Brief Review on Some Major Factors Affecting Visible Light Activity of Titania Photocatalyst
Burtrand I Lee
Department of Materials Science of Engineering, Clemson University, Clemson, SC, USA.
*Corresponding Author
Burtrand I Lee,
Department of Materials Science of Engineering,
Clemson University, Clemson,
SC 29634 USA
Email: lburtra@ yahoo.com
Article Type: Review Article
Received: September 10, 2014; Accepted: September 10, 2014; Published: September 10, 2014
Citation: Burtrand I Lee (2014) Brief Review on Some Major Factors Affecting Visible Light Activity of Titania Photocatalyst. Int J Nano Stud Technol. 3(5), 67-71. doi: dx.doi.org/10.19070/2167-8685-1400013
Copyright: Burtrand I Lee© 2014 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
As sunlight is an excellent source of clean energy, research for harnessing the energy via an efficient photocatalyst that is active under visible light is actively underway. Titania is well accepted as a photosemiconductor and photocatalyst under ultraviolet light. Effort to make it visible light active has been growing. This review article presents some factors involving the photocatalytic activity of titania which is active under visible light in terms of crystallography. Based on recent progress,several suggestions are made for a better visible light active titania.
2.Introduction
3.Results and Discussion
4.References
Key Words
Titania; Photocatalyst; Visible Light; Crystal Lattice; Hetero Junction; Excitons; Charge Separation.
Introduction
Titania is well known for its usefulness as a photocatalyst under ultraviolet light (UV). For more practical and broader applications, the photocatalytic activity (PCA) under visible light (VL) has been actively pursued over a decade. Interest in VL active photocatalysis has been motivated in part by the realization that sunlight is effectively an inexhaustible energy source. Most frequent effort to boost visible light photocatalytic activity (VLA) included doping the titania crystal lattice. More successes have been shown by anions and nitrogen in particular [1-3]. hydrogenated titania surface and increased the solar absorption. Dye-sensitization technique [4- 6] is well utilized. Surface incorporation of high electrical conductivity metal nanoparticles is well known for its surface plasmonics [7- 10].
For high PCA, one of the requirements for a catalyst is high specificarea (SSA). In the synthesis of high SSA nanoparticles, one chooses to use a low temperature method such as solution or solgel process. A dilemma encountered in this is that the particles produced at a low temperature contain much residual impurities such as water (H+ , H2O , OH- , H3O+ ), solvent, and any additives that had to be present to optimize the process [11, 12]. Conventionally these are removed by the subsequent calcinations steps which are also used to promote crystallinity. These steps involving elevated temperatures will cause the surface area reduction as well as any unwanted phase transformation. This may be overcome by nonaqueous solution process followed by low temperature drying or dehydration processes [12- 19].
Another major factor known to affect the PCA is efficient separation of the photogenerated charges, i.e., exctions of e-s and h+s. Heterojunctions are shown to be effective site in reducing the recombination rates of the photogenerated e-s and h+s [13, 20- 32]. Heterointerfaces between crystallographic phases are critical to many photocatalyses including solar energy conversion. Understanding structure/composition at the metal/metal oxide or metal oxide/metal oxide interfaces, the ability to tailor and tune those interfaces are then essential elements in developing more efficient photocatalysts [28- 32]. In this review article, the major factors contributing to the VLA are mentioned and crystallographic features directly related to VLA are discussed.
Titania is known to have three polymorphs: Anatase (An), rutile (Ru), and brookite (Br) [33, 34]. These three TiO2 crystallographic phases have similar chemical properties, but have different crystalline structures. An and Ru are tetragonal, whereas Br is orthorhombic [33]. An phase TiO2 is photocatalytically the most active under UV [29, 33- 37]. At an ambient condition, Ru TiO2 is thermodynamically the most stable phase [33- 35]. An is most widely utilized as photocatalyst under UV because it exhibits the highest SSA with an ease of production [33- 35]. As one calcines An, it begins to transform to Ru at a temperature near 400oC depending on the SSA and/or the crystallite size [33]. Br phase is metastable, least well known, and is the most difficult to synthesize [33- 35]. There are conflicting reports on the photocatalytic properties of Br phase of titania [33-40]. Some claim Br is the most photocatalytically active [34, 38- 40] and some report An is [28]. However, An exhibits the largest band gap energy and hence the least active under VL. A low temperature solution process often yields mixed phase of titania with different proportions depending on the experimental variables [11, 33, 38, 39]. By varying the synthesis conditions, e.g., time, temperature, and pH, the polymorphic phases in titania can be controlled [11, 33, 38, 39].
