Premature Exfoliation of Deciduous Teeth in a Four-Year-Old Child with Hypophosphatemic Rickets
Chalbi M1*, Gharbi I2, Jemmali B2
1 Resident in Pediatric Dentistry, La Rabta Hospital, Tunisia.
2 Professor in Pediatric Dentistry, La Rabta hospital, Tunisia.
*Corresponding Author
Manel Chalbi,
Resident in Pediatric dentistry, La Rabta Hospital, Tunisia.
Tel: 0021652204476
E-mail: manelchalbi88@gmail.com
Received: June 18, 2017; Accepted: July 25, 2017; Published: July 27, 2017
Citation: Chalbi M, Gharbi I, Jemmali B (2017) Premature Exfoliation of Deciduous Teeth in a Four-Year-Old Child with Hypophosphatemic Rickets. Int J Dentistry Oral Sci. 4(7), 508-512. doi: dx.doi.org/10.19070/2377-8075-17000100
Copyright: Chalbi M©2017. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Vitamin D is an essential hormone for calcium gut absorption. It is also involved in child growth, cancer prevention, immune system responses, and tooth formation. Vitamin D deficiency is a common cause of rickets, a condition that affects bone development in children and that can have serious dental complications.
Case Report: The following article presents a case report of a 4 year old Tunisian child, diagnosed with Hypophosphatemic rickets. Clinical features include growth failure, frontal bulging and genu valgum. Dental examination revealed spontaneous loss of deciduous teeth in the absence of dental caries or trauma.
Follow up: Two years on, we noted a markedly hypoplastic yellow to brownish enamel in the first permanent molars and permanent lower incisors.
Conclusion: The purpose of this article is to report and discuss the dental alterations observed: spontaneous loss of teeth and hypoplastic structure of enamel. Indeed, our patient presents the second case reported in literature with periodontal breakdown associated to the hypophosphatemic rickets. Genetic and molecular testing is needed to verify this.
2.Background
3.Case Report
4.Discussion
5.Conclusion
6.References
Keywords
Hypophosphatemic Rickets; Child; Periodontitis; Hypoplastic Enamel.
Background
Hypophosphatemic rickets (HR), is a group of hereditary metabolic diseases which is characterized by diminished phosphate reabsorption in renal tubules leading to chronic hyperphosphaturia and hypophosphatemia, associated with normal or low levels of 1,25(OH)2 Vitamin D3 [1, 2].
The different forms of Hypophosphatemic Rickets were classified according to their mode of inheritance into autosomic dominant (ADHR), autosomic recessive (ARHR) and X-linked dominant (XLH) from (Souza, et al 2010). XLH is the most frequent form of infantile rickets also known as vitamin D resistant rickets. The rate is usually reported to be around 1:20000 [2-4].
The basic physiological defect is impaired proximal renal TRP, due to an error in the X chromosome, especially The PHEX gene (phosphate regulating gene with homologies to endopeptidases on the X chromosome), promoting elimination of phosphates through the proximal renal tubules, causing phosphaturia and consequently hypophosphatemia.
This mutant gene is expressed in cells such as osteoblasts, osteocysts, odontoblasts, and parathyroid glands, resulting in abnormal osteogenesis and odontogenesis (Barconelli et al, 2006), which causes defective calcification of mineralized structures such as bones and teeth [1, 2].
Recently, one of the different modes of inheritance was described as autosomal recessive hypophosphatemia, also known as vitamin D dependent rickets, and it was found that is caused by mutations in Dentine Matrix Protein (DMP1). This protein is also highly expressed in osteocytes in bone as well as in teeth (Feng et al 2003) [5].
Rickets signs and symptoms are frequently evident when patients reach about 18 months [1, 4].
It can vary from patient to another owing to several factors: family history, the degree of hypophosphatemia, and the age at the start of systemic treatment. The main clinical symptoms are severe bone deformities, particularly a characteristic bowing of the legs. This is the major sign of the disease and is most obvious with the onset of the child’s first steps. In addition, these patients present frontal bulging due to sagittal suture synostosis, costrochondral joint enlargement, genu valgum, genu varum and short stature [1, 6].
Its dental significance was widely described in literature. The main dental manifestations reported were: spontaneous periapical infections, hypoplastic enamel, abnormal lamina dura, ricketly bone trabeculation [7].
However, the possible association between HR and periodontal breakdown was reported for the first time by Al-Jundi SH, Hammad MM, I. European [2011] [5].
One study using DMP1-null mice (a mouse model of HR) demonstrated an early onset periodontal defect in these mice with porous alveolar bone and defective cementum and ligament [8].
We report a case of child diagnosed with HR who showed excessive periodontal breakdown, which may indicate a possible association with mutation of DMP1.
