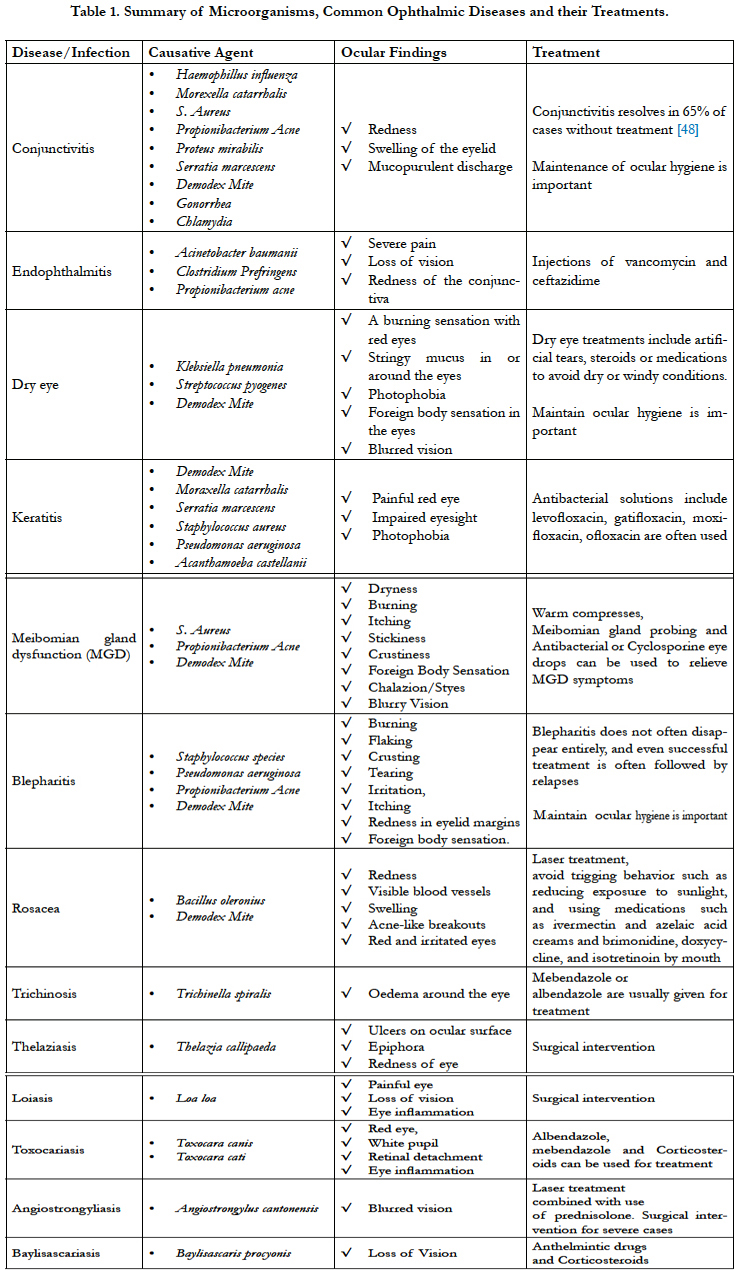
Microorganisms and Common Ophthalmic Diseases
Su CW1*, Tighe S2
1 TissueTech, Inc., Miami, FL, USA.
2 Florida International University, Herbert Wertheim College of Medicine, Miami, FL, USA.
*Corresponding Author
Chen-Wei Su MS,
TissueTech. Inc., 7000 SW 97th Avenue,
Suite 212, Miami, FL 33173, USA.