Along the line can a mixed phase titania be more photocatalytically active than a single or pure phase An? Based on hetrojunctions promoting the charge transport in the excitons across the phase boundary, a mixed phase structure could be more photocatalytically active [35]. One well known example of this is Degussa Aeroxide TiO2, also known as P25, which consists of 79% An and 21% Ru [41].
Results and Discussions
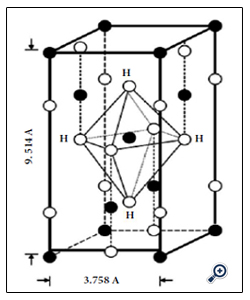
A low temperature synthesis method, called the “water based ambient condition sol” (WACS) process, yielded well crystallized high surface area polymorphic nanoparticle titania with Br as the predominant phase [11, 33, 38, 39]. However, this water based low temperature synthesis method incorporates water or protons from the solvent water into the lattice [11, 33, 38, 39] as shown in Figure 1. The extraneous protons will create cation vacancies, e.g., 4 H+ ions creating 1 Ti4+ ion vacancy by the charge neutrality principle. Upon calcination the protons bonded to oxygen anions can be removed as water by the reaction 2OH → H2O + O2-. Removing the lattice protons and/or synthesis in a lesser water environment has enhanced optical [42], dielectric [43-47], and/or catalytic properties in other metal oxides [17,47,48].
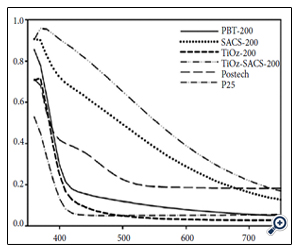
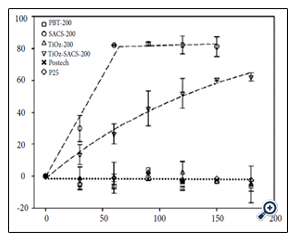
A polymorphic titania with Br as the major phase, i.e., polymorphic brookite Titania (PBT) - produced by WACS process [11-14] followed by a post-treatment in hot N-methylpyrrolidone (NMP) solvent, labeled as SACS200 - is shown to much enhance VLA [12-14]. Table 1 lists the titania samples discussed in this review with the corresponding phase compositions, crystallite sizes, and SSAs of PBT as compared with other reference titanias. Figure 2 shows UV-Vis spectra of these titania [38]. PBT200 which is titania with 53% Br and 45% An before the proton extraction shows some VL absorption while NMP treated PBT, i.e., SACS200 showed much increased VL absorption. Postech TiO2 that is N-doped An titania showed some VL absorption while P25 showed no VL absorption. Nanoparticle titania with crystallite size <10 nm of Br as the predominant phase exhibits a moderate VL absorption. A polar anhydrous solvent NMP treatment of PBT enhanced VLA significantly. These titanias tested for photocatalytic degradation of methyl orange (MO) under VL irradiation with a 14 W fluorescent lamp are shown in Figure 3 [12]. SACS200 which is An/Br mixed phase shows superior VLA to NMP treated TiOz (TiOzSACS200) despite its higher SSA. The difference between the two titanias is that SACS200 is An/ Br mixture and TiOzSACS200 is single phase An. P25 treated with NMP (not shown here) hardly showed any enhanced VLA [12-14]. This means that the superior VL PCA of SACS200 must be attributable to the mixed phase of Br and An.