Case Report
A 4-year-old male patient was been referred from the pediatric service to the pediatric dentistry department in March 2014 for a dental evaluation. The patient was diagnosed as having hypophosphatemic rickets. Upon arrival, it was been reported he was under treatment of vitamin D, phosphoneuros, and Alpha glob. The patient was complaining of premature loss of the deciduous incisors within a year since its eruption. Biological studies revealed phosphor in serum 20.4mg/L normal values (50- 70 mg/L), phosphatase alkaline 1011U/L normal values (1-720), Parathormone (PTH) 43pg/ml normal values (15 - 72 pg. /L) and 24-hour urinary calcium 22.95mg/24h normal values 2 - 4 mg/kg/24h.
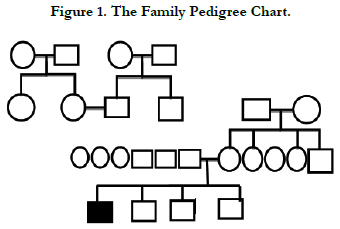
Those results confirmed the diagnosis of hypophosphatemic rickets. The patient case is issued from a consanguineous marriage. The family pedigree chart shown in (Figure 1) reveals typical autosomic transmission of the condition.
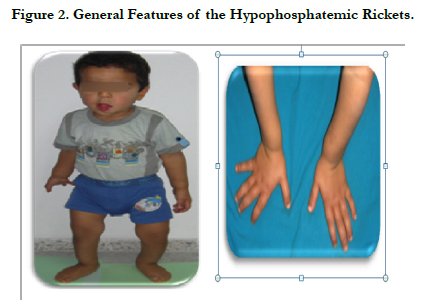
The general physical examination revealed the main clinical characteristic of base diagnosis emitted: bulging frontal bone, occipital hump, typical bowing legs marked a genu varum or “knock knee deformity” and enlargement wrists (Figure 2).
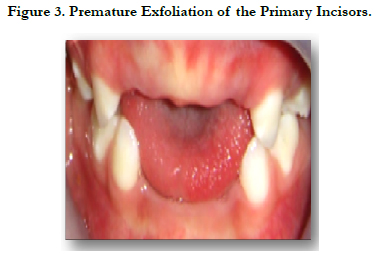
Intraoral examination revealed upper arch with caries free, incomplete primary dentition, absence of teeth 51, 61, 52 and 62 without history of trauma or decay. It is due probably to the Hypoplastic alveolar bone and the defective acellular cementum (Figure 3).
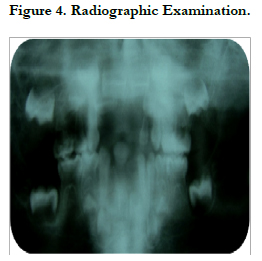
The radiographic examination: showed an enlarged pulp chamber with high pulp horns extending to the dentino-enamel junction in the lower second temporary molars (75, 85). Indeed, Persons with hypophosphatemic rickets have enlarged pulp chambers and increased predentin width (Goodman et al., 1998; Murayama et al., 2000; Pereira et al., 2004) [8].
The osseous structure showed less dense trabeculation particularly on the alveolar bone (Figure 4).
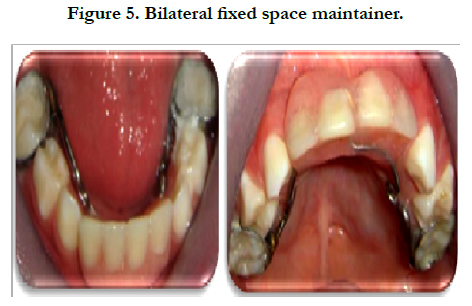
Treatment: In order to maintain the space of the primary maxillary and mandibular incisors, a bilateral fixed space maintainer was been made and cemented with glass ionomer cement (Figure 5).
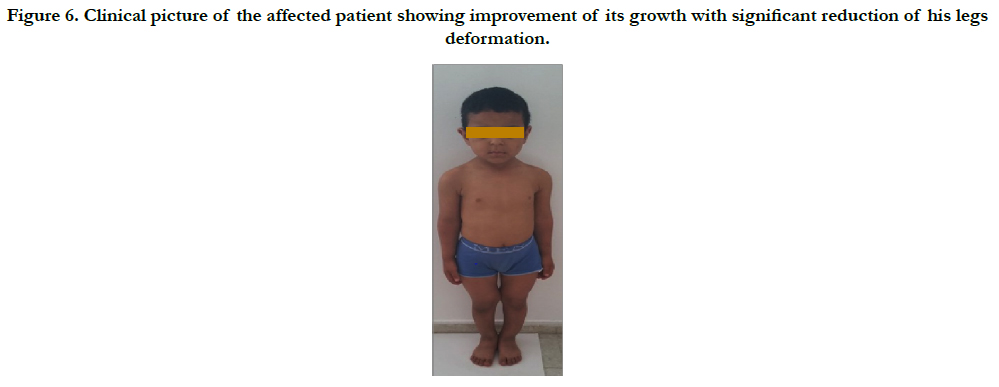
Follow up: In march 2016, when he was six years old, we noted a remarkable improvement in its growth, whose weight and height decreased for this period from 80cm to 1.13 m and from 14 kg to 20kg (Figure 6).
