Castelli Risk Index, Atherogenic Index of Plasma, and Atherogenic Coefficient: Emerging Risk Predictors of Cardiovascular Disease in HIV-Treated Patients
Adedokun AK1,6*, Olisekodiaka MJ1,4, Adeyeye DA2, Muhibi AM3, Ojokuku OH1, Adepeju AA2, Onifade AA5, Adetoro AT1, Ajibola AK1, Sheu MR7
1 Department of Biomedical Science, Ladoke Akintola University of Technology, Osogbo, Osun State, Nigeria.
2 Department of Chemical Pathology, Ladoke Akintola University of Technology Teaching Hospital, Osogbo, Osun State, Nigeria.
3 Department of Haematology and Blood Group Serology, Ladoke Akintola University of Technology Teaching Hospital, Osogbo, Osun State, Nigeria.
4 Department of Chemical Pathology, Nnamdi Azikwe University, Awka, Anambra State, Nigeria.
5 Department of Chemical Pathology, University of Ibadan, Ibadan, Oyo State, Nigeria.
6 Department of Surgical and Diagnostic Oral Maxillofacial Histopathology, DUH, King Saudi University, Riyadh, Saudi Arabia.
7 Department of Environmental Science, King Saud University, Riyadh, Saudi Arabia.
*Corresponding Author
Adedokun A. Kamoru,
Department of Biomedical Science,
Ladoke Akintola University of Technology,
Osogbo, Osun State, Nigeria.
Tel: +2348030517400/+966552442026
E-mail: adeolokun@yahoo.com/kadeokun@ksu.edu.sa
Received:October 04, 2017; Accepted: November 20, 2017; Published: November 23, 2017
Citation: Adedokun AK, Olisekodiaka MJ, Adeyeye DA, Muhibi AM, Ojokuku OH, Adepeju AA, et al., Castelli Risk Index, Atherogenic Index of Plasma, and Atherogenic Coefficient: Emerging Risk Predictors of Cardiovascular Disease in HIV-Treated Patients. Int J Clin Trails Case Stud. 2017;2(3):8-15.
Copyright: Adedokun AK© 2017. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Atherogenic cardiovascular risk assessment is important in the management of dyslipidaemia associated with antiretroviral therapy (ART). Generally, Castelli’s Risk Index (CRI), Atherogenic Index of Plasma (AIP) and Atherogenic coefficient (AC) are becoming useful indices as risk predictors for cardiovascular disease (CVD). We studied the importance of these indices in identifying at-risk HIV individuals receiving treatment. Eighty (80) HIV-patients comprising treatment (n=40) and treatment- naïve (n=40) groups with age- and sex-matched were enrolled. Traditional lipid parameters {Total cholesterol (TC), Triglycerol (TG), and HDLc} were analyzed using spectrophotometry. The LDLc was calculated using Friedewald equation. Non-HDLc, CRI-I (TC/HDLc ratio) and CRI-II (LDLc/HDLc ratio), AIP {Log10 (TG/HDL-c)} and AC {(Non-HDLc)/ HDLc} were also calculated. The CD4+ cell count was determined using flow cytometry. The correlations between atherogenic indices and lipid profile were determined using Pearson’s correlation. P<0.05 was considered as significant. Risk estimation by stratification based on number of altered lipid profile parameter showed no significant increase (P>0.05) in any of the traditional lipid profile parameters except for HDLc with significant elevation (P<0.001) across the strata. Remarkably, all estimated atherogenic indices were found significantly high (P<0.001) across the strata. However, these indices showed positive significant correlations with LDLc and nonHDLc (p<0.001), but negative significant correlations with HDLc (p<0.01). In this present study, the estimated risk indices were suggestive of atherogenicity that helped identifying individuals with dyslipidemia at risk of CVD. Findings from this study also indicated that without markedly deranged lipid profile these estimated indices could serve as more sensitive risk predictors.
2.Abbreviations
3.Introduction
4.Materials and Methods
4.1 Subject Selection and Sample Size
4.2 Inclusion and Exclusion Criteria
4.3 Laboratory Methods
4.4 Statistical Analysis
4.Results and Interpretation
5.Discussion
6.Conclusion
7.Limitations
8.Acknowledgments
9.References
Keywords
Antiretroviral Therapy; Atherogenic Coefficient; Atherogenic Index of Plasma; Castelli’s Risk Index; Cardiovascular Disease; Dyslipidemia.
Abbreviations
ART: Antiretroviral Therapy; AC: Atherogenic Coefficient; AIP: Atherogenic Index of Plasma; CRI: Castelli’s risk index; CVD: Cardiovascular Disease.
Introduction
Our recent study shows that abnormal lipid profile is associated with antiretroviral therapy (ART) and there are significant positive associations of dyslipidemia as well as some lipid ratios (LDL-c/ HDL-c and TC/HDL-c ratios) with ART in the management of human immunodeficiency virus (HIV) [1]. However, only few of these lipid ratios were laid to emphasis within the scope of the study. Currently, the emerging lipid ratios are Castelli’s risk index I and II (CRI-I and CRI-II), atherogenic risk of plasma (AIP) and atherogenic coefficient (AC), respectively [2].
On a general note, recent data have shown that CRI-I and CRI-II (estimated as TC/HDL-c and LDL-c/HDL-c ratios) are more accurate predictors of cardiovascular risk than traditional lipid parameters such as serum total cholesterol (TC), serum triglycerol (TG), serum high density lipoprotein cholesterol fraction (HDL-c) and serum low density lipoprotein cholesterol fraction (LDL-c). CRI-I, calculated from the ratio of two important parameters, TC and HDLc, both of which are reported independent risk factors for coronary artery disease (CAD) is similar with CRI-II CAD in risk evaluation. CRI-II, also calculated as the ratio of LDLc and HDLc, has been reported to involve in predicting risk of CAD [2]. Indeed, changes in these ratios were equally reported to be better predictors of successful coronary heart disease (CHD) risk reduction when compared with changes in absolute levels of lipids or lipoproteins [3].