Table 1. Titania samples with phase compositions, the corresponding crystallite sizes, and specific surface areas.
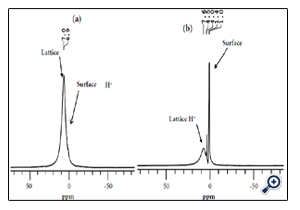
Figure 4. Solid state 1H MAS NMR spectra of (a) before NMP treatment and b) after NMP treatment of PBT.
The superior VL photocatalytic activity was explained as the effective charge separation by the intercrystalline electron transport from Br to An grains complemented by the strong VL absorption with the nitrogen species incorporated from NMP. The NMP here had a dual role of removing the entrapped protons from the crystal lattice of titania and introducing VL absorbing nitrogen doping effect. These lattice protons extracted by NMP at a low temperature was confirmed by TGA, FTIR and solid state proton MAS-NMRspectroscopy as shown in Figure 4 [14].
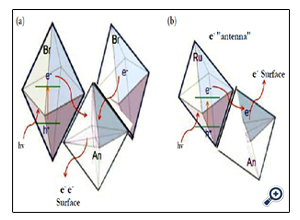
Figure 5. Illustration of charge separation mechanism for the enhanced VLA in (a) SACS200 analogous to (b) P25 under UV.
The significance of this technique is tailoring the defect chemistry without sacrificing the surface area and the optimum polymorphic composition. This crystallographic composition has also shown superior antimicrobial activity to single phase reference titanias [49] The Br/An mixed phase titania is analogous to the An and Ru mixed phase in P25 as shown in Figure. 5. The superior UV photocatalytic activity of P25 has been delineated by the efficient intercrystalline electron transport and charge separation by the minor rutile phase acting as the electron “antenna” [50]. Ozawa et al. [51] proposed strong coupling between An and Ru in their mixed phase titania. Xia et al. [52] supported the efficient charge separation in P-25 titania via molecular dynamic simulation and density functional theory along with Raman spectroscopy. Gai et al. [36] also showed superior PCA for an N-doped An/Br polymorph by efficient electron transport of the excited e- from Br to An. The probable mechanism, analogous to the commercial reference titania P25, suggests an effective intergrain electron transfer and charge separation. This, in turn, reduced the electron-hole recombination rate [12,13,35,36,39].
The importance of charge separation via heterojunction in photosemiconductors for a more efficient PCAhas been emphasized by many researchers [20-29,50-52]Xia et al. [52].showed by density of states computed by DFT that the band gap in a mixed phase titania decreased from 3.2 eV of An to 2.0 eV as the proportion of rutile phase increased up to about 50%.Li et al.[40] presented Br having indirect band gap energy of 2.85 eV. These suggest the band gap of Br/An polymorph is significantly below that of An (3.2 eV) to absorb VL.
As the calcination temperatures of PBT increased, Br and An in PBT transformed to Ru [11]. Also as Ru phase increased and Br phase decreased in the polymorph, the VLA decreased [33]. Postech sample, which is a hydrothermal nitrogen doped single phase An, absorbed some VL as shown in Figure 2 of UV/Vis spectra. However, it was VL inactive under the 14 W VL source for MO degradation (Figure 3). SACS200 titania has shown to be superior to all other reference titanias in generating hydrogen from aqueous EDTA solution under VL [13]. Hydrogen generation rate from water was shown to follow the same trend as shown for MO photocatalytic decomposition in Figure 3. HBK titania is a commercial anatase titania with the name Hombikat having SSA > 200 m2/ g. Despite the high surface area of HBK, the hydrogen generation under VL was only that of untreated PBT, which has SSA of ~160 m2/ g. The crystallographic difference between HBK and PBT is the phase composition. HBK is fully An but PBTis An with more than 50% brookite.
Therefore, the major contributing factors for the observed superior VL-PCA from the An/Br polymorphic SACS200 are listed as:
(1) The lower lattice hydroxyl content [11-13],
(2) nitrogen doping [12] from NMP for enhanced VL absorption, and
(3) the effective charge separation and intergrain transfer of VL excited electrons from one phase to the other [11-13,36].