Tel: (786) 456-7870
E-mail: csu@tissuetechinc.com
Received: December 22, 2016; Accepted: December 30, 2016; Published: January 02, 2017
Citation: Su CW, Tighe S (2017) Microorganisms and Common Ophthalmic Diseases. Int J Ophthalmol Eye Res. 5(1), 272-276. doi: dx.doi.org/10.19070/2332-290X-1600058
Copyright: Su CW© 2017. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
The human eye is in constant contact to environmental conditions that increase its likelihood in being exposed to a multitude of pathogens. Studies have revealed that the normal microbial flora found around the ocular area contribute to the well-being of ocular health as they play a critical role in inhibiting the proliferation of other pathogenic species and maintaining the homeostatic balance. However an imbalance in this status quo may lead to diseases including dry eye syndrome, meibomian gland dysfunction (MGD), blepharitis, rosacea, allergies, keratitis, conjunctivitis and immunological conditions such as mucous membrane pemphigoid and Sjogren’s syndrome. Thus, it is important to investigate and understand the role of various microorganisms that lead to these ocular diseases, and this knowledge may lead to better future treatments. This review focuses on recent advances and insights of pathogens and related ocular diseases which can help the audience further understand the etiology and the control of these diseases.
2.Introduction
3.Bacterial Homeostatic Balance and Eye Infection
4.Fungal Infections of the Eye
5.Parasitic Eye Infections
5.1. Acanthoemba Infection
5.2. Round Worms
5.3. Demodex Mites
6.Conclusion
7.References
Keywords
Microorganisms; Ocular Diseases; Acanthoemba; Bacteria; Demodex Mite; Fungi; Infection.
Introduction
Although the surface of the eye has the mechanical and immunologic functions to normally defend itself against pathogenic microorganisms that cause ocular diseases such as dry eye syndrome, meibomian gland dysfunction, and blepharitis [1], the eye is continuously exposed to the external environment and is therefore highly susceptible to a multitude of pathogens. In addition to the mechanical functions, previous studies have demonstrated the homeostatic balance of normal microbial flora found around the eye is important to maintain ocular health as these microorganisms are capable of preventing the colonization or the abnormal proliferation of pathogenic microorganisms [2]. In fact, it has been found that the number of microbial cells far exceeds the number of human cells in the body thus reaffirming their significance in the overall microbiome [3]. Hence, any manipulations or actions such as wearing unclean contact lenses or rubbing the eye may disturb the homeostatic balance and cause ocular diseases. Many studies have tried to examine the microbes from different parts of the ocular surface including a recent study [4] that reported the profile of microorganisms from 235 patients with various ophthalmic infections including dacrocystis, corneal ulcers, endophthalmitis and post-traumatic infections and found 34.5% of cases (81 patients) yielded bacterial growth, 13.6% (32 patients) yielded fungi growth and the presence of Acanthamoeba. Thus, in this review, we emphasize on the clinically important species of bacteria, fungi, and parasitic microorganisms with their associated ocular diseases to provide readers an integral summary to further understand the etiology and the control of these diseases.
Bacterial Homeostatic Balance and Eye Infection
Traditionally, to identify and characterize bacteria of the ocular area has resorted to swabbing the skin, eyelid or ocular surface and analyzing these bacterial strains under microscopy. Recently, DNA sequencing-based technology of bacterial population has been reported and conducted wildly to reveal a diversity of bacteria from healthy individuals. This thorough sequencing of conjunctival DNA was conducted by researchers in University of Northern Texas in 2011 [5] and they found the combined bacterial community was able to be classified into 5 phyla and 59 distinct genera. The 12 most common genera were Pseudomonas, Propionibacterium, Bradyrhizobium, Corynebacterium, Acinetobacter, Brevundimonas, Staphylococci, Aquabacterium, Sphingomonas, Streptococcus, Streptophyta, and Methylobacterium which composed about 96% of the classified bacterial detected. Despite these bacteria’s normal presence, Staphylococcus, Pseudomonas and Propionibacterium species have been reported the most common bacterial species for causing ocular infections such as blepheratis [6, 7]. These same pathogens may also invade the ocular surface to cause conjunctivitis, keratitis, and even sight-threatening endophthalmitis [8, 9]. In fact, these bacterial pathogens are isolated in approximately 50% of the swabs taken from the conjunctiva and tears, and more than 50% from the eyelids [10]. This above data demonstrate bacterial pathogens commonly found in healthy eyes may also led to infection, thus suggesting the infections are linked to the change of the homeostatic balance and/or abnormal proliferation that can be caused by multiple factors. For example, the pathogenesis of ocular infections can be caused by neutropenia, surgical intervention, improper hygiene, the use of broad spectrum antiobiotics or even wearing contaminated contact lenses. In fact, contact lens wearers were found to have a significantly higher number of bacterial species identified on their lid margin than control healthy subjects (non-contact lens wearers) and these same subjects were more likely to develop ocular surface diseases [11]. Hence, it is important to keep the homeostatic balance of local bacterial flora, avoid improper use of makeup and maintain the cleanliness of the ocular area to prevent the onset or recurrence of these ocular diseases.
