The Correlation between the Plasma Level of NT-Pro BNP and Left Ventricular Diastolic Dysfunction
Osama Sanad Arafa1, Mohamed Ahmed Hamouda2, Mohamed Abd El-Kader Elian3, Shaimaa Ahmed Mostafa3*, Mahmoud Said Mahmoud4
1 Professor of Cardiology, Benha University, Faculty of Medicine, Benha, Egypt.
2 Assistant Professor of Cardiology, Benha University, Faculty of Medicine, Benha, Egypt.
3 Lecturer of Cardiology, Benha University, Faculty of Medicine, Benha, Egypt.
4 MSC of Cardiology, Benha University, Faculty of Medicine, Benha, Egypt.
*Corresponding Author
Shaimaa Ahmed Mostafa,
Lecturer of Cardiology, Benha University,
Faculty of Medicine, Benha,
Egypt.
Tel: 01229122843
E-mail: shaimaamustafa2011@gmail.com
Article Type: Research Article
Received: September 19, 2015; Accepted: December 08, 2015; Published: December 11, 2015
Citation: Shaimaa Ahmed Mostafa, et al., (2015) The Correlation between the Plasma Level of NT-Pro BNP and Left Ventricular Diastolic Dysfunction. Int J Cardiol Res. 02(5), 41-46. doi: dx.doi.org/10.19070/2470-4563-1500010.
Copyright: Shaimaa Ahmed Mostafa© 2015. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: To study the correlation between plasma level of N-terminal pro-BNP and left ventricular diastolic dysfunction in asymptomatic patients.
Methods: The study included 35 patients divided into control group: included 10 patients with normal systolic and diastolic function and patient group: included 25 patients with normal systolic and asymptomatic diastolic dysfunction. The patients were evaluated by conventional echocardiography (diameters, systolic and diastolic function) and tissue Doppler (early diastolic velocity) then E/e` was measured. Serum pro BNP was measured and correlated with demographic and echocardiographic parameters and its sensitivity, specificity were evaluated.
Results: Both groups were of comparable age, gender, BMI, risk factors and systolic function. By Doppler echocardiography there was no difference in E velocity between two groups (0.64+0.18 m/sec Vs 0.67+0.14 P = 0.302), but by tissue Doppler there was significant difference in the mean value of e` velocity (m/sec) (0.14+0.06 Vs 0.06+0.01 P=0.001) and /eˋ (5.03+0.149 Vs 0.55+1.79 P= 0.001). There was significant difference between two groups according to serum NT pro BNP (ng/ml) was (59.1) in the control group Vs (154.1) in patient group (P = 0.001). There was significant correlation between serum NT pro BNP and E (P= 0.001) and E/e` (P = 0.001). The sensitivity of serum NT Pro BNP was 94.2, specificity was 88.1, positive predictive value was 90.6 and its accuracy was 86.2.
Conclusion: Serum level of NT pro BNP can be used as innovative method in predicting asymptomatic diastolic dysfunction and so prevent the progression to symptomatic diastolic heart failure.
2.Introduction
3.Aim
4.Methods
4.1 Group (1) (Control group)
4.2 Group (2) (Patient group)
4.3 Statistics
5.Result
5.1 Comparison between Control Group and Patient group according to (serum NT pro BNP)
5.2 Comparison between Control Group and Patient group according to (serum NT pro BNP)
6.Discussion
7.Study limitations
8.Conclusions
7.References
KeyWords
NT-Pro BNP; Asymptomatic Diastolic Dysfunction.
Introduction
For years, scientists and clinicians believed that systole was the cornerstone mechanism of heart functioning. As a result, all treatments for heart failure (HF) were directed at treating systolic dysfunction. But the heart is actually in diastole two-thirds of the time. In the 1980s, the medical community began to recognize that some patients with a normal ejection fraction were experiencing signs and symptoms of heart failure. Scientists evaluated these patients and found that even though systolic function was adequate, diastolic function was suboptimal. At this time, diastolic dysfunction was deemed to be clinically significant [1].