In addition, comparison of individual lipid ratio in subjects from the report of Framingham Study clearly indicates that lipid ratios are significantly more useful as predictors of cardiovascular diseases (CVD) than the individual levels of LDLc or HDLc [4]. Furthermore, earlier investigation, the Helsinki Study, reported that the lipid ratio defined as LDLC/HDL-C (known here as CRI-II) presented more prognostic value when compared with ordinary LDLc or HDLc fraction [5].
In a related but different term, another lipid ratio, AIP defined as log (TG/HDLc) was proposed as a marker of plasma atherogenicity [6]. Since then, series of reports have been emerging; studies revealed that in circumstances where other atherogenic risk factors such as TG and HDLc not altered significantly or show no changes, AIP is a useful diagnostic tool for possible replacement [7]. Again, findings have favourably proved that AIP is highly important in predicting CVD risk and for effective therapeutic monitoring [6].
In 2012, Ghuge and Zine reported that AIP was increased in people at higher risk of CAD thus a diagnostic alternative to assess the effect of risk factors related to CVD [9]. Currently, AIP is gradually receiving attention from some practitioners as a vital prognosticator or predictor of developing atherosclerosis in metabolic diseases. The suggestion of AIP values that fall within -0.3 to 0.1 are associated with low, while 0.1 to 0.24 and above 0.24 are associated with medium and high cardiovascular risk, respectively [10].
The fourth lipid ratio known as Atherogenic Coefficient (AC), estimated as {(non-HDLc)/HDL-C} and sometimes as {(TCHDLc)/ HDL-C}, represents total sums of cholesterol found in VLDLc, IDLc and LDLc lipoprotein fractions in relation to the protective value of HDLc fraction. This therefore mirrors the atherogenic tendency that entire lipoprotein fractions are bound to generate. Once more, in 2011, Hermans and colleagues reported that non-HDLc mimics the role of Apo-B in determining atherogenicity level as well as lipoprotein burden and consequently termed a valid surrogate of Apo-B apolipoprotein [11].
On the other hand, traditional lipid parameters relating dyslipidemia with resultant CAD have been repeatedly reported, especially in HIV management using highly active antiretroviral therapy (HAART). The commonly estimated of such parameters are TC, TG, LDLc, and HDLc respectively. Dyslipidemia has been consistently recognized in HIV-patients receiving treatment with HAART as a notable risk for developing CAD often with elevated levels of TC, TG, and LDLc, reduced HDLc and typically hypertriglyceridemia in most severe cases [1,12].
Several important trials have stressed on the role of raised LDLc and reduced HDLc in the pathogenesis of atherosclerosis [12]. Also, other but prospective meta-analysis studies pointed it out that high TGs are correspondingly an independent risk factor for developing CHD [14, 15].
However, there are scenarios where the usefulness of traditional risk factors is limited. Bhardwaj and co-workers (2013) [2] reported that calculating certain emerging lipid ratios using these common lipid parameters, particularly in such circumstances where LDLc levels are under the target range, brings about possibility of raising the chance of identifying at-risk individuals that could be missed if ordinary lipid profile is employed. In this same study, it was noted that no significant difference in TC and LDLc levels was observed between the two studied groups, conversely, further estimation using calculated ratio based on these reported parameters later uncovered a significant difference between the groups. This, therefore, suggests that application of lipid ratios provides further utility over ordinary individual lipid parameters [2]. In addition, studies have indicated that in situations where other common predictors of atherogenic risk such as TG and HDLc appear normal, further estimation with AIP indicates that this lipid ratio is a more sensitive tool for diagnosis [7].
Furthermore, considering the running cost and operationalization for some important CAD plasma markers such as apolipoproteins (apo-CIII, apo-B100 etc) in resource-limited centers, these lipid ratios can add significant value to the assessment of CAD risk without headache [2, 16].
Finally, applications of these lipids ratios are becoming more recognized on daily basis, however, up till present moment very few studies have investigated their benefits in the management of HIV, whereas there is accumulating evidence of associated risk of developing CVD using ART in the Southwest Nigeria. Also, the use of more sensitive diagnostic markers such as apolipoproteins is rarely in routine operation in most treatment centers in the developing countries due to financial implications, for this reason, obtainability of simple, more sensitive and specific, and at the same time of no-cost lipid ratios as alternatives (or adjuncts) and better predictors of CVD risk will contribute immensely to this assessment.
Materials and Methods
The recruitment was conducted at the Institute of Human Virology of Nigeria (IHVN) clinic center and the analyses were carried out at the chemical pathology laboratory, both at Ladoke Akintola University of Technology Teaching Hospital, LAUTECH, Osogbo located in south-west of Nigeria.
Eighty (80) voluntary participants with age range of 18-55 years were recruited for the study. These include;
(i) Fourty HIV-positive patients (n= 40) on HAART treatment for three months and above,
(ii) Fourty HIV-positive patient but treatment naïve (n=40).
The participants comprise both sexes and ages within the same bracket, all living in the same geographical area. The study was initiated after the proposal had been reviewed and successfully approved by LAUTECH Ethical Committee with the help of the laboratory scientist in-charge.
Based on their clinical history and clinical records, subjects with the following manifestations were excluded;
DM (Diabetes Mellitus), hypertensions, hepatitis, jaundice, hypothyroidism, and nephrotic syndrome. Similarly, participants placed on contraceptives and other corticosteroid therapies were equally excluded.