Fungal Infections of the Eye
Fungal eye infections are commonly caused by environmental factors [12], post-surgical complications or ocular injuries [13]. A previous report [14] has shown the most common fungi species involved in eye infections are Fusarium (67% of cases), Aspergillus (10.5% of cases), and Candida species (10% of cases). Notably, approximately 40% of these infections were related to trauma [14]. These fungi can invade and affect different parts of the eye thus leading to conditions such as keratitis or endophthalmitis. There are two types of endophthalmitis: exogenous and endogenous. Exogenous fungal endophthalmitis occurs after fungal spores enter the eye from an external source and may be caused by surgical procedures that disrupt the integrity of the eye such as cataract, keratoplasty, glaucoma, or retinal, and intravitreal injections. Symptoms normally present 1 week after the procedure. Endogenous endophthalmitis is a much less prevalent occurring in less than 15% of endophthalmitis cases and occurs when a bloodstream infection (for example, candidemia) spreads to one or both eyes. The most common cause of endogenous endophthalmitis is Candida albicans [15]. These conditions may lead to partial or total loss of vision and may resort to surgical correction to resolve ocular pain [1]. As fungi eye infections can be very severe and difficult to treat, antifungal drugs are often used. Prevention of endophthalmitis is normally performed by administering pre-, peri- and post-surgical antibiotic regimens, however their effectiveness in prevention has not been proven in a well-controlled prospective study.
Antibiotics are also used for the early treatment of bacterial keratitis to prevent superficial keratitis (only involving the uppermost layers of the cornea) from spreading deep in the stroma that may cause subsequent scarring. Due to the increased prevalence of keratitis in contact lens wearers, antibiotics can also be used to the soak contact lenses to prevent contamination from organisms such as Fusarium and Candida albicans which are the most common fungi found on contact lens or in contact lens care solution [16].
Parasitic Eye Infections
Parasitic infections are major causes of ocular diseases all over the world especially in third world countries where poor sanitary conditions favor the parasitism between man and animals [17]. Parasitic infection can be caused by a range of organisms including single-celled Protozoan to large, multi-cellular organisms such as Helminth or Ectoparasites [18]. Herein, we list the some of the most common parasites causing ocular infection:
Acanthoemba strains are one of the most common protozoa in soil, and are also frequently found in contaminated water and other habitats [19]. When present in the eye, acanthamoeba strains can cause keratitis [20] which may subsequently lead to corneal ulcers or even blindness [21, 22]. Acanthamoeba keratitis is usually linked with the use of contact lens in advanced countries [23] but is more commonly induced by exposure to contaminated water in countries like India [24]. A recent study conducted by researchers in Austria demonstrated that 89% of acanthamoeba keratitis patients were contact lens wearers and 19% of the patients required a corneal transplant [25]. The major symptoms of acanthamoebic keratitis are painful radial neuritis, eyelid ptosis, conjunctival hyperemia, epithelial ulcers, and the symptoms are usually linked with a formation of a central ring-shaped corneal infiltrate. Acanthamoebic keratitis represents one of the most difficult ocular conditions to treat since there are no known anti-microbial agents that can exert a reproducible, effective killing effect. Several agents such as polyhexamethylene biguanide (PHMB) [26], propamidine isethionate [27] or chlorhexidine [28] have been used to treat acanthamoebic keratitis in some cases, yet they are not always effective and this leaves the corneal surface susceptible to severe corneal scarring [29]. Proper prevention should be followed by maintaining ocular hygiene and properly maintaining the cleanliness of contact lenses by avoiding the use of tap water to wash or store contact lenses.