Today, patients with signs and symptoms of HF and diastolic dysfunction are diagnosed with heart failure with a normal ejection fraction (HFNEF), or diastolic HF. Perhaps as much as 50% of patients with heart failure have diastolic dysfunction and a normal ejection fraction [2].
Some data suggest that mortality rates among individuals with HFNEF are similar to those with HF and systolic dysfunction [3]. Bhatia, et al. found that among patients with heart failure, the one year mortality and hospitalization for HF didn’t differ among those with an EF > 50% vs. EF < 40% [3].
Interestingly, Owan, et al. observed that the prevalence of HFNEF has increased over the last 15 years and the rate of death related to this entity hasn’t decreased. This is in contrast to HF secondary to decreased EF, where a decrease in mortality over time was observed. This is likely related to evolving therapies for systolic heart failure, improved diagnosis and development of new therapies for HFNEF. Furthermore, even in the absence of clinical HF, diastolic dysfunction (DD) is associated with increased rates of future hospitalizations, development of HF and all-cause mortality [4].
Worsening stages of DD on echocardiography are associated with incremental risk of adverse outcomes including the development of clinical HF. Accurately diagnosing DD could possibly lead to improved treatments and may have substantial health care implications. Moreover, reproducible, widely applicable and prognostically meaningful approaches to defining DD are important for clinical trials where the use of complex methods to assess diastolic function may be difficult to standardize and implement [5].
In routine clinical practice Doppler echocardiography is the method of choice to diagnose DD. Numerous algorithms have been proposed, most based on transmitral Doppler patterns. However, transmitral Doppler derived indices of diastolic function are dependent on loading conditions and accurate measurements are operator dependent. Tissue Doppler imaging is a newer technique that can be used in combination with transmitral Doppler to determine the presence and severity of DD [5].
However, this assessment of DD is more complex and requires expert interpretation. Many parameters have been shown to be associated with DD, including echocardiographic measurements, various clinical characteristics, increased left atrial (LA) volume and elevated levels of B-type natriuretic peptide (BNP) and N-terminal (NT)-pro BNP. Identifying simple clinical and/ or biochemical and/or echocardiographic measurements that can reliably identify the presence and severity of DD is particularly important for patients with HF-PLEF [6].
Therefore, we aimed to determine the best set of clinical parameters, tissue Doppler echocardiography and N-terminal pro-BNP (NT pro BNP) that could accurately predict DD. If needed a simple set of such parameters could be shown to be strongly associated with DD on echocardiography, use of such parameters could circumvent the need for detailed, difficult and costly echocardiographic assessments (in situations where echo is not readily available) to determine the presence of prognostic important DD.
Aim
To study the correlation between plasma level of N-terminal pro-BNP (NT pro BNP) and left ventricular diastolic dysfunction as determined by tissue Doppler echocardiography in asymptomatic patients.
Methods
The study included 35 patients divided into two groups:
Included 10 patients with normal left ventricular systolic function (EF ≥ 50%) and normal left ventricular diastolic function by estimation of (E/e` < 8) by tissue Doppler echocardiography.
Group (2) (Patient group):
Included 25 Patients with normal left ventricular systolic function (EF ≥ 50%) and asymptomatic left ventricular diastolic dysfunction by estimation of (E/e` ≥ 8) by tissue Doppler echocardiography.
Exclusion criteria are:
• Poor quality echocardiographic study.
• Moderate to severe mitral and /or Aortic regurgitation.
• Prosthetic mitral valve.
• Renal impairment.
• Major arrhythmias including atrial fibrillation.
• Patients with symptomatic heart failure.
• Patients with acute coronary syndrome.
• Uncontrolled hypertension.
They were submitted to:
1. History taking including history of diabetes mellitus, atrial fibrillation, coronary heart disease, hypertension and history of underlying medical treatment including (ACE inhibitors, Beta blocker, Aspirin, statins and diuretic).