Following an overnight fast, ten milliliters (10mls) of venous blood was collected from the cubital vein using a sterile needle and syringe into EDTA bottle. The blood was centrifuged and the plasma was then separated after 10 minutes at 4000 rpm. The plasma was kept in plain bottle and stored at -20°C before the time of analyses.
The plasma concentrations of TC, TG, and HDLc were estimated using spectrophotometry method.
Plasma TC was estimated by an enzymatic method [17]. Cholesterol level was determined following an enzymatic hydrolysis and oxidation processes. The mixture was incubated for ten (10) minutes at room temperature, and then the absorbance was finally read at wavelength of 500nm.
\
Plasma TG concentration was estimated by an enzymatic method [18]. TG level was determined following enzymatic hydrolysis process with lipase enzymes. The mixture was incubated for 10 minutes at room temperature following thorough mixing, and then the absorbance was finally read at wavelength of 500nm.
HDLc was estimated by precipitated method [19]. LDLc, VLDLc and chylomicrons were precipitated by centrifugation for 10 minutes in the presence of phosphotungstic acid and magnesium chloride, at 4000 rpm. The top supernatant was extracted immediately while the cholesterol concentration was determined. Here, centrifugation was left with only the HDLc in the supernatant. The HDLc fraction in the supernatant was removed as sample containing HDLc fraction which was in turn estimated by cholesterol assay method.
LDL-c: After the estimation of TC, TG and HDLc as stated above, LDLc was calculated using Friedewald formula as follows;
LDLc = Total cholesterol – (TG/5 +HDL).
This was scientifically applicable since the combined concentration of both cholesterol and TG was less than 400mg/dl [20].
Non HDLc: Non HDL cholesterol is estimated as total cholesterol minus HDLc.
Mathematically, it is estimated as;
Non HDLc = TC-HDLc.
Castelli’s Risk Index (CIR): Castelli’s Risk Index is based on three important lipid profile parameters i.e. TC, LDLc and HDLc and it is categorized into two; CIR-1 and CIR-11 [21].
CRI-1: CRI-I is established on the ratio of two important parameters of lipid profile, TC and HDLc. Mathematically, it is estimated as;
CRI-I = TC/HDLc ratio
CRI-11: CRI-II is calculated as the ratio of LDLc to HDLc. Mathematically, it is estimated as;
CRI-II = LDLc/HDLc ratio
Atherogenic Index of Plasma (AIP) = Atherogenic Index of Plasma was proposed by Dobiasova and Frohlich in 2001. AIP is a logarithmically transformed molar ratio of TG to HDLc [6].
AIP= Log10 (TG/HDLc) ratio.
Atherogenic Coefficient (AC) = Atherogenic coefficient is an indirect measure of cholesterol in VLDLc, IDLc, and LDLc lipoprotein fractions in reference with HDLc fraction. Mathematically, it is expressed as;
AC= {(TC- HDLc)/HDLc} or {(Non-HDLc)/HDLc} ratio
CD 4+ T-cell count was determined using Partec flow cytometer (TUV Rheinland) by method of flow cytomery. Flow cytometry was a very useful method of estimating CD4+ cells through which cell or micro particles in liquid suspension were differentiated and determined according to their cell sizes, fluorescence emission as well as their internal structures [22].
The study data were statistically analyzed using the Statistical Package for Social Science version program (SPSS programversion 10.0 - SPSS Inc., Chicago, IL. USA). The data were expressed as means (± SEM) using student’s t-test and Analysis of Variance (ANOVA). Means were also analyzed using Post Hoc test to compare the significant variables. Pearson’s correlation coefficient (r) was used to determine the association between and across means of the variables. Results were regarded as significant at P<0.05.
Results and Interpretation
A total number of 80 patients infected with HIV/AIDS disease met overall inclusion criteria and were randomly selected into two groups; treatment-naïve group (n=40) and another age and sex-matched group already on steady treatment for not less than three months (treatment group, n=40), all living within the same geographical area. The need for matching a treatment-naïve group was necessary to assess the effect of the drugs on both lipid profile and atherogenic indices before further evaluations.
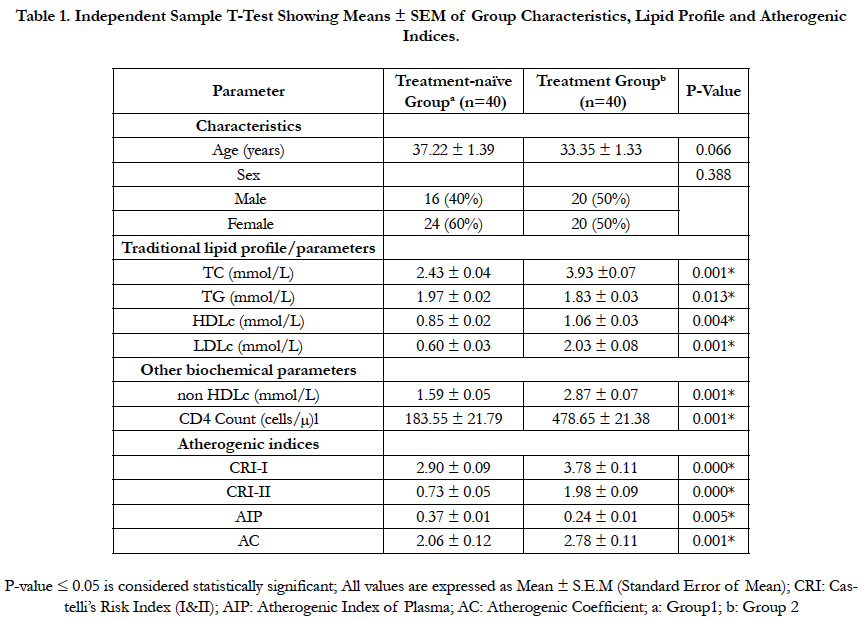
Data illustrated in table (1) show the effects of ART in matching group of treatment-naïve with another group of HIV-patients on ART treatment. There were significant differences (P ≤ 0.05) in the mean values of traditional lipid parameters (TC, TG, LDLc and HDLc), non HDL cholesterol, CD4 cell count and atherogenic indices (CRI-1 and II, AIP and AC) when comparisons between the two groups were carried out. In other words, there were significant increases (P≤0.05) in all these estimated parameters except the plasma triglyceride (TG) level and atherogenic index of plasma (AIP) where significant reductions (P ≤ 0.05) were observed.