So several opportunities to further enhance VL-PCA and photochemical reactions in titania are:
a) Tailoring the crystallography of polymorphic titania with respect to crystallographic phase composition during the synthesis, though the optimum phase composition is yet to be determined [13],
b) Deprotonation or dehydroxylation from the crystal lattice at a low temperature not losing the high SSA and without altering the optimum phase composition/distribution for the maximum PCA, and
c) Introducing or incorporating VL absorbing chromophore in titania that extracts the entrapped protons at the same time.
References
- Chen X, Mao SS (2007) Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications. Chem Rev 107: 2891-2959.
- Zhou L, Deng J, Zhao Y, Liu W, An L, et al, (2009) Preparation and characterization of N–I co-doped nanocrystal anatase TiO2 with enhanced photocatalytic activity under visible-light irradiation Materials Chem and Physics 117: 522.
- Chen X, Liu L, Yu PY, Mao SS (2011) Increasing solar absorption for photocatalysis with black hydrogenated titanium dioxide nanocrystals. Science. 331: 746-750.
- Kruth A, Antje Quade, Volker Brüser, Klaus-Dieter Weltmann (2013) Plasma- enhanced synthesis of poly(allylamine)-encapsulated ruthenium dyesensitized titania photocatalysts. J Phys Chem. C. 117: 3804-3811.
- Lee CH, Shie JL, Tsai CY, Yang YT, Chang CY (2014) Photoelectrochemical characteristics of N-and S-doped TiO2 with coumarin. Journal of Clean Energy Technologies 2: 38-42.
- Li Z, Fang Y, Zhan X, Xu S (2013) Facile preparation of squarylium dye sensitized TiO2 nanoparticles and their enhanced visible-light photocatalytic activity. Journal of Alloys and Compounds 564:138.
- Chen Q, Shi H, Shi W, Xu Y, Wu D (2012) Enhanced visible photocatalytic activity of titania–silica photocatalysts: effect of carbon and silver doping. Catal Sci Technol 2: 1213-1220.
- Seery MK, George R, Floris P, Pillai SC (2007) Silver doped titanium dioxide nanomaterials for enhanced visible light photocatalysis. Journal of Photochemistry and Photobiology A: Chemistry. 189: 258-263.
- Kuhn JN, Qayyum E, Castillo VA, Warrington K, Barakat MA (2013) Immobilization of silver-based bimetallic nanoparticles on titania-support for photocatalysis . J. of Physics: Conference Series. 431: 012020.
- Dunnill CW, Ansari Z, Kafizas A, Perni S, Morgan DJ, et al, (2011) Visible light photocatalysts-N-doped TiO2 by sol-gel, enhanced with surface bound silver nanoparticle islands. J Mater Chem. 21: 11854-11861.
- Lee BI, Wang XY, Bhave R, Hu M (2006) Synthesis of brookite TiO2 nanoparticles by ambient condition sol process. Mater Lett. 60: 1179-1182.
- Sujaree Kaewgun, Daniel Mckinney, Jamie White, Andrew Smith, Michael Tinker, et al. (2009) Study of visible light photocatalytic activity achieved by NMP solvent treatment of polymorphic titania. J of Photochem & Photobio.A: Chemistry. 202: 154-158.
- Lee BI, Kaewgun S, Kim W, Lee J, Choi W (2009) Visible light photocatalytic properties of polymorphic brookite titania. J Renew & Sustainable Energy.1: 023101.
- Kaewgun S, Nolph CA, Lee BI, Wang LQ (2009) Influence of hydroxyl contents on photocatalytic activities of polymorphic titania nanoparticles.Mater Chem Phys.114: 439-445.
- Sato T, Aita Y, Komatsu M, Yin S (2006) Solvothermal synthesis of visible light responsive nitrogen-doped titania nanocrystals. J Mater Sci. 41: 1433-1438.