2. Complete cardiovascular examination including heart rate, systolic and diastolic blood pressure and thorough cardiac examination.
3. Laboratory tests:
A blood sample was obtained for:
• Serum urea and creatinine level, glycosylated hemoglobin (Hb A1C) was estimated for diabetic patients.
• Serum level of NT-ProBNP was estimated using Elecys pro-BNP sandwich immunoassay.
(NT-proBNP reference ranges were reported as < 125 pg/ml for individuals aged < 75 years and < 450 pg/ml for individuals aged ≥ 75 year).
4. Electrocardiography (ECG)
Resting 12 leads ECG was obtained at the time of examination.
5. Echocardiography:
a. Using GE vivid S5, the M-mode study of the LV was performed using the parasternal long axis view. The LV endsystolic dimension (LVDs) and end-diastolic (LVDd), interventricular septum thickness during diastole (IVSd) and systole (IVSs), posterior wall thickness (PWT) during diastole (LVPWd) and systole (LVPWs), ejection fraction (EF), Aortic root diameter (AOD) and Left atrial diameter (LAD) were measured.
b. Using these measurements, the LV mass (LVM) according to the Devereux formula: LVM=1.05× [PWT+IVST+LVDd) 3-(LVDd) 3]-13.6 (Lang et al., 2005). The LVM index (LVMI) was calculated as the ratio between LVM and body surface area (normal values, 110 g/m2 for women and 130 g/m2 for men). The severity of LV hypertrophy (LVH) was assessed by LVMI.
c. Pulsed-wave Doppler was done and the peak velocities of the E wave of the trans-mitral flow was estimated.
d. Tissue Doppler echocardiography measurement:The apical four-chamber view was used to obtain the TDI of the mitral annulus. A sample volume of the pulsed-wave Doppler was positioned at the lateral side of the mitral annulus and then the lateral signal of the mitral annular velocity was recorded. The peak early mitral annular velocity (é) was measured and E/é was calculated as E divided by é.
Statistics
Using Microsoft Excel 2003 and SPSS v19.0 for Microsoft Windows 7 the clinical and laboratory data were statistically analyzed to obtain descriptive statistics (mean, Standard deviation, range) and Analytical studies:
a. Comparisons between means using:
1. Unpaired student t-test for comparison between 2 groups with independent parametric data.
2. Mann – Whitney (U) test for comparison between 2 groups of independent non-parametric data.
b. Comparisons between qualitative variables using:-chi-square test (x²)
c. Correlation statistics using:-
1. Pearson correlation coefficient between parametric data.
2. Spearman correlation coefficient between non-parametric data.
3. Sensitivity: The sensitivity of a clinical test refers to the ability of the test to correctly identify those patients with the
disease.
4. Specificity: The specificity of a clinical test refers to the ability of the test to correctly identify those patients without the disease.
• Positive predictive value: probability that the disease is present when the test is positive (expressed as a percentage).
• Negative predictive value: probability that the disease is not present when the test is negative (expressed as a percentage).
Result
This study included 35 patients divided into 2 groups: Control group which included 10 patients with normal systolic and diastolic function and Patients group which included 25 patients with normal systolic and asymptomatic diastolic function as documented by tissue Doppler echocardiography based on the valueof (E/e` ≥ 8).
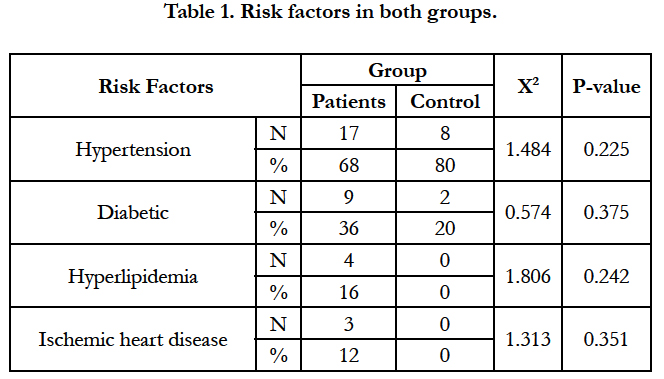
Both groups were of comparable age and gender; mean of age was (48.56) years in the control group vs (43.40) years in the patient group (P = 0.142) and as regard to gender, female percent was more in patient group while male percent was more in control group without significant difference (P = 0.225).
