Epidemiology of Eales Disease in the Central Western India
Shah MA*, Shah SM, Kalyani PJ, Shah AH, Shah PD, Pandya JS
Drashti Netralaya, Dahod, Gujarat, India.
*Corresponding Author
Mehul A. Shah MD,
Drashti Netralaya, Nr. GIDC,
Chakalia Road, Dahod-389151,
Gujarat, India.
Tel: 00-91-2673-645364
Fax: 00-91-2673-221232
E-mail: omtrust@rediffmail.com
Article Type: Review Article
Received: October 05, 2014; Accepted: December 30, 2014; Published: January 08, 2015.
Citation: Shah MA, et al., (2015). Epidemiology Of Eales Disease In The Central Western India. Int J Ophthalmol Eye Res. 03(1), 90-94. doi:dx.doi.org/10.19070/2332-290X-1500017
Copyright: Shah MA© 2015. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To investigate the clinical features and propose a new staging system based on the clinicopathological correlation and formulate new guidelines for management of Eales’ disease in healthy young males.
Design: Prospective cohort study.
Setting: Vitreo Retinal Department of a tertiary eye care center in Western Central India.
Participants: Seventy-four eyes diagnosed with Eales’ disease.
Materials and methods: From 2004 to 2010, patients clinically diagnosed with Eales’ disease were enrolled in this study using specific inclusion and exclusion criteria. We examined the patients’ anterior and posterior segments thoroughly. We performed the necessary ocular and systemic investigations. We divided the investigations into three stages: inflammatory, ischemic and complications and the patients were treated accordingly. We treated the patients using medical management, photocoagulation or pars plana vitrectomy. The patients were monitored according to standard schedules and formats. All information was documented using a pre-tested online format and statistical analyses were performed using SPSS ver. 17. A p-value < 0.05 was considered to indicate significance.
Outcome measures: Visual acuity.
Results: The cohort comprised 74 cases with a mean age of 30 ± 8.73 years. The visual acuity of the presenting cohort
was < 3/60 in 64.9% of eyes. The final visual outcome was > 6/12 in 40 eyes (54.1%), 6/60 to 6/18 in 14 eyes (18.9%) and < 1/60 in the remaining 9 eyes at a mean follow-up of 592 days. The visual parameters differed significantly pre-and post-treatment. We evaluated the visual outcome following surgical management.
Conclusion: We studied epidemiological facts about anterior and posterior segment findings.
2.Introduction
3.Methods
4.Results
5.Discussion
6.Conclusion
7.References
Keywords
Eales Diseases; Grading System; Periphlebitis; Vitreous Hemorrhage; Systemic Corticosteroids.
Introduction
Eales’ disease was first described by Henry Eales, a British ophthalmologist, in 1880 and 1882. [1,2] He identified this disease in seven male patients ranging in age from 14 to 29 years with recurrent retinal hemorrhages. In addition, these patients had histories of headache, variation in peripheral circulation, chronic constipation, and epistaxis. [2]
Eales’ believed that these symptoms were caused by vasomotor neurosis with constriction of the alimentary vessels and compensatory dilatation of the retinal and nasal vessels. Eales’ did not describe retinal vasculitis. [2] However, 5 years later, Wadsworth (1887) described signs of retinal vasculitis. [3] Eales’ has subsequently been honored with the eponym for idiopathic retinal Periphlebitis. [4]
Eales’ disease is an idiopathic inflammatory vasoproliferative disorder of the retinal veins, which leads to the occlusion of these vessels. [5] It affects primarily healthy young adults between the ages of 20 and 40 years. It affects primarily males, (80-90%), but can affect females. Elder et al. (1971) reported the possibility of bilateral involvement in 90% of cases. [6] It may rarely affect the arterioles. This vascular occlusion, induced by the inflammation of veins, can lead to proliferative vascular retinopathy and subsequent sequelae, such as recurrent vitreous hemorrhage, and tractional or combined retinal detachment. [5,7]
Eales’ disease was common a century ago, but its incidence has declined in western countries. However, Eales’ disease is widespread in the Indian subcontinent and certain areas of the Middle East. It is usually seen unilaterally, but can present bilaterally. Eales’ disease is characterized by three overlapping stages of venous inflammation (vasculitis), occlusion, and retinal neovascularization. Diagnosis is mostly clinical and requires exclusion of other systemic or ocular conditions that could present with similar retinal features. Recurrent vitreous hemorrhage is the hallmark of Eales’ disease. [7]
