Central Retinal Artery Occlusion during Cisplatin and Etoposide Chemotherapy for Small Cell Lung Cancer
Kitamura Y1, Oshitari T1*, Kato F1, Sakurai M1, Baba T1, Tawada A1, Shirato S1, Takiguchi Y2, Sekine I2, Iwasawa S2, Kurimoto R2, Yoshino I3, Yamamoto S1
1 Department of Ophthalmology and Visual Science, Chiba University Graduate School of Medicine, Chiba, Japan.
2 Department of Medical Oncology, Chiba University Graduate School of Medicine, Chiba, Japan.
3 Department of General Thoracic Surgery, Chiba University Graduate School of Medicine, Chiba, Japan.
*Corresponding Author
Toshiyuki Oshitari, MD, PhD.,
Department of Ophthalmology and Visual Science,
Chiba University Graduate School of Medicine,
Inohana 1-8-1, Chuo-ku, Chiba 260-8670,
Chiba, Japan.
Tel: 81-43-226-2124
Fax: 81-43-224-4162
E-mail: Tarii@aol.com and oshitari@faculty.chiba-u.jp
Article Type: Case Report
Received: March 30, 2015; Accepted: May 28, 2015; Published: May 29, 2015
Citation: Oshitari T et al., (2015) Central Retinal Artery Occlusion during Cisplatin and Etoposide Chemotherapy for Small Cell Lung Cancer. Int J Ophthalmol Eye Res 03(4), 107-109. doi: dx.doi.org/10.19070/2332-290X-1500021.
Copyright: Oshitari T© 2015. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
We present our findings in a case of central retinal artery occlusion (CRAO) that developed in a patient while being treated with intravenous cisplatin (CDDP) and etoposide (VP16) for small cell lung cancer. The patient was a 67-year-old woman who had lung lobectomy for small cell lung cancer. She began adjuvant chemotherapy with CDDP and VP16 after the surgery, and after 13 days of chemotherapy, she developed a sudden painless decrease of vision in her left eye. She was referred to Department of Ophthalmology, and our examination found that her decimal visual acuities were 1.0 OD and light perception OS. Fluorescein angiography showed a CRAO in her left eye. Two months later, she had a sharp pain in the left eye because of neovascular glaucoma, and cyclophotocoagulation was immediately performed. Although her visual function did not recover completely (light perception OS), the pain was alleviated after the cyclophotocoagulation. Physical examinations showed no additional abnormalities of the cardiovascular system. Although a CRAO during chemotherapy is extremely rare, ophthalmologists and internists should remember that a CRAO can develop in patients undergoing combined chemotherapy even though other cardiovascular events may not be present.
2.Introduction
3.Case Report
4.Discussion
5.Conclusion
7.Acknowledgement & Declarations
8.References
Keywords
Central Retinal Artery Occlusion; Cancer; Chemotherapy.
Introduction
A central retinal artery occlusion (CRAO) is considered to be an acute stroke of the eye that results in profound visual reduction. The majority of CRAOs are caused by platelet fibrin thrombi and emboli that develop in individuals with atherosclerotic disease [1-2]. The risk factors for CRAO include arterial hypertension, diabetes mellitus, carotid artery disease, cerebral vascular accidents, and tobacco smoking. Other risk factors that contribute to CRAO are proatherogenic states, hyperhomocystenemia, factor V Leiden, protein C and S and anti-thrombin deficiencies, antiphospholipid antibodies, and sickle cell disease [1]. In addition, patients with cancer who receive chemotherapy are at risk for vascular complications such as veno-occlusive disease, venous thrombosis, and vascular ischemia [3]. Pharmacological agents are also considered to be the independent risk factors for vascular events because some, such as platinum based compounds, vinca alkaloids, bleomycin, and tamoxifen, have been shown to cause vaso-occlusive complications. However, retinal vascular occlusions in patients with cancer who receive chemotherapy is a rare complication and has not been frequently reported.
We describe a case of CRAO that developed in a cancer patient while undergoing intravenous cisplatin(CDDP) and etoposide(VP16) chemotherapy.
Case Report
A 67-year-old woman with stage IIIA small cell lung cancer underwent left lung lobectomy 2 months before first our examination. She began adjuvant chemotherapy with intravenous CDDP and VP16 after the surgery. After 13 days of chemotherapy, she noted a sudden painless loss of vision in the left eye on awakening. She was referred to our Ophthalmology Department.