80% of the control group were hypertensive Vs 68% in the patient group (P = 0.225), diabetes mellitus was (20% Vs 36% P = 0.375), hyperlipidemia was (0 Vs 14, 16% P = 0.242). The mean value of BMI was (27.10) in control Vs (28.64) in the patient group (P = 0.412) and Ischemic heart disease was (0 Vs 3, 12% P = 0.351) Table 1.
There was no statistically significant difference between 2 groups as regard to the systolic function EF% was 69.4+7.54 in the control group Vs 63.8+4.38 in the patient group (P = 0.019). Also no significant difference between two groups according to LVM (gm) and LVMI (g/m²). The mean value of LVM was (156.1+39.7 Vs 161.1+50.8, P = 0.963). The mean value of LVMI was (71.8+15.5 Vs 79.8+20.8, P = 0.885).
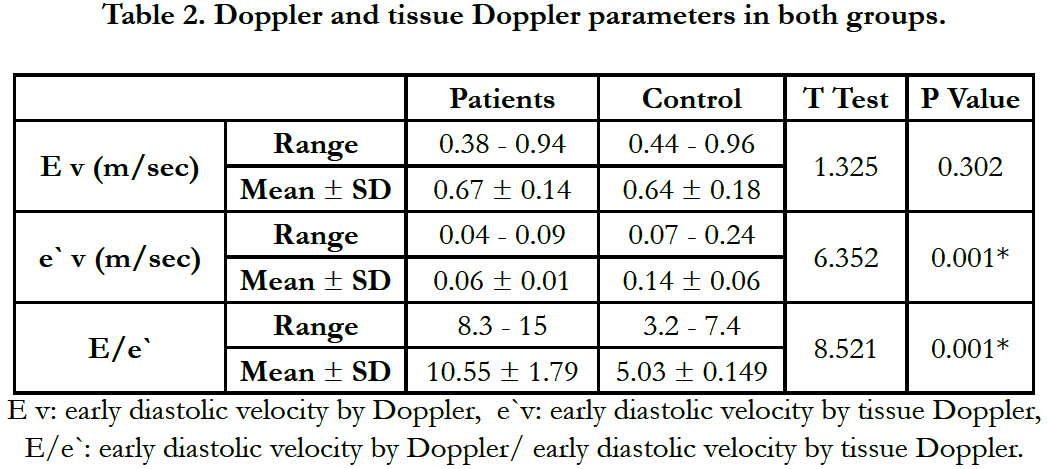
By Doppler echocardiography there was no difference in the E velocity between two groups the mean value of E velocity was (0.64 + 0.18m/sec) in the control group Vs (0.67 + 0.14) in the patient group (P = 0.302), but by tissue Doppler the difference was statistically significant, the mean value of e`velocity (m/sec) was (0.14 + 0.06 Vs 0.06 + 0.01P = 0.001). And also the difference of mean value of E/e` was significant (5.03 + 0.149 Vs 0.55 + 1.79 P = 0.001) Table 2.