- Yin S, Sato T (2000) Synthesis and photocatalytic properties of fibrous titania prepared from protonic layered tetratitanate precursor in supercritical alcohols. Ind Eng Chem Res. 39: 4526-4530.
- Yin S, Fujishiro Y, Wu JH, Aki M, Sato T (2003) Synthesis and photocatalytic properties of fibrous titania by solvothermal reactions. J Mater Process Technol. 137: 45-48.
- Yin S, Inoue Y, Uchida S, Fujishiro Y, Sato T (1998) Crystallization of titania in liquid-media and photochemical properties of crystallized titania. J Mater Res. 13: 844.
- Yin S, Aita Y, Komatsu M, Wang JS, Tang Q, et al, (2005) Synthesis of excellent visible-light responsive TiO2−xNy photocatalyst by a homogeneous precipitation-solvothermal process. J Mater Chem.15: 674-682.
- Cowan SR, Roy A, Heeger AJ (2010) Recombination in polymer- fullerene bulk heterojunction solar cells. Phys Rev B. 82: 245207.
- Reusch M, Bivour M, Hermle M, Glunz SW (2013) Fill factor limitation of silicon heterojunction solar cells by junction recombination. Energy Procedia.38: 297-304.
- Zhou S, Sun J, Zhou C, Deng Z (2013) Comparison of recombination models in organic bulk heterojunction solar cells. Physica B: Condensed Matter. 415: 28-33.
- Ishihara H, Kannarpady GK, Woo J, Biris AS (2013) Enhanced photocurrent response of titania-nanotube hetero junction devices capped with titanium disilicide. Energy Technology.1: 412-418.
- Etacheri V, Michlits G, Seery MK, Hinder SJ, Pillai SC (2013) A highly efficient TiO(2-x)C(x) nano-heterojunction photocatalyst for visible light induced antibacterial applications. ACS Appl Mater Interfaces. 5: 1663-1672.
- Avasthi S, McClain WE, Man G, Kahn A, Schwartz J, et al, (2013) Holeblocking titanium-oxide/silicon heterojunction and its application to photovoltaics. Appl. Physic Letters. 102: 203901.
- Zhang J, Zhu Z, Tang Y, Müllen K, Feng X (2014) Titania nanosheet-mediated construction of a two-dimensional titania/cadmium sulfide heterostructure for high hydrogen evolution activity. Adv Mater. 26: 734-738.
- Niraula M, Adhikari S, Lee DY, Kim EK, Yoon SJ, et al. (2014) Titania nanotube-silver phosphate hybrid heterostructure for improved visible light induced photocatalysis. Chemical Physics Letters. 593: 193-197.
- Haruta M, Yamada N, Kobayashi T, Iijima S (1989) Gold catalysts prepared by coprecipitation for low-temperature oxidation of hydrogen and of carbon monoxide. J Catal. 115: 301-309.
- Haruta M, Date M (2001) Advances in the catalysis of Au nanoparticles. Appl Catal. 222: 427-437.
- Liu L, Ouyang S, Ye J (2013) Gold-nanorod-photosensitized titanium dioxide with wide-range visible-light harvesting based on localized surface plasmon resonance. Angew Chem Int Ed Engl. 52: 6689-6693.
- Jianjun Guo, Shuxin Ouyang, Peng Li, Yuanjian Zhang, Tetsuya Kako, et al, (2013) A new heterojunction Ag3PO4/Cr-SrTiO3 photocatalyst towards efficient elimination of gaseous organic pollutants under visible light irradiation. Appl Catal. 134: 286-292.
- Tong H, Ouyang S, Bi Y, Umezawa N, Oshikiri M, et al, (2012) Nanophotocatalytic materials: possibilities and challenges. Adv Mater. 2012; 24:229-251.
- Bhave RC, Lee BI. (2007) Experimental variables for the formation of brookite phase TiO2 nanoparticles Mater Sci Eng. A 467: 146-149.
- Kandiel TA, Robben L, Alkaim A, Bahnemann D (2013) Brookite versus anatase TiO2 photocatalysts: phase transformations and photocatalytic activities. Photochem Photobiol Sci. 12: 602-609.