Table 1. Independent Sample T-Test Showing Means ± SEM of Group Characteristics, Lipid Profile and Atherogenic Indices.
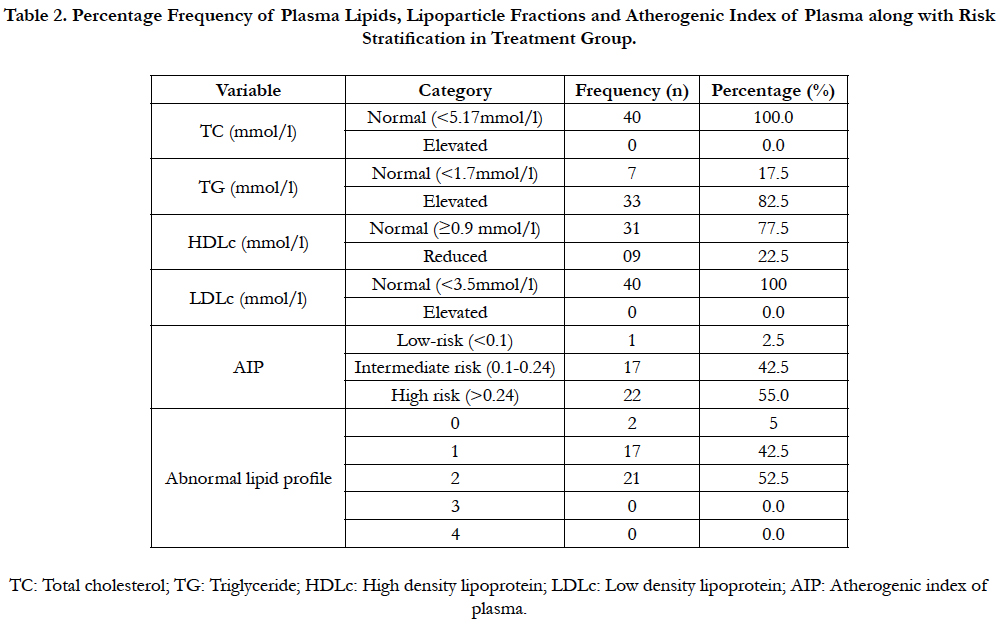
Table (2) shows the results of percentage frequency of referenced lipid profile along with risk stratifications using AIP and abnormal lipid profile in ART-experienced patients infected with HIV/ AIDS.
Reflecting the interpretations from routine clinical assessment and perspectives, plasma total cholesterol (TC) and low density lipoprotein cholesterol fraction (LDLc) were all normal and less than 5.17mmol/l and 3.5mmol/l respectively in the entire treatment group {n= 40; 100%}.
However, plasma TG level indicated that only 17.5% of this group (n=7) showed normal value less than 1.7mmol/l and majority of about 82.5% (n=33) of the group showed elevated value, while plasma high density lipoprotein cholesterol fraction (HDLc) were normal with ≥ 0.9 mmol/l value in 31 (77.5%) of the group. Surprisingly, stratification of this group of ARTexperienced patients that accorded predominantly normal values of lipid/lipoprotein levels under traditional lipid profile showed various risk strata using AIP.
Table 2. Percentage Frequency of Plasma Lipids, Lipoparticle Fractions and Atherogenic Index of Plasma along with Risk Stratification in Treatment Group.
AIP in predicting cardiovascular risk revealed that only 2.5% (n=1) proportion of the group was within low risk (<0.1), 42.5% (n=17) were in intermediate risk level (0.1-0.24) while majority in the group, 55% (n=22) posed with high risk (>0.24) respectively. Again, the group was further stratified into number of abnormal lipid/lipoprotein present. Patients proportion with all lipid/lipoprotein normal, that is, “0” abnormal lipid constituted 5% (n=2), the proportion with “1” abnormal lipid/ lipoprotein constituted 42.5% (n=17) while the proportion with “2” abnormal lipid/lipoprotein was 52.5% (n=21) respectively. In addition, none of the patients consisted of abnormal lipid/ lipoprotein values beyond “2” of the four lipid profile parameters.