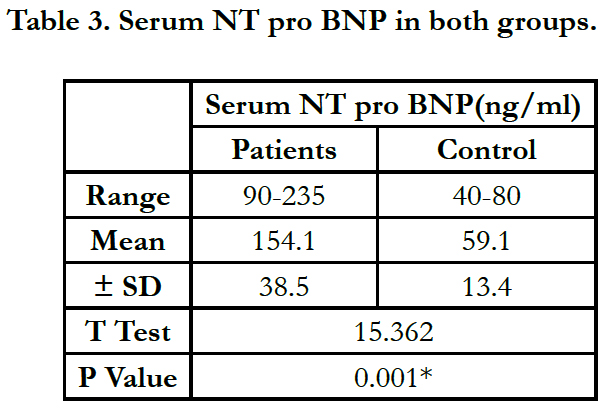
There was significant difference between two groups according to serum NT pro BNP. The mean value of serum NT pro BNP (ng/ ml) was (59.1) in the control group Vs (154.1) in patient group (P = 0.001) Table 3.
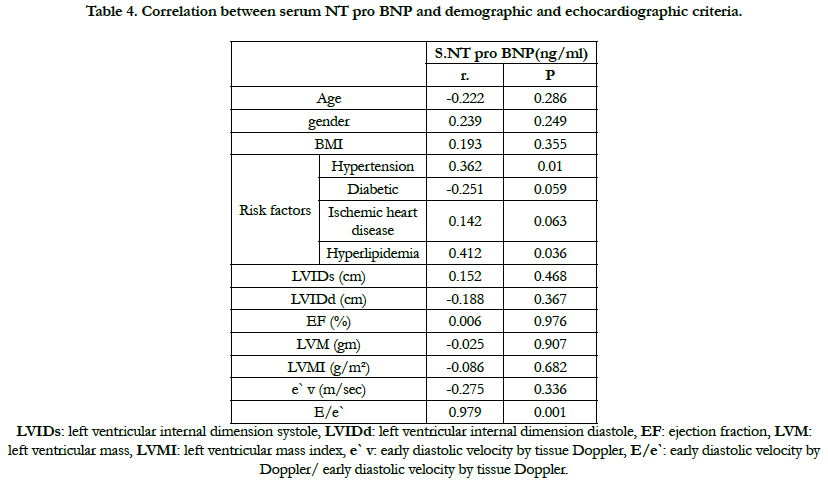
There was insignificant correlation between serum NT pro BNP and age (P=0.286, r=-0.222), gender (P=0.249, r=0.239), BMI (P=0.355, r=0.193), hypertension (P=0.010, r=0.362), diabetes (P=0.059, r=-0.251), hyperlipidemia (P=0.036, r=0.412), EF% (P=0.976, r=0.006), LVM (p=0.907, r=-0.025) but there was significant correlation between serum NT pro BNP and E velocity (P = 0.001, r=0.504) and E/e` (P = 0.001, r=0.979) Table 4.
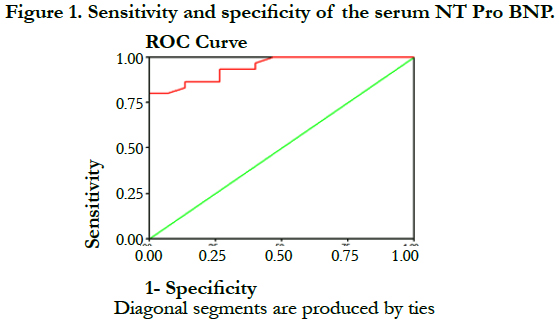
3.25ng/ml with sensitivity of serum NT Pro BNP 94.2, specificity was 88.1, positive predictive value was 90.6, negative predictive value was 28 and its accuracy was 86.2 Figure (1).
Discussion
Heart failure is a growing worldwide epidemic and is associated with substantial morbidity and mortality. Left ventricular systolic function was often considered to be the main abnormality in heart failure. However up to 50% of patients with heart failure has a preserved left ventricular ejection fraction (HF-PLVEF), suggesting that isolated diastolic dysfunction (DD) is the pathological mechanism underlying the clinical syndrome of heart failure in these patients [7].
B-type natriuretic peptide (BNP) is a neurohormone produced mainly by ventricular monocytes in response to increased enddiastolic pressure, or volume of the ventricles (Maeda K, et al. 1998). It produces diuresis, natriuresis, and vasodilatation which reduce the load on the heart. Patients with ventricular dysfunction tend to have high levels of plasma BNP; therefore BNP has been used as a diagnostic test for systolic dysfunction [8].