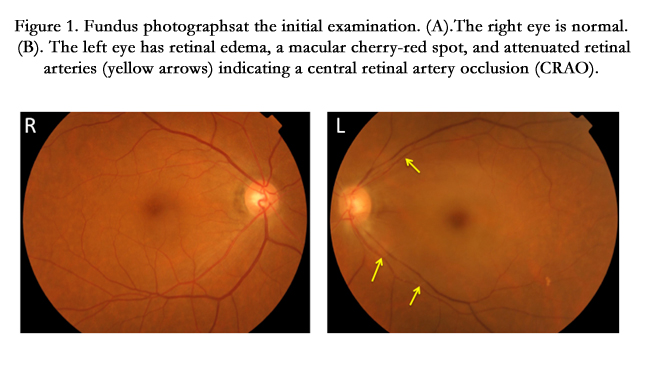
Her medical history showed that she had systemic hypertension and diabetes mellitus which were well-controlled. She had smoked for forty years. On her first examination, her visual acuity was 20/20 in the right eye and no light perception in the left eye. The left eye had a relative afferent pupillary defect. Slit-lamp examination and the intraocular pressure (IOP) were normal. Ophthalmoscopy of the left eye showed cloudy swelling of the nerve fiber layer throughout the posterior pole, a cherry-red spot in the macula, and attenuated retinal arteries (Figure1). The right fundus was normal. A diagnosis of left central retinal artery occlusion was made.
Figure 1. Fundus photographsat the initial examination. (A).The right eye is normal. (B). The left eye has retinal edema, a macular cherry-red spot, and attenuated retinal arteries (yellow arrows) indicating a central retinal artery occlusion (CRAO).
We performed eyeball massage, paracentesis, and oral administration of prostaglandin. However, there was no recovery of visual function. Optical coherence tomographic (OCT) images showed an increase in the reflectivity and thickness of the inner retina and a corresponding decrease of reflectivity in the outer layer of the retina of the left eye (Figure2). Fluoresce in angiography showed markedly delayed filling of the retinal arteries and increased arterio-venous transit time (Figure3). An echocardiogram and carotid artery ultrasound showed no evidence of a thrombus. The electrocardiogram was normal without arterial fibrillation. Coagulation markers such as the platelet count, prothrombin time, and activated partial thrombin time were within normal limits. Diffusion-weighted magnetic resonance imaging of the head and magnetic resonance angiography (MRA) showed no abnormalities.
Figure 2. Optical coherence tomographic (OCT) image. The image of the left eye shows increased reflectivity and thickness of the inner retina. The image of the right eye shows a normal retina.
Figure 3. Fluorescein angiogramstaken 30 hours after vision loss in the left eye. (A). Right eye. The filling time and pattern are normal. (B). Left eye. The filling time of the retinal arteries is markedly delayed.
Two months later, she had a sharp pain in her left eye which we diagnosed to be caused by neovascular glaucoma (NVG). The IOP in her left eye was increased to 58mmHg (Figure4), and cyclophotocoagulation was immediately performed. Although her visual function did not recover completely (light perception OS), the pain was alleviated after the cyclophotocoagulation. After six months, her vision was no light perception.
Figure 4. Slit-lamp examination showed mild corneal edema and iris neovascularization at the pupillary margin.
Discussion
Chemotherapy has been associated with the development of several vascular alterations in patients with cancer. This is probably because chemotherapeutic drugs can damage the vascular endothelium, cause a disequilibrium between procoagulant and anticoagulant molecules, induce tumor/endothelial apoptosis, activate cytokines, and increased tissue factor activity [4]. Although a recent systematic review and meta-analysis indicated that there was no significant increase in arterial thromboembolic events associated with CDDP [5], CDDP has been frequently associated with other adverse vascular events [6-10]. The vascular complications associated with CDDP include Raynaud’s phenomenon, cardiac ischemia, arterial thrombosis, and ischemic cerebrovascular events [7-9]. A previous study reported that arterial thrombosis was detected in 2.6% of patients receiving CDDP chemotherapy [10]. However, a retinal artery occlusion associated with CDDP is extremely rare. To the best of our knowledge, only two cases of retinal artery occlusion following CDDP chemotherapy have been reported [11, 12]. Both cases did not have any known risk factors for atherosclerosis such as smoking history, hypertension, or diabetes mellitus. Because one of the two cases had a recovery of visual function after the discontinuation of CDDP chemotherapy, the authors suspected an involvement of CDDP in the retinal artery occlusion.
In our case, the patient had several vascular risk factors, however there were no clinical signs of systemic atherosclerosis. This suggested that CDDP probably contributed to the vascular events in some way.
The mechanism of how CDDP chemotherapy causes retinal arterial occlusion was not determined. However, several studies have reported that CDDP can induce platelet activation and elevate the von Willebrand factor, which can cause endothelial injury and potentiate arterial thrombosis [13]. Furthermore, hypomagnesaemia and autonomic dysfunction cause vasospasm [8, 11]. A histological examination of the blood vessels after intravenous CDDP showed vascular intimal edema and detachment with pyknosis of the endothelial cells and thrombus formation [8]. Other ophthalmic complications associated with CDDP chemotherapy include pigmentary maculopathy, altered color perception attributable to cone dysfunction, retinal ischemia with neovascularization, optic neuropathy, and cortical blindness [14-18].