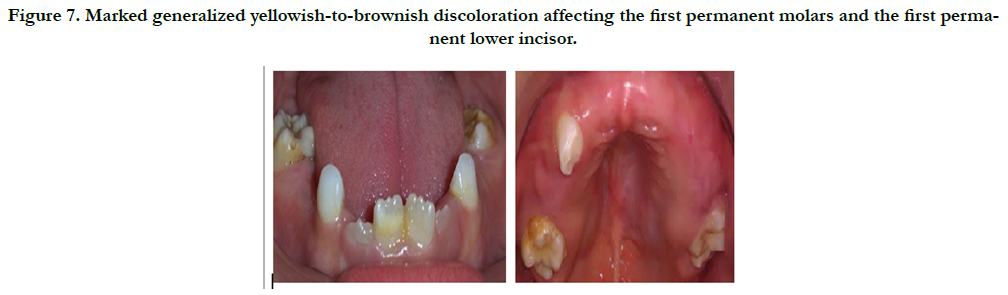
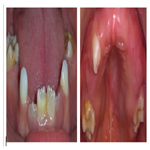
The intraoral examination revealed spontaneous loss of the upper primary canine tooth (53), the primary molars and markdely hypoplastic yellowich to brownich enamel in the first permanent molars and the permannet lower incisor (Figure 7).
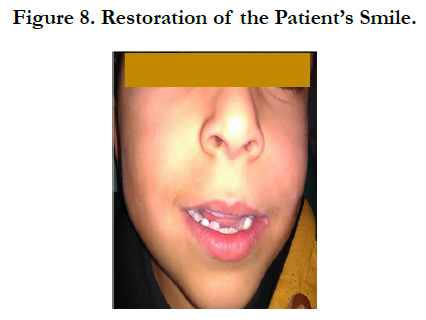
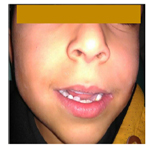
We restored those teeth with dental composite materials. In order to maintain the space of the deciduous teeth and to restore the smile of our child, a removable space maintainer was been made (Figure 8).
Figure 6. Clinical picture of the affected patient showing improvement of its growth with significant reduction of his legs deformation.
Figure 7. Marked generalized yellowish-to-brownish discoloration affecting the first permanent molars and the first permanent lower incisor.
Discussion
Vitamin D is an essential hormone for the absorption of calcium, bone growth of the child and for the formation of dental tissues. In order to exert its effects, vitamin D must be activated by a metabolic pathway that requires the enzymatic activity of 25-hydroxylase and 1 hydroxylase [12].
Hypophosphatemic rickets is a rare inherited disorder characterized by a disorder in phosphocalcic metabolism following vitamin D deficiency.
Three modes of genetic transmission have been described in the literature:
X-linked dominant (XLH), Autosomic dominant (ADHR), and autosomic recessive (ARHR).
The most common laboratory findings in the last form include hypocalcaemia, hypophosphatemia, elevated parathormone levels, and high alkaline phosphatase.
Moreover, the serum levels of 25(OH) vitamin D are normal or raised, while the levels of 1, 25(OH)2 vitamin D are low or undetectable [9].
In our case, on the basis of the family pedigree, the biological exams and the patient’s response to the treatment, the diagnosis of Vitamin -D dependent rickets (VDDR) was probably established.
The dental alterations were observed: premature loss of deciduous teeth and markedly hypoplastic enamel in the first permanent molars and the permanent lower incisors.
Systemic diseases associated with periodontitis may include Papillon-Lefevre syndrome, hypophosphatasia, leukaemias, Chediak-Higashi syndrome, Langerhan’s cell histocytosis, and neutropenia and leukocyte adhesion deficiency [10].
Spontaneous loss of teeth without history of trauma or decay or abscesses is not the reported feature described with Hypophosphatemic rickets.
More recently an association between hyposphatemic rickets and aggressive periodontitis has been reported for the first time by [Al-Jundi et al., 2011] in the European Archives of Paediatric Dentistry [5].