There are several types of round worms that may cause ocular diseases such as Anagiostrongyliasis, Baylisascariasis, Dirofilariasis, Loiasis Thelaziasis, Toxocariasis or Trichinosis [17]. The main causative agents are Angiostrongylus [30, 31], Baylisascaris procynois [32, 33], Dirofilaria repens [34], Loa loa [35], Thelazia callipaeda [36, 37], Toxocara canis [38], and Trichinella spiralis. Although these worms are usually observed in certain areas geographically, travel and co-habitation with pets have facilitated the spreading of certain parasitic diseases. Hence, it is important to ask patients if they have pets or traveled recently when they present with symptoms. These diseases may lead to blurred vision, pain, conjunctival congestion, conjunctivitis, keratitis, glaucoma, oedema around the eye, exophthalmoses or even loss of vision. For treatment of round worm infection, it may require surgical intervention to manually remove the worms with the combination of topical steroids or even laser treatment.
Demodex is a genus of tiny parasitic mites that live in or near animal’s hair or eyelashes. Two distinct species of demodex mites that have been found and characterized in humans are demodex folliculorum and demodex brevis [39, 40]. The life cycle of the demodex mites is approximately 14–18 days from the egg to the larval stage followed by the adult stage [39]. The infestation of demodex mites in the ocular and facial areas has been demonstrated to be the cause of rosacea [41, 42], chalazia, meibomian, gland dysfunction, dry eye, keratitis and blepharitis [43-45]. In fact, a study indicates that demodex mite is found in 42.1% of patients with blepharitis [46]. Moreover, infestation of demodex mites is usually linked with mite-harboring cylindrical dandruff in eyelashes [47]. In order to treat such complications, it is important to reduce the amount of adult mites and their eggs, prevent further mating, and avoiding re-infestation. In addition, it is important to avoid dog or cat contact and maintain hygiene of facial and ocular areas to avoid infestation of demodex mites.
Conclusion
Microorganisms can be naturally found on the ocular surface and contribute to the eye’s overall well-being, however the improper number and balance of species can lead to infections due to daily manipulations or actions. The pathogenesis of ocular infections can be multi-factorial including the use of broad spectrum antiobiotics that disrupt the normal balance of microbial flora, neutropenia, contact lens wear, surgical intervention, ocular trauma or even improper hygiene for ocular area. In addition, although it may be different geographically, microorganisms such as parasites can also invade after contact with contaminated water or soil in certain area and cause different type of ocular infections. Thus it is important to know the common, opportunistic pathogens and their etiology to better prevent and treat ocular diseases. This review captures this information and aims to provide the audience with further understandings of microorganisms and their associated ocular disease so as to establish stepping stones for facilitating further development of therapeutic modalities for ocular diseases.
References
- Klotz SA, Penn CC, Negvesky GJ, Butrus SI (2000) Fungal and parasitic infections of the eye. Clin Microbiol Rev. 13(4): 662-85.
- Lu LJ , Liu J (2016) Human Microbiota and Ophthalmic Disease. Yale J Biol Med. 89(3): 325-330.
- Turnbaugh PJ, Ley RE, Hamady M, Fraser-Liggett CM, Knight R, et al., (2007) The human microbiome project. Nature. 449: 804-10.
- Hemavathi, Sarmah P, Shenoy P (2014) Profile of microbial isolates in ophthalmic infections 234 and antibiotic susceptibility of the bacterial isolates: a study in an eye care hospital, bangalore. J Clin Diagn Res. 8(1): 23-5.
- Dong Q, Brulc JM, Iovieno A, Bates B, Miller D, et al., (2011) Diversity of bacteria at healthy human conjunctiva. Invest ophthalmol vis sci. 52(8): 5408-13.