Some studies have shown that patients with diastolic dysfunction had high levels of plasma BNP [9].
Elevation of circulating natriuretic peptides is known to portend adverse outcomes in the setting of heart failure [9]. Communitybased studies have demonstrated that incremental elevations in B-type natriuretic peptide (BNP) even below current thresholds used to diagnose heart failure and are also associated with risk for incident heart failure in asymptomatic individuals [7].
The physiology underlying this relationship between natriuretic peptides and the eventual development of heart failure in the general population remains unclear. However, it may be related to the cardio-protective effects of natriuretic peptides, such as vasodilation, natriuresis and diuresis, and inhibition of the renin– angiotensin–aldosterone axis. In addition, it may be related to antihypertrophic and antifibrotic properties mediated by activity at the guanylyl cyclase type A receptor and signaling pathways such as calcineurin/nuclear factor of activated T cells (NFAT), the sodium- hydrogen exchanger 1 (NHE-1), and transforming growth factor beta (TGF-β) pathways [10].
Elevated BNP in apparently healthy individuals may represent an aggregate marker of subclinical abnormalities in cardiac structure and function that can develop during the progression from risk factors to heart failure. Natriuretic peptides have been shown to be associated with left ventricular (LV) hypertrophy and cardiac functional abnormalities in individuals with chronic kidney disease without overt heart failure as well as LV systolic and diastolic dysfunction in coronary artery disease [11].
Furthermore, prior studies have shown that BNP is increased in the setting of symptomatic LV systolic dysfunction, defined as the presence of a reduced LV ejection fraction [12]. However, the overall prevalence of reduced ejection fraction in the community is low and the association between BNP and cardiac abnormalities in the setting of preserved ejection fraction is less clear [12].
Conventional approaches to assessing cardiac dysfunction have limited sensitivity. However, the use of advanced methods for quantifying myocardial deformation may help to elucidate the relationship between BNP and LV function [13].
Mitral annular motion detected on tissue Doppler imaging (TDI) is considered a sign that the TDI correctly reflects the influence of myocardial relaxation on transmitral flows (Hillis, et al. 2004). Some studies reported that the TDI-derived index, the ratio of early trans-mitral flow velocity to early mitral annular velocity (E/ É), correlates with LV filling pressure and pulmonary capillary wedge pressure (PCWP). An E/ É ratio < 8 is associated with normal filling pressures and a ratio > 15 is associated with increased filling pressures [14].
Therefore, the purpose of our study was to evaluate the correlation between the plasma level of N-terminal pro-BNP (NT pro BNP) and the left ventricular diastolic dysfunction in asymptomatic patients as determined by tissue Doppler echocardiography.
We investigated 35 patients who were divided into two groups (control group and patient group), control group included 10 patients with normal systolic and diastolic function and patient group included 25 patients with normal systolic and asymptomatic diastolic dysfunction.
Both groups were of comparable age and gender, BMI and risk factors also in both groups the systolic function was preserved. By Doppler echocardiography there was no difference in the E velocity between two groups the mean value of E velocity was (0.64+0.18 m/sec) in the control group Vs (0.67+0.14) in the patient group (P = 0.302), but by tissue Doppler the difference was statistically significant, the mean value of e` velocity (m/sec) was (0.14+0.06 Vs 0.06+0.01 P = 0.001). And also the difference of mean value of E/e` was significant (5.03+0.149 Vs 0.55+1.79 P = 0.001).
The mean value of serum NT pro BNP (ng/ml) was (59.1) in the control group Vs (154.1) in patient group (P = 0.001). There was significant difference between two groups according to serum NT pro BNP.
There was significant correlation between serum NT pro BNP and Ev (P. value = 0.001) and E/e` (P = 0.001).