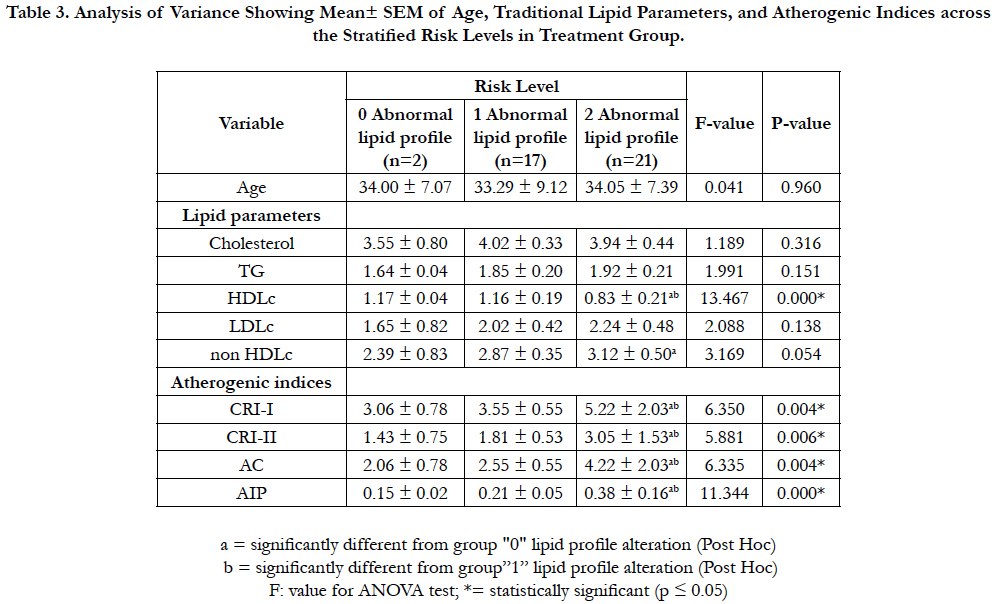
Table (3) illustrates analysis of variance for the mean (±SEM) values of age, traditional lipid parameters and atherogenic indices across the stratified risk levels based on burden of abnormal lipid profile in ART-experienced group infected with HIV/AIDS.
There was no significant difference (P>0.05) both across and between the groups when the ages of patient subgroups in all the three categories of abnormal lipid profile were compared.
Table 3. Analysis of Variance Showing Mean± SEM of Age, Traditional Lipid Parameters, and Atherogenic Indices across the Stratified Risk Levels in Treatment Group.
Considering traditional lipid parameters, only plasma HDLc level showed significant changes (P<0.05) both across and between the groups of the three categories of abnormal lipid profile. Similarly, there was significant difference (P<0.05) in plasma non HDLc level between subgroup of “2” abnormal lipid profile against “0” abnormal lipid profile, although no significant difference (P>0.05) was observed across the three categories of abnormal lipid profile.
On the contrary, all indices of atherogenicity; Castelli’s risk index I & II (CRI-1 & CRI-II), atherogenic coefficient (AC) and AIP showed significant differences (P<0.05) both across and between the subgroups of abnormal lipid profile.
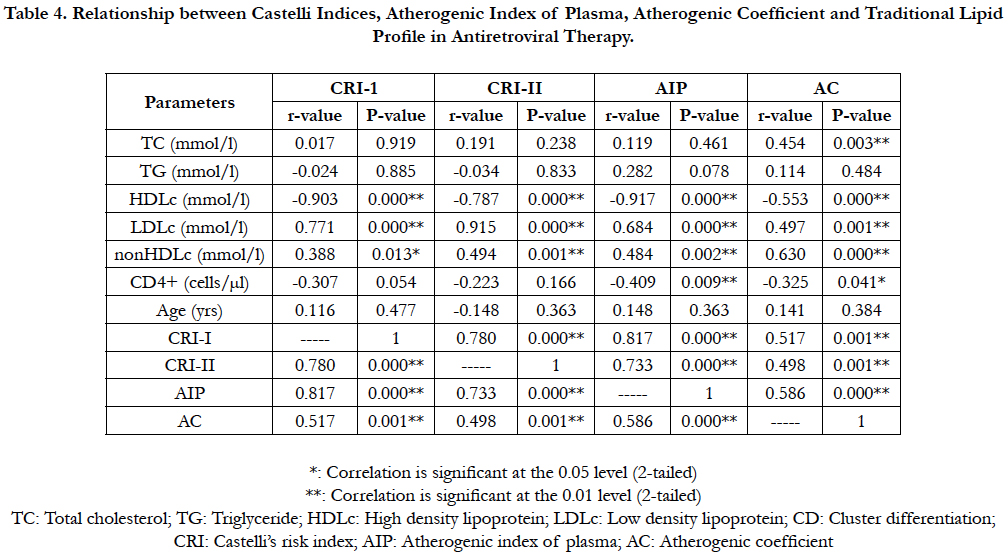
Table (4) shows relationship between traditional lipid parameters and atherogenic indices in a group of ART-experienced patients infected with HIV/AIDS.
Table 4. Relationship between Castelli Indices, Atherogenic Index of Plasma, Atherogenic Coefficient and Traditional Lipid Profile in Antiretroviral Therapy.
Plasma TC level showed significant positive association with AC (P<0.01; r=0.454) in ART-treatment group. Meanwhile, no significant association (P>0.05) was observed between TG and any index of atherogenicity. However, plasma HDLc level showed significant negative associations with CRI-I (P<0.01; r = -0.903), CRI-II (P<0.01; r = -0.787), AIP (P<0.01; r = -0.917) and AC (P<0.01; r = -0.553) correspondingly. Conversely, plasma LDLc level showed significant positive associations with CRI-I (P<0.01; r=0.771), CRI-II (P< 0.01; r=0.915), AIP (P<0.01; r=0.684) and AC (P<0.01; r=0.497) respectively. Similarly, plasma non HDLc level showed significant positive association with CRI-I (P<0.05; r=0.388), CRI-II (P<0.001; r=0.494), AIP (P<0.01; r=0.484) and AC (P<0.01; r=0.630), while CD4 cell counts showed significant negative associations with AIP (P<0.01; r = -0.409) and AC (P<0.05; r = -0.325) respectively. Individually, all indices of atherogenicity show significant positive associations (P<0.01) with one another; CRI-I vs CRI-II (P<0.01; r=0.780), CRI-1 vs AIP (P<0.01; r=0.817), CRI-I vs AC (P<0.01; r=0.517), CRI-II vs AIP (P<0.01; r=0.733), CRI-II vs AC (P<0.01; r=0.498) and AIP vs AC (P<0.01; r=0.586) respectively.
