- Simara P, Motl JA, Kaufman DS (2013) Pluripotent stem cells and gene therapy. Translational Research 161: 284-92.
- Klein S, Seidler B, Kettenberger A, Sibaev A, Rohn M, et al. (2013) Interstitial cells of Cajal integrate excitatory and inhibitory neurotransmission with intestinal slow-wave activity. Nature Communications 4: 1630.
- Goldenberg RC, de Carvalho AC (2013) Resident stem cells and regenerative therapy. (1stedn). Academic Press, Massachusetts.
- Min KW, Leabu M (2006) Interstitial cells of Cajal (ICC) and gastrointestinal stromal tumor (GIST): facts, speculations, and myths. Journal of Cellular and Molecular Medicine 10: 995-1013.
- Zheng Y, Bai C, Wang X (2012) Telocyte morphologies and potential roles in diseases. Journal of Cellular Physiology 227: 2311-7.
- Rusu MC, Mirancea N, Manoiu VS, Valcu M, Nicolescu MI, et al. (2012) Skin telocytes. Annals of Anatomy 194: 359-67.
- Ceafalan L, Gherghiceanu M, Popescu LM, Simionescu O (2012) Telocytes in human skin--are they involved in skin regeneration? Journal of Cellular and Molecular Medicine 16: 1405-20.
- Manetti M, Ruffo M, Guiducci S, Faussone-Pellegrini MS, Matucci- Cerinic M, et al. (2012) Telocytes in normal and scleroderma skin. Italian Journal of Anatomy and Embryology 117: 112.
- Manetti M, Guiducci S, Ruffo M, Rosa I, Faussone-Pellegrini MS, et al. (2013) Evidence for progressive reduction and loss of telocytes in the dermal cellular network of systemic sclerosis. Journal of Cellular andlecular Medicine 17: 482-96.
- Pais V (2005) Moartea programata a celulelor umane in imagini ultrastructurale. Cartea Universitara, Bucuresti.
- Pais V, Danaila L, Pais E (2013) Ultrastructural patterns of the activated cell death programs in the human brain. Ultrastructural Pathology 37: 110-20.