The sensitivity of serum NT Pro BNP in our study was 94.2, specificity was 88.1, positive predictive value was 90.6, negative predictive value was 28 and its accuracy was 86.2.
While previous studies of natriuretic peptides have focused on patients with a history of or concomitant symptoms consistent with heart failure [13], our analysis extends these findings to individuals with subclinical disease, suggesting a possible role for natriuretic peptide levels in identifying the presence of subclinical cardiac dysfunction among individuals without history of heart failure.
Several possible biological mechanisms may be responsible for the observed relation between subclinical myocardial dysfunction and elevated natriuretic peptide levels. In particular, natriuretic peptides may be markers of the pathologic processes associated with hypertension, such as increased interstitial fibrosis and/or abnormalities in myocardial calcium homeostasis. Alternatively, BNP may be directly involved in underlying changes in cardiac mechanics [15].
Imran et al. studied 313 asymptomatic patient with diastolic dysfunction and examined the associations of plasma N-terminal pro-B-type natriuretic peptide (NT-pro BNP) with both conventional and advanced echocardiographic measures of systolic and diastolic function and found that higher (NT-pro BNP) was associated with greater left ventricular mass index (P = 0.003), lateral É velocity (P < 0.001) and E/e` ratio (P < 0.001). Imran, et al. had also reported age related increase in BNP levels [16].
The results of our study were in agreement with (Imran, et al., according to the correlation between plasma level of NT-pro BNP and E/e` ratio and that there was no correlation between left ventricular mass and NT pro BNP. But our results were not in agreement with Imran, et al., who showed that there is correlation between age and plasma level of NT-pro BNP. The possible reason of this disagreement was due to the difference in the rangeof age as our patients were younger than those included in that study [16].
In our study, we found a positive correlation between E velocity and plasma level of NT-pro BNP which was not in agreement with (Imran, et al. 2013) who showed a positive correlation between É velocity and plasma level of NT-pro BNP.
Kasner, et al. who worked on 50 patients were included in crosssectional, case-controlled study and were divided into two groups. Group 1 included 30 hypertensive patients with asymptomatic diastolic dysfunction and group 2 included 20 patients who hadnormal diastolic flow patterns on echocardiography, conventional echo was done and blood samples were taken for serum BNP level from all patients. Karaca, et al. found that there was strong correlation between left ventricular mass index and plasma BNP levels (P < 0.05) and the sensitivity of BNP was 80%, its specificity
was 100%, positive predictive value was 100%, negative predictive value was 23% and its accuracy was 88% in identifying asymptomatic prolonged relaxation pattern [17].
The results of our study were not in agreement with Kasner, et al. as we found no correlation between plasma level of NT-pro BNP and left ventricular mass index (P. value = 0.682) [17]. and this result were not in agreement with Troughton who showed that increased natriuretic peptide levels in relation to abnormalities in left ventricular structures including alterations in LV dimensions, volumes, mass and mass index [12]. This was because the mean values of left ventricular mass and wall thickness in our sample were within normal ranges and individuals included in our study may represent patients at an earlier stage of cardiac dysfunction in the progression from underlying cause of diastolic dysfunction to heart failure.
Study limitations
Several limitations of this study deserve consideration. Patients in our study were generally young and of small number. The number of medications taken by study participants at time of entry into the study was not reported. Right ventricular systolic pressure was not measured due to inadequate tricuspid regurgitant jet velocity and Long-term clinical outcomes were not collected in our study precluding any analysis of the predictive value of NT-proBNP and/or myocardial function parameters in this sample. The extent to which elevated natriuretic peptides in this specific setting portends the eventual development of heart failure warrants further investigation.
Conclusion
Serum level of NT pro BNP can be used as innovative method in predicting asymptomatic diastolic dysfunction and so prevent the progression to symptomatic diastolic heart failure.
References
- Ferreira-Martins J, Leite-Moreira AF (2010) Physiologic basis and pathophysiologic implications of the diastolic properties of the cardiac muscle. BioMed Research International.