The rate of NVG after CRAO is likely around 15%, which we would not count as rare [19, 20]. The CRAO most likely contributed to the NVG because the MRA findings did not show severe stenosis of the internal carotid artery that would cause the ocular ischemic syndrome. In addition, there was no evidence of ischemic retinopathy caused by diabetic retinopathy because the contralateral eye had no non-perfusion areas caused by diabetic retinopathy. We assume that the retina was not completely ischemic and the metabolism of the inner retina was partially maintained. This would then induce angiogenic factors such as vascular endothelial growth factor which could cause the NVG.
Conclusion
In conclusion, the development of a CRAO in a patient undergoing chemotherapy with intravenous CDDP for small cell lung cancer indicates that clinicians should advise patients to immediately consult an ophthalmologist if they have a sudden decrease in their vision.
Acknowledgements and Declarations
We thank Professor Duco Hamasaki for editing the manuscript. We have no conflict of interest regarding this case report.
References
- Chen CS, Lee AW (2008) Management of acute central retinal artery occlusion. Nat Clin Pract Neurol 4:376-383.
- Rudkin A, Lee A, Chen C (2009) Vascular risk factors for central retinal artery occlusion. Eye 24:678-681.
- Cool RM, Herrington JD, Wong L (2002) Recurrent Peripheral arterialthrombosis induced by cisplatin and etoposide. Pharmacotherapy 22:1200-1204.
- Haddad TC, Greeno EW (2006) Chemotherapy-induced thrombosis. Thromb Res 118:555-568.
- Proverbs-Singh T, Chiu SK, Liu Z, Seng S, Sonopavde G, et al. (2012) Arterial thromboembolism in cancer patients treated with cisplatin: a systematic review and meta-analysis. J Natl Cancer Inst 104:1837-1840.
- Cheng E, Berthold DR, Moore MJ, Duran J (2009) Arterial thrombosis after cisplatin-based chemotherapy for metastatic germ cell tumors. Acta Oncol 48:475-477.
- Molloy RG, Welch GC, Drury JK, Abel BJ (1995) Arterial thrombosis after chemotherapy with cisplatin, vinblastine and methotrexate. Br J Clin Pract 49:50-51.
- Içli F, Karaoğuz H, Dinçol D, Demirkazik A, Günel N, et al. (1993) Severe vascular toxicity associated with cisplatin-based chemotherapy. Cancer 72:587-593.
- Longo F, Mansueto G, Poggi A (1999) Acute arterial thrombosis in a patient with small cell lung cancer after a cycle of chemotherapy with cisplatin and etoposide. Tumori 85:214-215.
- Czaykowski PM, Moore MJ, Tannock IF (1998) Highrisk of vascular events in patients with urothelial transitional cell carcinoma treated with cisplatin based chemotherapy. J Urol 160:2021-2024.
- Arijit M (2011) Reversible branch retinal artery occlusion following intravenous cisplatin chemotherapy for cervical carcinoma. Int Ophthalmol 31:429-432.
- Wang MY, Arnold AC, Vinters HV (2000) Bilateral blindness and lumbosacral myelopathy associated with highdose carmustine and cisplatin therapy. Am J Ophthalmol 130:367-368.
- Licciardello JT, Moake JL, Rudy CK, Karp DD, Hong WK (1985) Elevated plasma von Willebrand factor levels and arterial occlusive complications associated with cisplatin-based chemotherapy. Oncology 42: 296-300.
- Kupersmith MJ, Seiple WH, Holopigian K, Noble K, Hiesiger E, et al. (1992) Maculopathy caused by intra-arterially administered cisplatin and intravenously administered carmustine. Am J Ophthalmol 113:435-438.
- Kwan AS, Sahu A, Palexes G (2006) Retinal ischemia with neovascularization in cisplatin related retinal toxicity. Am J Ophthalmol 141:196-197.
- Johnson DW, Cagnoni PJ, Schossau TM, Stemmer SM, Grayeb DE, et al. (1999) Optic disc and retinal microvasculopathy after high-dose chemotherapy and autologous hematopoietic progenitor cell support. Bone Marrow Transplant 24:785-792.
- Wilding G, Caruso R, Lawrence TS, Ostchega Y, Ballintine EJ, et al. (1985) Retinal toxicity after high-dose cisplatin therapy. J Clin Oncol 3:1683-1689.
- Li Y, Li J, Pi G, Tan W (2014) Paclitaxel- and/or cisplatin-induced ocular neurotoxicity: a case report and literature review. Onco Targets Ther 7:1361- 1366.
- Yamamoto K (2005) Neovascular glaucoma after branch retinal artery occlusion. Jpn J Ophthalmol 49:388-390.
- Duker JS (1991) A prospective study of acute central retinal artery obstruction. The incidence of secondary ocular neovascularization. Arch Ophthalmol 109:339-342.