- Spyridon S Marinopoulos (2014) Blepharitis. Johns Hopkins Antibiotic (ABX) Guides.
- Dougherty JM , Mc Culley JP (1984) Comparative bacteriology of chronic blepharitis. Br J Ophthalmol. 68(8): 524-8.
- Jackson WB (2008) Blepharitis: current strategies for diagnosis and management. Can J Ophthalmol. 43(2): 170-9.
- John T, Shah AA (2008) Use of azithromycin ophthalmic solution in the treatment of chronic mixed anterior blepharitis. Ann Ophthalmol (Skokie). 40(2): 68-74.
- Willcox MD (2013) Characterization of the normal microbiota of the ocular surface. Exp Eye Res. 117: 99-105.
- Larkin DF , Leeming JP (1991) Quantitative alterations of the commensal eye bacteria in contact lens wear. Eye (Lond). 5 ( Pt 1): 70-4.
- Srinivasan M (2004) Fungal keratitis. Curr Opin Ophthalmol. 15(4): 321-327.
- Keay L, Gower EW, Cassard SD, Tielsch JM, Schein OD (2012) Postcataract surgery endophthalmitis in the United States: analysis of the complete 2003 to 2004 Medicare 253 database of cataract surgeries. Ophthalmology. 119(5): 914-22.
- Ibrahim MM, Vanini R, Ibrahim FM, Fioriti LS, Furlan EM, et al., (2009) Epidemiologic aspects and clinical outcome of fungal keratitis in southeastern Brazil. Eur J Ophthalmol. 19(3): 355-61.
- Wilhelmus KR (1987) The pathogenesis of endophthalmitis. Int ophthalmol clin. 27(2): 74-81.
- Sugarman JR, Dennis LK , White E (1994) Cancer survival among American Indians in western Washington State (United States). Cancer Causes Control. 5(5): 440-8.
- Nimir AR, Saliem A, Ibrahim IA (2012) Ophthalmic parasitosis: a review article. Interdiscip Perspect Infect Dis. 2012: 587402.
- Rath S, Basu S, et al., (2016) Ocular Parasitoses: A Comprehensive Review. Surv Ophthalmol. S0039-6257(16): 30013-3.
- Marciano-Cabral F, Cabral G (2003) Acanthamoeba spp. as agents of disease in humans. Clin Microbiol Rev. 16(2): 273-307.
- Nunes TE, Brazil NT, Fuentefria AM , Rott MB (2016) Acanthamoeba and Fusarium interactions: A possible problem in keratitis. Acta Trop. 157: 102- 7.
- Lorenzo-Morales J, Khan NA, Walochnik J (2015) An update on Acanthamoeba keratitis: diagnosis, pathogenesis and treatment. Parasite. 22: 10.
- Auran JD, Starr MB, Jakobiec FA (1987) Acanthamoeba keratitis. A review of the literature. Cornea. 6(1): 2-26.
- Cheung N, Nagra P, Hammersmith K (2016) Emerging trends in contact lens-related 273 infections. Curr Opin Ophthalmol. 27(4): 327-32.
- Sharma S, Pasricha G, Das D, Aggarwal RK (2004) Acanthamoeba keratitis in non-contact lens wearers in India: DNA typing-based validation and a simple detection assay. Arch Ophthalmol. 122(10): 1430-4.
- Walochnik J, Scheikl U, Haller-Schober EM (2015) Twenty years of acanthamoeba diagnostics in Austria. J Eukaryot Microbiol. 62(1): 3-11.
- Sharma S, Garg P, Rao GN (2000) Patient characteristics, diagnosis, and treatment of non-contact lens related Acanthamoeba keratitis. Br J Ophthal-mol. 84(10): 1103-8.
- Lindsay RG, Watters G, Johnson R, Ormonde SE, Snibson GR (2007) Acanthamoeba keratitis and contact lens wear. Clin Exp Optom. 90(5): 351-60.