Treatment options include systemic steroids during the inflammatory stage, retinal photocoagulation usually in the proliferative stage to the non-perfused retina, and early vitrectomy for recurrent vitreous hemorrhage. [6,7,11] Oral steroids are the primary agents used most frequently for the management of Eales’ disease, but prolonged systemic use of steroids is associated with serious ocular and systemic side effects. [11] Photocoagulation is the mainstay of treatment in the proliferative stage of Eales’ disease. [12] Intravitreal steroids and anti-VEGF have been used in many posterior segment ocular pathologies. [13,14] Visual prognosis is good if treated early in the course of the disease. [7,12,13] Recently, anti-metabolite therapies, especially methotrexate, have been used in Eales’ disease with some success. [12]
The prognosis of patients with Eales’ disease is variable and depends on the availability of medical care. Many of the patients in the region in which this study was conducted live in areas that are underserved by medical care. Therefore, the treatment outcome may also vary. This study assessed the demographics, clinical presentation and treatment outcome of Eales’ disease in Central West India, which is inhabited by mainly tribal populations.
Methods
This study was a prospective interventional case series, designed to evaluate the epidemiology of Eales’ disease patients in a region of Central Western India; data were collected from January 2004 to December 2012. The study was conducted in a tertiary care teaching hospital and a tribal center for ophthalmic care, which serves the second largest tribal belt in India. It is located at the junction of three states, Gujarat, Rajasthan and Madhya Pradesh.
Approval from the Institutional Ethics Committee was obtained and all efforts were made to adhere to the guidelines of the Declaration of Helsinki.
Patients were briefed about the procedures of the study and consent was obtained before enrolment.
Patients suspected to have Eales’ disease aged between 20 and 40 years underwent exhaustive ocular and medical evaluation for confirmation of a diagnosis of Eales’ disease. Our cohort comprised patients diagnosed with Eales’ disease with no other systemic or ocular disease.
Subjects with a history of diabetes mellitus, hypertension, collagen vascular disease, sarcoidosis, Bechet’s disease, systemic lupus erythematosus, Coats’ disease, or syphilis were excluded from the study.
Ophthalmic examinations included best-corrected visual acuity using Snellen’s chart, intraocular pressure measurement by applanation tonometry, and anterior segment evaluation by slit-lamp examination.
Evaluations of the posterior segment at the initial and each subsequent visit were performed by direct and indirect ophthalmoscopy, slit-lamp biomicroscopy with 90-D and three-mirror lenses, and fundus fluorescein angiography.
Investigations included complete blood count with sickle-cell screening, erythrocyte sedimentation count, liver function test, fasting plasma sugar level, blood coagulation profiles, Mantoux test using 2 TU/0.1 mL purified protein derivative (PPD), and chest X-ray.
Ultrasonography was performed to detect fractional retinal detachment in eyes affected by vitreous hemorrhage with no fundus view. The location and extent of retinal involvement due to Eales’ disease, neovascularization, and fibrovascular traction were documented in all patients by fundus drawing and digital fundus color photography. Optical coherence tomography (OCT) was performed using high-density OCT (Cirrus, 2000, Carl Zeiss
Meditec, CA,) as required after it has became available.
The inflammatory stage was treated with medical management, periocular steroids, systemic steroids or methotrexate. Completeblood counts and liver function tests were performed at least once per month in patients prescribed methotrexate. Prior to prescribing methotrexate, a thorough medical consultation was performed by a medical oncologist. The ischemic stage was treated with anti- VEGF or photocoagulation according to availability. The stage of complications was treated with surgical management. The details were documented in a pre-tested, online Eales’ disease form and then exported to a Microsoft Excel™ spreadsheet.