Discussion
Dyslipidemia has been recognized as an important risk factor associated with coronary artery disease (CAD) [23]. A number of lipid parameters have been employed in predicting risk of coronary atherosclerosis and cardiovascular diseases (CVD). These chiefly comprise plasma concentrations of lipids and lipoproteins, while occasionally involves plasma apolipoproteins. Of more recent, the use of lipid ratios as risk indices involving the lipids/lipoproteins has been noteworthy [1, 24, 25]. Many reports show the relevance of evolving lipid ratios above traditional lipid parameters [2, 7].
In our study, we assessed atherogenic status of HIV-individuals at risk of CVD during the treatment, by first comparing a group of HIV-treated patients against another without prior exposure to treatment, indicative of drug effect. The outcomes greatly indicated lipid alteration reflected on both lipid ratios and traditional lipid profile. However, when classified by plasma total cholesterol (TC), LDL-cholesterol (LDLc), and non HDLcholesterol (non HDLc) levels along with Castelli’s risk indexes I and II (CRI-1 & II) and atherogenic coefficient (AC), CVD risk seemed to be more likely in HIV-treated patients. Whereas, when considering HDL-cholesterol (HDLc) and triglyceride (TG) levels in the company of atherogenic index of plasma (AIP), there seemed to be high risk of CVD in antiretroviral therapy (ART) treatment-naïve group of the patients.
Meanwhile, attention has been drawn to different lipid abnormalities in both treatment-naïve HIV patients and ARTexperienced [1] and that corroborates our study. More so, our present study is also in accordance with earlier study, where high risk factors such as significant increases in TC and LDLc levels along with increased HDLc level (cardinal marker of improved dyslipidemic risk) were reported in ART treatment [26].
On the other hand, in ART-naïve patients, hypertriglyceridemia, reduced TC and LDL levels have been observed [12]. Rose and colleagues (2008) also reported reduced HDLc level, indicating an increase in the activity of the cholesterol ester transfer protein (CETP) which acts in transfer of cholesterol from HDLc to apo‑B lipoproteins, hence consequential reduction in HDLc [27]. More interestingly, a study conducted on newly diagnosed HIVpositive individuals revealed that ART resulted in an improved dyslipidemic profile that was earlier observed before initiating treatment [28].
On the contrary, in one closely related study from the same study location with our study, Oduola et al., (2009) [29] followed a group of HIV-infected urban dwellers, and the lipid profile was monitored for 15 months in categorized groups of HIV-patients on ART and treatment-naive who were hitherto asymptomatic. When finally matched with another set of apparently healthy HIV-negative participants, TG, TC, HDLc, and LDLc levels in the ART-treated patients were in significantly different, while the TC and HDLc levels significantly reduced in the HIV-treatment naïve against ART-treated group [29].
Nevertheless, consistent with our findings, some studies reported significant increases in TC, HDLc, and non HDLc levels after ART treatment of HIV positive patients [30, 31]. Although, our study expanded beyond the scope of the existing literature with annexation of well-validated risk predictors such as CRI-I & II indexes, AC and AIP. It is bit difficult to make succinct comparison due to different study design. However, Kalkinami et al., (2012) [32] reported CRI index in a separate but retrospective cohort study on HIV-monoinfection group of their study, where apart from significant increases in TC, HDLc and non HDLc levels, CRI index also showed elevation but not statistically significant.
On these conflicting reports, we were therefore obliged to further assessment of the ART-experienced individuals in our study to understand interrelationship between the common lipid profile parameters and the evolving lipid ratios on ART therapy, succinctly. In this regard, advanced stratifications of the ART-experience group clearly refined individual risk levels using AIP index and burden of abnormal lipid and lipoproteins. Interestingly, we did not observe significant alterations in TC, TG, LDLc and non HDLc levels in this group considering the risk factors by means of abnormal lipid profile, although HDLc showed significant reduction, whereas the CRI-indexes, AIP and AC all indicated significant increased risk levels across the group strata.
The reduction in HDLc levels has been linked with increased rate of catabolism. Again, alterations in lipid metabolism involving reduced HDLc has been associated with CAD [33], while reduced level of HDLc is described an independent risk factor for enhanced susceptibility to CAD [34]. Our study outcome is very typical of dyslipidemia observed in angiographically confirmed patients in Indians where the absolute values of lipid profile parameters such as TC, LDLc, HDLc and nonHDLc levels were not markedly deranged but showed significant elevations in CRI-I, CRI-II, AIP and AC indices [2] that have been repeatedly reported as probable predictors of CVD in literature [2, 8].
Besides, studies have revealed that in circumstances where other atherogenic risk parameters such as TG seem normal, AIP may possibly serve as the diagnostic alternative [7] while da Luz and coworkers established that a high ratio of TG/HDL strongly indicated correlation with coronary disease than lipid variables [35]. Furthermore, studies have indicated the role of AIP in predicting CVD risk and effectiveness of therapy [8]. In our study, we could not observe significant changes in TC, TG, LDLc and non HDLc levels in our ART-experience group whereas, AIP along with other lipid ratios (CRI-I, CRI-II, and AC indices) showed significant increases. This clearly suggests the relevance of the risk predictors over individual lipid parameters.
In a related study, studies have revealed the association between TC/HDLc ratio (here known as CRI-I) and coronary plaques development [36], this is supported by the Canadian study group that TC/HDLc is a more sensitive, specific and CVD risktargeting index than ordinary TC [37].
\
AIP correlated well with HDLc, LDLc and non HDLc fractions in our study in the same way with CRI-I, CRI-II, and AC indices. According to Grover’s model, either the ratio of LDLc/HDLc or TC/HDLc (that is, CRI-I & II) is the best lipid related predictive risk calculators of future cardiovascular events [38].