- Hammersmith KM (2006) Diagnosis and management of Acanthamoeba keratitis. Curr Opin Ophthalmol. 17(4): 327-31.
- Hirano K , Sai S (1999) Severe Acanthamoeba sclerokeratitis in a non-contact lens wearer. Acta Ophthalmol Scand. 77(3): 347-8.
- Prommindaroj K, Leelawongs N, Pradatsundarasar A (1962) Human angiostrongyliasis of the eye in Bangkok. Am J Trop Med Hyg. 119(6): 759-61.
- Sawanyawisuth K, Limpawattana P, Busaracome P, Ninpaitoon B, Intapan PM, et al., (2004) A 1-week course of 290 corticosteroids in the treatment of eosinophilic meningitis. Am J Med. 117(10): 802-3.
- Park SY, Glaser C, Murray WJ, Rowley HA, Bass N, et al., (2000) Raccoon roundworm (Baylisascaris procyonis) encephalitis: case report and field investigation. Pediatrics. 106(4): E56.
- Mets MB, Noble AG, Basti S, Gavin P, Davis AT, et al., (2003) Eye findings of diffuse unilateral subacute neuroretinitis and multiple choroidal infiltrates associated with neural larva migrans due to Bbaylisascaris procyonis. Am J Ophthalmol. 135(6): 888-90.
- Latifoglu O, Ozmen S, Sezer C, Yavuzer R, Altintas K, et al., (2002) Dirofilaria repens presenting as a premasseteric nodule. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 94(2): 217-20.
- Geldelman D, Blumberg R, Sadun A (1984) Ocular Loa Loa with cryoprobe extraction of subconjunctival worm. Ophthalmology. 91(3): 300-3.
- Otranto D, Traversa D (2005) Thelazia eyeworm: an original endo- and ecto-parasitic nematode. Trends Parasitol. 21(1): 1-4.
- Ikeme MM (1967) Kerato-conjunctivitis in cattle in the plateau area of Northern Nigeria. A study of Thelazia rhodesi as a possible aetiological agent. Bull Epizoot Dis Afr. 15(4): 363-7.
- Baquera-Heredia J, Cruz-Reyes A, Flores-Gaxiola A, Lopez-Pulido G, Diaz- Simental E, et al., (2002) Case report: Ocular gnathostomiasis in northwestern Mexico. Am J Trop Med Hyg. 66(5): 572-4.
- Rufli T, Mumcuoglu Y (1981) The hair follicle mites Demodex folliculorum and Demodex brevis: biology and medical importance. A review. Dermatologica. 162(1): 1-11.
- English FP, Nutting WB (1981) Demodicosis of ophthalmic concern. Am J Ophthalmol. 91(3): 362-72.
- Erbagci Z, Ozgoztasi O (1998) The significance of Demodex folliculorum density in rosacea. Int J Dermatol. 37(6): 421-5.
- Forton F, Seys B (1993) Density of Demodex folliculorum in rosacea: a casecontrol study using standardized skin-surface biopsy. Br J Dermatol. 128(6):650-9.
- Coston TO (1967) Demodex folliculorum blepharitis. Trans Am Ophthalmol Soc. 65: 361-92.
- Fulk GW, Clifford C (1990) A case report of demodicosis. J Am Optom Assoc. 61(8): 637-9.
- Heacock CE (1986) Clinical manifestations of demodicosis. J Am Optom Assoc. 57(12): 914-9.
- Dadaci Z, Kilinc F, Ozer TT, Sahin GO, Acir NO, et al., (2015) Periodic acid-Schiff staining demonstrates fungi in chronic anterior blepharitis. Eye (Lond). 29(12): 1522-7.
- Gao Y-Y, Di Pascuale MA, Li W, Liu DT, Raju Vk, et al., (2005) High prevalence of ocular demodex in lashes with cylindrical dandruffs. Invest ophthalmol vis sci. 46(9): 3089-94.
- Rose P (2007) Management strategies for acute infective conjunctivitis in primary care: a systematic review. Expert Opin Pharmacother. 8(12): 1903- 21.