Data were analyzed using the SPSS software (ver. 17.0; IBM SPSS Inc., Chicago, IL, USA). Univariate parametrical analyses were used. A p-value of <0.05 was considered to indicate statistical significance.
Results
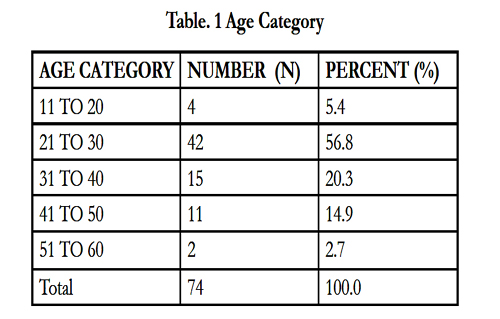
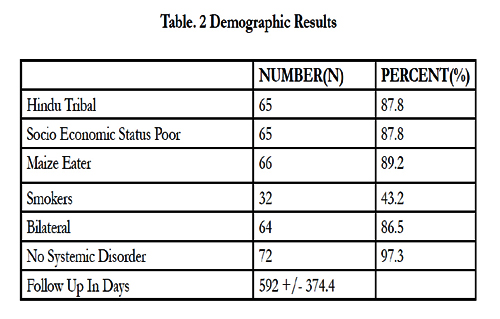
The cohort comprised 74 male patients with a mean ages of 30 ± 8.73 years (Table 1). Demographic factors including age, diet, smoking, religion and race were evaluated (Table 2). No significant differences in the demographic factors were observed, with the exception of age of presentation (p = 0.02).
Within the cohort, the presenting visual acuity was < 3/60 in 64.9% of eyes. The final visual outcome was > 6/12 in 40 eyes (54.1%), 6/60 to 6/18 in 14 eyes (18.9%) and < 1/60 in 9 eyes at a mean follow-up of 592 days.
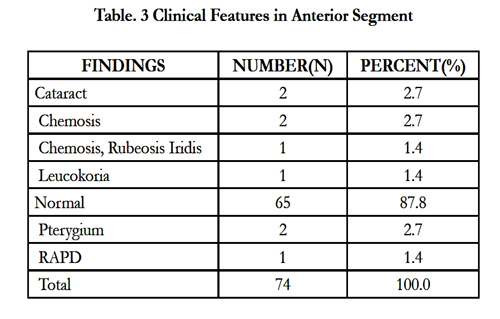
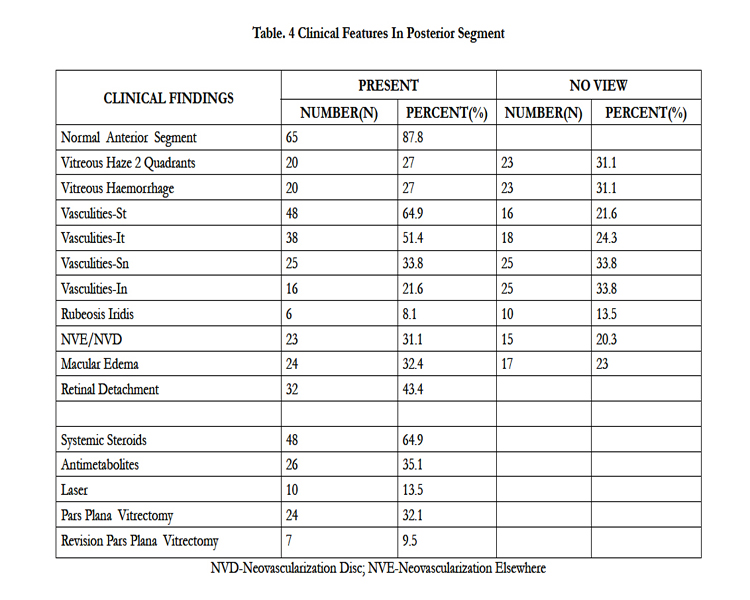
We found the anterior segment normal in 65 (87.8%) cases (Table 3). We then evaluated the findings in the posterior segment (Table 4). The treatments prescribed were systemic steroids (64.9%),anti-metabolites (35.1%), photocoagulation (13%), pars plana vitrectomy (32.1%) and revision vitrectomy (9.5%) (Table 4).