Similarly, Dobiásová and Frohlich demonstrated correlations of AIP with HDL size and LDL particles and with the fractional esterification rate of cholesterol catalyzed by lecithincholesterolacyl transferase (LCAT) in plasma. By indication, index ratio perfectly reveals the presence of atherogenic small LDLc and HDLc particles, and therefore an accepted sensitive predictor of atherosclerotic cardiovascular risk [6]. Likewise, CRI-I and II are predictive of AIP [24] as well as AC [2] and therefore are likely to be related in their ability to pinpoint patients with risk of CVD.
Conclusion
The findings in this study were in conformity with mammoth of evidence showing that calculated lipid ratios- Castelli’s risk indexes, atherogenic index of plasma, and atherogenic coefficient could serve as more sensitive risk predictors for atherosclerotic cardiovascular disease. Many of ART-experienced patients might be wrongly captured within low risk status using traditional lipid profile in desirable range consequently escape right medical decision. Meanwhile, sudden coronary death is the first manifestation of atherosclerotic disease in large proportion of the cases (WHO, 1990); further estimation of lipid ratios beyond ordinary lipid profile could avert sudden death in HIV-patients well receiving treatment.
Limitations
Our study was not without limitations, one of which was the sample size. Further studies will require large sample size and better still of multicenter design to better validate this conclusion. More so, since information concerning certain other potential risk factors for cardiovascular dieases such as- diet, physical activity, classes of drug, genetic factors and other demographic and anthropometric variables was not collected during the recruitment for our study, this may limit the validity of our outcomes to populations similar to our study alone.
Acknowledgments
The authors are grateful to Scientists Akande ‘Seun, Laluwoye Opeyemi and other staff members who have contributed in one way or the other during the stages of recruitment and analyses, all from LAUTECH Teaching Hospital, Osogbo (as at the time of this study).
References
- Kamoru AA, Japhet OM, Adetunji AD, Akinlawon AA, Musa MA, Onifade AA, et al. CD4+ Cell Count, Lipid And Lipoprotein Levels In Hiv Patients On Drug Treatment. Int J AIDS Res. 2017 Apr 5;4(1):140-146.
- Bhardwaj S, Bhattacharjee J, Bhatnagar MK, Tyagi S. Atherogenic index of plasma, castelli risk index and atherogenic coefficient new parameters in assessing cardiovascular risk. Int J Pharm Biol Sci. 2013;3(3):359-364.
- Millán J, Pintó X, Muñoz A, Zúñiga M, Rubiés-Prat J, Pallardo LF, et al., Lipoprotein ratios: Physiological significance and clinical usefulness in cardiovascular prevention. Vasc Health Risk Manag. 2009;5:757–765. PubMed PMID: 19774217.
- Kannel WB. Risk stratification of dyslipidemia: insightsfrom the Framingham Study. Curr Med Chem Cardiovasc Heamatol Agents. 2005 Jul;3(3):187-193. PubMed PMID: 15974883.
- Manninen V, Tenkanen L, Koskinen P, Huttunen JK, Mantarri M, Heinomen OP, et al. Joint effects of serum triglyceride and LDL cholesterol and HDL concentrations on coronary heart disease risk in the Helsinki Heart Study. Implications for treatment. Circulation. 1992 Jan;85(1):37-45. PubMed PMID: 1728471.
- Dobiasova M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FERHDL). Clin Biochem. 2001 Oct;34(7):583–588. PubMed PMID: 11738396.
- Nwagha UI, Ikekpeazu EJ, Ejezie FE, Neboh EE, Maduka IC. Atherogenic index of plasma as useful predictor of cardiovascular risk among postmenopausal women in Enugu, Nigeria. Afri Health Sci. 2010 Sep;10(3):248–252. PubMed PMID: 21327136.
- Dobiášová M, Frohlich J, Šedová M, Cheung MC, Brown BG. Cholesterol esterification and atherogenic index of plasma correlate with lipoprotein size and findings on coronary angiography. J Lipid Res. 2011 Mar;52(3):566–571. Pubmed Central PMCID: PMC3035693.
- Ghuge GD, Zine R. Atherogenic index of plasma in myocardial infarction in rural population of Marathwada region. J Evol Med Dent Sci. 2012 Sep;1(3):237-240.
- Dobiasova M. AIP-atherogenic index of plasma as a significant predictor of cardiovascular risk: from research to practice. Vnitr Lek. 2006 Jan;52(1):64-71. PubMed PMID: 16526201.
- Hermans MP, Sacks FM, Ahn SA, Rousseau MF. Non-HDL-cholesterol as valid surrogate to apolipoprotein B100 measurement in diabetes: Discriminant Ratio and unbiased equivalence. Cardiovasc Diabetol. 2011; 10:20. PubMed Central PMCID: PMC3056766.
- El‑Sadr WM, Mullin CM, Carr A, Gibert C, Rappoport C, Visnegarwala F, et al. Effects of HIV disease on lipid, glucose and insulin levels: Results from a large antiretroviral‑naive cohort. HIV Med. 2005 Mar;6(2):114‑121. PubMed PMID: 15807717.
- National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002 Dec 17;106(25): 3143–3421. PubMed PMID:12485.
- Patel A, Barzi F, Jamrozik K, Lam TH, Ueshima H, Whitlock G, et al. Serum triglycerides as a risk factor for cardiovascular diseases in the Asia‑Pacific region. Circulation. 2004 Oct 26;110(17):2678‑2686. PubMed PMID: 15492305.
- Sarwar N, Danesh J, Eiriksdottir G, Sigurdsson G, Wareham N, Bingham S, et al., Triglycerides and the risk of coronary heart disease: 10,158 incident cases among 262,525 participants in 29 Western prospective studies. Circulation. 2007 Jan 30;115(4):450‑458. PubMed PMID: 17190864.