- Di Paola, Bellardita M, Palmisano L (2013) Brookite, the least known TiO2 photocatalyst. Catalysts. 3: 36-73.
- Gai L, Duan X, Jiang H, Mei Q, Zhou G, et al. (2012) One-pot synthesis of nitrogen-doped TiO2 nanorods with anatase/brookite structures and enhanced photocatalytic activity Cryst Eng Comm. 14: 7662-7671.
- Lin H, Li L, Zhao M, Huang X, Chen X, et al. (2012) Synthesis of highquality brookite TiO2 single-crystalline nanosheets with specific facets exposed:tuning catalysts from inert to highly reactive. J Am Chem Soc. 134:8328-8331.
- Kaewgun S, Nolph CA, Lee BI (2008) Enhancing photocatalytic activity of polymorphic titania nanoparticles by NMP solvent-based ambient condition process. Catalysis Letters. 123: 173-180.
- Nolph CA, Sievers DE, Kaewgun S, Kucera CJ, McKinney DH, et al. (2007) Photocatalytic study of polymorphic titania synthesized by ambient condition sol process. Catalysis Letters.117: 102-106.
- Li JG, Ishigaki T, Sun XD (2007) Anatase, brookite, and rutile nanocrystals via redox reactions under mild hydrothermal conditions: phase-selective synthesis and physicochemical properties. J Phys Chem C. 111: 4969.
- Degussa, AEROXIDE and AEROPEAL (2005) Titanium dioxide as photocatalyst. Degussa Technical Information. 1243.
- Joshi P, Richards B, Jha A (2013) Reduction of OH− ions in tellurite glasses using chlorine and oxygen gases. Journal of Materials Research. 28: 3226-3233.
- Maie H, Lee BI (2009) Hydroxyl effects on dielectric properties of nanoparticle barium titanate. J Mater Sci. Materials in Electronics. 20: 619-627.
- Qi L, Lee BI, Badheka P, Wang LQ, Gilmour P, Samuels WD, Exarhos GJ (2005) Low temperature paraelectric-ferroelectric phase transformation in hydrothermal BaTiO3 particles. Mater Lett. 59: 2794-2798.
- Kota R, Lee BI (2007) Effect of lattice hydroxyl on the phase transition and dielectric properties of barium titanate particles. J Mater Sci. Electronics 18:1221-1227.
- Badheka P, Magadala V, Devaraju NG, Lee BI, Kim ES (2006) Effect of dehydroxylation of hydrothermal barium titanate on dielectric properties in polystyrene composite. J Appl Polymer Sci. 99: 2815–2821.
- Nam CT, Yang WD, Duc L (2013) Solvothermal synthesis of TiO2 photocatalysts in ketone solvents with low boiling points. Journal of Nanomaterials,627385.
- 48.Tong H, Umezawa N, Ye J (2011) Visible light photoactivity from a bonding assembly of titanium oxide nanocrystals. Chem Commun (Camb).47:4219-4221.
- Shah RR, Kaewgun S, Lee BI, Tzeng TR (2008) The antibacterial effects of biphasic brookite-anatase titanium dioxide nanoparticles on multiple-drugresistant staphylococcus aureus. J Biomedical Nanotechnology. 4: 339-349.
- Hurum DC, Agrios AG, Gray KA, Rajh T, Thurnauer MC (2003) Explaining the enhanced photocatalytic activity of Degussa P25 mixed-phase TiO2 using EPR. J Phys Chem B. 107: 4545-4549.
- OzawaT, Iwasaki M, Tada H, Akita T, Tanaka K, et al. (2005) Low-temperature synthesis of anatase-brookite composite nanocrystals: the junction effect on photocatalytic activity. J Colloid Interface Sci. 281: 510-513.
- Xia T, Li N, Zhang Y, Kruger MB, Murowchick J, et al. (2013) Directional heat dissipation across the interface in anatase-rutile nanocomposites. ACS Appl Mater Interfaces. 5: 9883-9890.