- Maeder MT, Kaye DM (2009) Heart failure with normal left ventricular ejection fraction. J Am Coll Cardiol 53(11): 905-918.
- Bursi F, Weston S, Redfield MM, Jacobsen SJ, Pakhomov S, et al. (2006) Systolic and diastolic heart failure in the community. JAMA 296(18): 2209-2216.
- Owan TE, Hodge DO, Herges RM, Jacobsen SJ, Roger VL, et al. (2006) Trends in prevalence and outcome of heart failure with preserved ejection fraction. N Engl J Med 355(3): 251-259.
- Weber M, Hamm C (2006) Role of B-type natriuretic peptide (BNP) and NT-proBNP in clinical routine. Heart 92(6): 843-849.
- Dahlstorm U (2004) Can natriuretic peptides be used for the diagnosis of diastolic heart failure? Eur J Heart Fail 6: 281-287.
- Kurl S, Ala-Kopsala M, Ruskoaho H, Makikallio T, NyyssonenK, et al. (2009) Plasma N-terminal fragments of natriuretic peptides predict the risk of stroke and atrial fibrillation in men. Heart 95(13): 1067-1071.
- Ng AC, Delgado V, Bertini M, van der Meer RW, Rijzewijk LJ, et al. (2009) Findings from Left Ventricular Strain and Strain Rate Imaging in Asymptomatic Patients With Type 2 Diabetes Mellitus. Am J Cardiol 104(10):1398-1401.
- Dokainish H, Nguyen JS, Sengupta R, Pillai M, Alam M, et al. (2010) Do additional echocardiographic variables increase the accuracy of E/e′ for predicting left ventricular filling pressure in normal ejection fraction? An echocardiographic and invasive hemodynamic study. J Am Soc Echocardiogr 23(2): 156-161.
- Calvieri C, Rubattu S, Volpe M (2012) Molecular mechanisms underlying cardiac antihypertrophic and antifibrotic effects of natriuretic peptides. J Mol Med 90(1): 5-13.
- Mishra RK, Li Y, Ricardo AC, Yang W, Keane M, et al. (2013) Association of N-Terminal pro-B-type natriuretic peptide with left ventricular structure and function in chronic kidney disease (from the Chronic Renal Insufficiency Cohort [CRIC]). Am J Cardiol 111(3): 432-438.
- Troughton R, Richards AM (2009) B-type natriuretic peptides and echocardiographic measures of cardiac structure and function. JACC Cardiovasc Imaging 2(2): 216-225.
- Yoneyama A, Koyama J, Tomita T, Kumazaki S, Tsutsui H, et al. (2008) Relationship of plasma brain-type natriuretic peptide levels to left ventricular longitudinal function in patients with congestive heart failure assessed by strain Doppler imaging. Int J Cardiol 130(1): 56-63.
- Ommen SR, Nishimura RA, Appleton CP, Miller FA, Oh JK, et al. (2000) Clinical utility of Doppler echocardiography and tissue Doppler imaging in the estimation of left ventricular filling pressures: A comparative simultaneous Doppler-catheterization study. Circulation 102(15): 1788-1794.
- Solomon SD, Verma A, Desai A, Hassanein A, Izzo J, et al. (2010) Effect of intensive versus standard blood pressure lowering on diastolic function inpatients with uncontrolled hypertension and diastolic dysfunction. Hypertension 55(2): 241-248.
- Imran U, Susan C, Chung-Leih H, Anil V, James D, et al. (2013) Relation of N-Terminal Pro-B-Type Natriuretic Peptide with Diastolic Function in hypertensive Heart Disease. Am J Hypertens 26(10): 1234-1241.
- Kasner M, Westermann D, Steendijk P, Gaub R, Wilkenshoff U, et al. (2007) Utility of Doppler echocardiography and tissue Doppler imaging in the estimation of diastolic function in heart failure with normal ejection fraction: a comparative Doppler conductance catheterization study. Circulation 116(6): 637-647.