In deed Vitamin D dependent Rickets is caused by mutations in Dentine Matrix Protein (DMP1): an extracellular matrix protein and a member of the small integrin binding ligand N-linked glycoprotein family. This protein is essential in the maturation of odontoblasts and osteoblasts, as well as in mineralization via local and systemic mechanisms [Feng et al., 2003]. It is highly expressed in alveolar bone and cementum, which are important components of the periodontium.
An early-onset periodontal defect was observed in the Dmp1 null mouse, a mouse model of hypophosphatemic rickets. The alveolar bone is porous, with increased proteoglycan expression. The cementum is also defective, as characterized by irregular, punctuate fluorochrome labeling and elevated proteoglycan. The osteocyte and cementocyte Lacuno-canalicular system of both alveolar bone and cementum is abnormal, with irregular lacunar walls and fewer canaliculi [8].
Therefore, we hypothesized that Dmp1 is critical for the integrity of the periodontium, and that deletion may lead to increased susceptibility to disease.
This may explain the periodontal breakdown observed in our case.
Dental examination revealed also markedly hypoplastic, yellowish-to-brownish enamel in first permanent molars and lower permanent incisors.
In a study about children with chronic disorders of calcium and phosphate homeostasis enamel hypoplasia was found in hereditary vitamin D-dependency rickets and in hypoparathyroidism [11].
The enamel hypoplasia was occurred only in case of children who had hypocalcaemia. In fact, crown mineralization of primary teeth occurs from gestation to 12 months of age, and that of permanent teeth extends from birth to 8 years of age, except for third molars [9]. For our patient, hereditary vitamin D-dependency rickets was diagnosed at the age of two years, after the beginning of mineralization of the first molars and incisors, which explains the structural defect already described.
Clearly, the genetic counseling is essential to establish early diagnosis in order to institute the necessary medicinal therapy (supplements in vitamin D and phosphorus) and thus minimize dental defects.
Conclusion
In conclusion, we have described hypophosphatemic rickets in a 4-year-old Tunisian boy. Patient serologic, general, clinical, oral, dental and radiologic features are delineated.
Our patient is the second case reported in literature diagnosed with HR with premature exfoliation of teeth associated to the hypophosphatemic rickets. Genetic and molecular testing is needed to verify this.
References
- Gabriela Godina H, Francisco Belmont L (2013) Dental characteristics of hypophosphatemic rickets. Case report. Revista Odonatological Mexicana. 17(2): 101-108.
- I Cremonesi I, Nucci C, Piana G, D’Alessandro G, Marchionni S, et al., (2014) Dental findings in X-linked hypophosphatemic rickets. Scanning. 36(4): 456–461.
- Carpenter TO, Imel EA, Holm I, Suzanne MJ, Insoqna K, et al., (2011) A Clinician’s Guide to X-Linked Hypophosphatemia. J Bone Miner Res. 2(7): 1381–1388.
- Carvalho CN, Freire LG, Bauer J, Azevedo G, Carolina F, et al., (2014) Hypophosphatemic vitamin D resistant rickets: clinical,radiographic and histologic findings. Indian j dent. 5: 100-113.
- SH Al-Jundi, MM Hammad, I Dabous (2011) Case Report: Hypophosphatemic rickets and aggressive periodontitis: a review of the role of dentine matrix protein 1 in the pathogenesis. Eur Arch Paediatr Dent. 12(1): 46-50.
- Diane D, Joseph C, Gaucher C, Chaussain C, Courson F (2009) Familial hypophosphatemic vitamin D-resistant rickets-prevention of spontaneous dental abcesses on primary teeth. A case report. Oral Surg Oral Med Patho Oral Radiol Endod. 107(4): 525-530.
- Ziaee V, Rabbani A (2013) Hypophosphatemic Rickets and its Dental Significance. Iran J Pediatr. 23(6): 715.
- L Ye, S Zhang, H Ke, LF Bonewald, Feng JQ (2008) Periodontal Breakdown in the Dmp1 Null Mouse Model of Hypophosphatemic Rickets. J Dent Res. 87(7): 624-629.
- Zambrano M, Nikitakis NG, Sanchez-Quevedo M, Sauk J, Rivera H, et al., (2003) Oral and dental manifestations of vitamin D–dependent ricketstype I: Report of a pediatric case. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 95(6): 705-9.
- G Sharma, R Whatling (2011) Case Report: Premature exfoliation of primary teeth in a 4-year-old child, a diagnostic dilemma. Eur Arch Paediatr Dent. 12(6): 312-317.
- Nikiforuk G, Fraser D (1981) The etiology of enamel hypoplasia: a unifying concept. J Pediatr. 98(6): 888-93.
- Tiphaine D, Julie G, Pauline A, Jeanne M , Maryse W (2014) Dental Complications of Rickets in Early Childhood: Case Report on 2 Young Girls. Pediatrics. 133(4): e1077-1081.