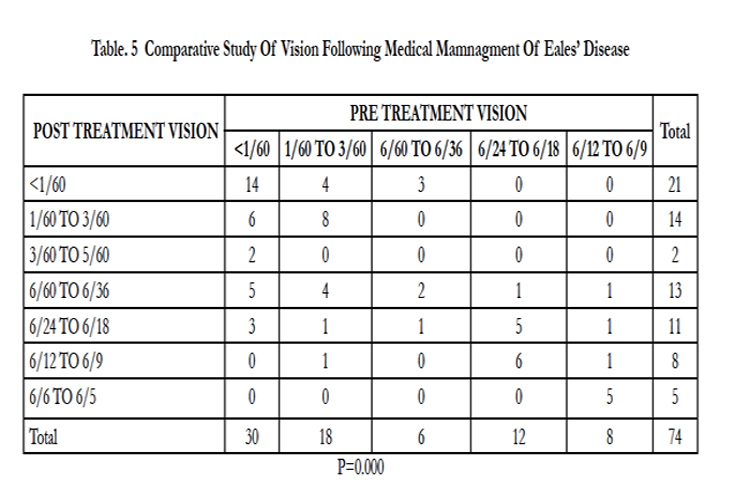
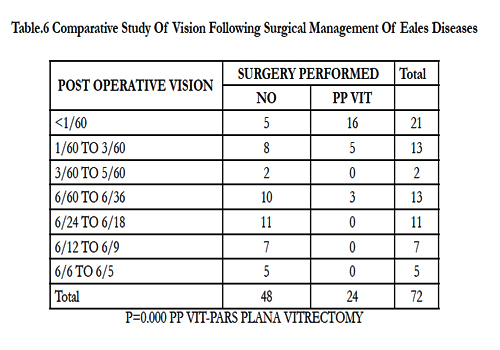
We evaluated visual parameters pre- and post-treatment, and found a significant difference (p = 0.000, Table 5). We next compared visual outcome following surgical management, and again found a significant difference (p = 0.000, Table 6).
Discussion
Our cohort comprised 74 cases with a mean age of 30 ± 8.73 years. Other reports with a similar demographic profile, including age and gender reported similar findings. [7,15-17] In this study, we have simplified the classification according to the clinical findings. In the inflammatory stage (stage 1), patients with vitritis and vasculitis were treated with systemic and periocular steroids. In the ischemic stage (stage 2), patients with neovascularization and ischemia were treated with laser or anti-VEGF therapy. During the stage of complications (stage 3), cases of traction or mixed retinal detachment were treated with pars plana vitrectomy. Saxena et al. reported a classification and grading system, the proposed classification system may provide guidelines for the management of Eales’ diseases. [15] Periphlebitis and vitreous hemorrhage have been reported, but the incidences of vitritis, vasculitis and vitreous hemorrhage have not been described. [7,15-17]
We observed total posterior vitreous detachment in 24.3% of cases. Badrinath et al. reported multifocal vitreo retinal adhesions in 87.8% of cases. [18,19] There was no difference in the response to treatment in the Mantoux-positive and -negative groups. [20]
Oral corticosteroids and deep sub-tenon triamcinolone, used for quadrants two and three reported by Biswas et al., has a significant effect on visual outcome. [21,22] We used anti-metabolites in the form of methotrexate in 35.1% of cases .[7,22] As suggested by many investigators we did not use anti-tubercular therapy for the treatment of Eales’ disease.[7] We used laser photocoagulation in the ischemic stage (stage 2) or in those with neovascularization, as reported by Biswas et al. [7,23,24]
In the current study, vitrectomy was performed in 32.1% of cases, which is in contrast to the 6–18% reported by Badrinath et al. [19] This may be due to the greater number of patients being in the stage of complications (stage 3) in our study.