- Gotto A, Whitney E, Stein EA, Shapiro DR, Clearfield M, Weis S, et al. Relation between baseline and on-treatment lipid parameters and first acute major coronary event in the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS). Circulation. 2000 Feb 8;101(5):477–484. PubMed PMID: 10662743.
- Trinder P, Barham D. Estimation of LDL-cholesterol in plasma. Clin Chem. 1972;78: 19-22.
- Buccolo G, David H. Quantitative determination of serum triglyceride by the use of enzymes. Clin Chem. 1973May;19(5):476-482. PubMed PMID: 4703655.
- Benzie A. An evaluation of a routine assay of serum High-density lipoprotein cholesterol using dextran sulphate and magnesium chloride. Med lab Sci. 1979 Jul;36(3):289-291. PubMed PMID: 229379.
- Friedwald WT, Levy IR, Friedrickson DS. Estimation of the concentration of low density lipoprotein cholesterol in plasma without use of the preparative ultracentrifugation. Clin Chem. 1972 Jun;18(6):499-502. PubMed PMID: 4337382.
- Castelli WP, Abbott RD, McNamara PM. Summary estimates of cholesterol used to predict coronary heart disease. Circulation. 1983 Apr;67(4):730- 734. PubMed PMID: 6825228.
- Greeve B. A new No-lyse, No-Wash Flow-Cytometric Method for the determination of CD4 T Cells in Blood Samples. Transfus Med Hemother. 2003;30:8-13.
- Yusuf S, Hawken S, Ounpuu S, Dans T, Avezum A, Lanas F, et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet. 2004 Sep;364(9438):937-952. PubMed PMID: 15364185.
- Onyedum CC, Young EE, Iroezindu MO, Chukwuka CJ, Nwagha UI. Atherogenic index of plasma in highly active antiretroviral therapynaïve patients with human immunodeficiency virus infection in Southeast Nigeria. Indian J Endocr Metab. 2014 Sep;18(5):631-636. PubMed PMID: 25285278.
- Kim T, Park AY, Baek Y, Cha S. Genome-wide association study reveals four loci for lipid ratios in Korean population and the constitutional Subgroup. PLoS One. 2017;12(1):e0168137. PubMed Central PMCID: PMC5207643.
- Zephy D. Lipid profile among Art treated and untreated patients in HIV positive cases. Arch Med. 2015;8:2.
- Rose H, Hoy J, Woolley I, Tchoua U, Bukrinsky M, Dart A, et al. HIV infection and high density lipoprotein metabolism. Atherosclerosis. 2008 Jul;199(1):79-86. pubMed PMID: 18054941.
- Fourie CMT, Van Rooyen JM, Schutte AE. HIV infection and cardiovascular risk in black South Africans. Cardiovasc J Afr. 2011 Jun;22(3):117–119. PubMed Central PMCID: PMC3734753.
- Oduola T, Akinbolade AA, Oladokun LO, Adeosun OG, Bello IS, Ipadeola TI. Lipid profiles in people living with HIV/AIDS on ARV therapy in an urban area of Osun State, Nigeria. World J Med Sci. 2009 Jan;4:18-21.
- Levy AR, McCandless L, Harrigan PR, Hogg R, Bondy G, Iloeje UH, et al. Changes in lipids over twelve months after initiating protease inhibitor therapy among persons treated for HIV/AIDS. Lipids Health Dis. 2005 Feb 10;4:4. PubMed PMID: 15705191.
- Asztalos BF, Schaefer EJ, Horvath KV, Cox CE, Skinner S, Gerrior J, et al. Protease inhibitor- based HAART, HDL, and CHD-risk in HIV-infected patients. Atherosclerosis. 2006 Jan;184(1):72–77.
- Kakinami L, Adams JM, Block RC, Cohn SE, Maliakkal B, Fisher SG. Short Communication. Risk of Elevated Total Cholesterol/High-Density Lipoprotein Cholesterol Ratio After Antiretroviral Therapy in HIV/Hepatitis C Virus Patients. AIDS Res Hum Retrov. 22012 Dec 1;28(12):1552-1556.
- Brunzell JD. Hypertriglyceridemia. N Engl J Med. 2007;357:1009-1017.
- Von Eckardstein A, Nofer JR, Assmann G. High density lipoproteins and arteriosclerosis: Role of cholesterol efflux and reverse cholesterol transport. Arterioscler Thromb Vasc Biol. 2001 Jan;21(1):13–27. PubMed PMID: 11145929.
- da Luz PL, Favarato D, Faria‑Neto JR, Lemos P, Chagas AC. High ratio of triglycerides to HDL‑cholesterol predicts extensive coronary disease. Clinics (Sao Paulo). 2008 Aug;63(4):427‑432. PubMed Central PMCID: PMC2664115.
- Nair D, Carrigan TP, Curtin RJ, Popovic ZB, Kuzmiak S, Schoenhagen P, et al. Association of total cholesterol/ high-density lipoprotein cholesterol ratio with proximal coronary atherosclerosis detected by multislice computed tomography. Prev Cardiol. 2009;12(1):19-26. PubMed PMID: 19301687.
- Genest J, Frohlich J, Fodor G, McPherson R. The Working Group on Hypercholesterolemia and Other Dyslipidemias. Recommendations for the management of dyslipidemia and the prevention of cardiovascular disease: 2003 update. CMAJ. 2003 Oct 28;169(9):921–924. PubMed PMID: 14581310.
- Grover SA, Levington C, Paquet S. Identifying adults at low risk for significant hyperlipidemia: a validated clinical index. J Clin Epidemiol. 1999 Jan;52(1):49–55. PubMed PMID: 9973073.