Conclusion
We have studied newer aspects of demographic details, anterior segment findings and posterior segment findings. We have found that final visual outcome significantly improved following medical or surgical treatment.
References
- Eales’ H. (1880) Cases of retinal hemorrhage associated with epistaxis and constipation. Birmingham Medical Review 9: 262-3.
- Eales H. (1882) Primary retinal hemorrhage in young men. Ophthalmic Rev 1:41.
- Elliot AJ. (1975) 30-year observation of patient with Eales’ disease. Am J Ophthalmol 80:404–408.
- Wardsworth. (1887) Recurrent retinal haemorrhage followed by the development of blood vessels in the vitreous Ophthalmic Rev 6: 289.
- Das T, Biswas J, Kumar A. (1994) Eales’ disease. Indian J Ophthalmol 42:3–28.
- Duke-Elder S, Dobree JH. System of Ophthalmology Vol X. London: Henry Kimpton.
- Biswas J, Sharma T, Gopal L, Madhavan HN, Sulochana KN, et al. (2002) Eales’ disease: an update. Surv Ophthalmol 47:197–214.
- Madhavan HN, Therese KL, Gunisha P, Jayanthi U, Biswas J. (2000) Polymerase chain reaction for detection of Mycobacterium tuberculosis in epiretinal membranes in Eales’ disease. Invest Ophthalmol Vis Sci 41:822–825.
- Biswas J, Rao NA. (1990) Epiretinal membrane in Eales’ disease and other vascular retinopathies. Invest Ophllmlmol Vis Sci 31: 369-70.
- Badrinath SS, Biswas J, Sharma T, Rao N. (1992) Immunophenotyping of inflammatory infiltrates in epiretinal (ERM) and sub retinal membrane (SRM) in Eales’ disease. Invest Ophthalmol Vis Sci 33: 33-35.
- Das T, Namperumalsamy P. (1987) Combined photocoagulation and cryotherapy in treatment of Eales’ retinopathy. Proc All Ind Ophthalmol Soc 45: 108.
- Saxena S, Kumar D. (2000) Safety of oral methotrexate pulsed therapy. Indian J Ophthalmol 48:251.
- Pathengay A, Pilli S, Das T. (2005) Intravitreal triamcinolone acetonide in Eales’ disease: a case report. Eye 19: 711–713.
- Saxena S, D. Kumar. (2007) New classification system-based visual outcome in Eales' disease. Indian J Ophthalmol 55: 267-9.
- Davis J, S. H. Schecter. (2009) Eales' disease: the great masquerader. Optometry 80: 354-9.
- Shrestha J. K, D. Khadka. (2009) Retinal vasculitis. Nepal J Ophthalmol 1:66-71.
- Stoffelns B. M, C. Kramann. (2009) Eales' disease--10 years experience with a rare disease. Klin Monbl Augenheilkd 226: 299-304.
- Badrinath SS, Gopal L, Sharma T. (1999) Vitreoschisis in Eales disease:pathogenic role and significance in surgery. Retina: 19:51–4.
- Badrinath SS, Gopal L, Sharma T. (1996) Morphological description Of surgical anatomy of Eales disease–A pilot study. Presented at 100th annual meeting of American Academy, Chicago.
- Das TP, Namperumalsamy P. (1990) Photocoagulation in Eales disease. Results of prospective randomized clinical study. Presented at XXVI Int Cong Ophthalmol, Singapore.
- Howe LJ, Stanford MR, Edelsten C, Graham EM. (1994) The efficacy of systemic corticosteroids in sight-threatening retinal vasculitis. Eye 8:443–7.
- Greenwood AJ, Stanford MR, Graham EM. (1998) The role of azathioprinein the management of retinal vasculitis. Eye 12:783–8.
- Dayton GO Jr, Straatsma BR. (1962) Eales disease and photocoagulation. Trans Pac Coast Ophthalmol Soc 43:12–129.
- Gopal L, Abraham C. (1985) Efficacy of photocoagulation in Eales disease. Trans Asia-Pacific Acad Ophthalmol 10:689-90.