Phenytoin Induced Cerebellar Ataxia and Orofacial Dyskinesia in a Case of Disseminated Cysticercosis: A Case Report
P.N.Venkatarathnamma1*, M.N.Chandra Shekara2, S.V.Srinivasa3, Jagadish basavaih4
1* Professor of Medicine, Sri Devaraj Urs Academy of Higher Education and Research, Tamaka, Kolar,Karnataka, India.
2 Assistant Professor of Neurology, Sri Devaraj Urs Academy of Higher Education and Research, Tamaka,Kolar, Karnataka, India.
3 Assistant Professor of Medicine, Sri Devaraj Urs Academy of Higher Education and Research, Tamaka,Kolar, Karnataka, India.
4 Assistant Professor of Radiology, Sri Devaraj Urs Academy of Higher Education and Research, Tamaka,Kolar, Karnataka, India.
*Corresponding Author
P.N.Venkatarathnamma
Professor of Medicine,
Sri Devaraj Urs Academy of Higher Education and Research,
Tamaka, Kolar, Karnataka,India.
E-mail: drpnvr@rediffmail.com
Article Type: Case Report
Received: September 15, 2013; Accepted: October 3, 2013; Published: October 7, 2013
Citation: P.N.Venkatarathnamma, M.N.Chandra Shekara, S.V.Srinivasa, Jagadish basavaih(2013) Phenytoin Induced Cerebellar Ataxia and Orofacial Dyskinesia in a Case of Disseminated Cysticercosis: A Case Report. Int J Clin Pharmacol Toxicol. 2:702, 102-105. doi: dx.doi.org/10.19070/2167-910X-1300019
Copyright: P.N.Venkatarathnamma© 2013. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
We report a 25 year old male patient who developed phenytoin induced cerebellar ataxia and orofacial dyskinesias that resolved after discontinuation of phenytoin. This patient also had disseminated cysticercosis, i.e. diffuse involvement of the brain, spinal cord, intramuscular and subcutaneous tissues. This case report is interesting because cerebellar ataxia is a known manifestation of both neurocysticercosis and phenytoin toxicity, while it is reversible in case of phenytoin toxicity.
2.Introduction
3.Case Report
4.Discussion
5.Conclusion
6.References
Keywords
Phenytoin Toxicity; Cerebellar Ataxia; Disseminated Cysticercosis.
Introduction
Phenytoin is a hydantoin compound related to the barbiturates that are used for the treatment of seizures. Nystagmus, ataxia, and drowsiness, are the most frequently encountered dose-related side effects of phenytoin toxicity and are correlated with plasma levels. It was revealed that in the majority of the patients, nystagmus, ataxia, and drowsiness appear when plasma levels reach 20, 30, and 40 mcg/dL, respectively, but dose-related side effects may show a great diversity among patients [1].
Cysticercosis is caused by cysticercus cellulosae,the larval form of the tapeworm Taenia solium.Humans acquire cysticercosis through faeco-oral contamination with taenia solium eggs from tapeworm carriers. Disseminated cysticercosis is an uncommon manifestation of this common disease [2]. Widespread dissemination of the cysticerci can result in involvement of almost any organ of the body including the central nervous system, skeletal muscles, subcutaneous tissue, and the eyes. Neurocysticercosis, is a pleomorphic disease whose clinical manifestations vary with the number, size, location and stage of cysticerci as well as the intensity of the host’s immune response [3]. Common manifestations include intractable seizures, cognitive decline, cerebellar ataxia, focal neurological signs, hydrocephalus and features of spinal cord compression.
Case Report
A 25 year old male, resident of a rural place near Kolar (Karnataka, India), an employee at a cold storage, was on treatment with phenytoin for history of recurrent seizures (generalized tonic clonic type) for one year duration. Now he presented with giddiness, vomiting, unsteady gait and slurring of speech of one week. He is a non vegetarian (consumes pork). There was no relevant past medical history. The patient had not undergone any prior radio imaging and was on Tablet phenytoin - 300mg/day in divided doses started by a private practitioner.
On examination the patient had multiple subcutaneous nodules over the right cheek, neck and upper trunk (Figure.1). Neurological examination revealed a wide based ataxic gait, scanning dysarthria, horizontal gaze nystagmus, bilateral cerebellar signs (left more than right) and occasional choreiform movements involving the left upper limb and orofacial dyskinesias.
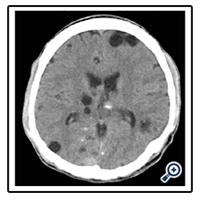
Routine laboratory examination was unremarkable except for a macrocytic normochromic blood picture. Computed tomography of the brain showed multiple hypodense lesions with central hyperdensity (more than 100 in number), noted diffusely in the brain parenchyma, largest measuring 3x2 cms in the left frontal region, and was suggestive of infective granulomas.(Figure 2).
MRI showed multiple lesions throughout the brain parenchyma in both cerebral hemispheres, cerebellar hemispheres, brain stem and spinal cord. The lesions were also noted in the intramuscular and subcutaneous plane. These lesions were hypointense on T1 weighted images, with enhancement of the scolices post gadolinium administration. (Figure 3A & 3B) On T2 weighted images the lesions were hyperintense and showed restriction of diffusion on diffusion weighted imaging. (Figure 3C & 3D).
Figure 3A: T1W MRI precontrast image showing multiple scattered neurocysticercosis in the brain parenchyma.
Figure 3B: T1 W postcontrast image showing multiple scattered neurocysticercosis in the brain parenchyma.
Ultrasonography of the nodules showed multiple well defined cystic lesions, largest measuring 2x1x1.5 cms in the subcutaneous plane of right chest wall (Figure 4). Microscopic examination of the aspirated fluid from a nodule showed pale eosinophilic proteinaceous material, with no parasite structure or hooklets. An excision biopsy with microscopic study of a subcutaneous nodule in the right chest revealed cystic areas, fibro fatty tissue and a structure of tiny lymph node within the cystic cavity consistent with cysticercosis.
A serum phenytoin level (assessed by kinetic interaction of microparticles in a solution) was 32.9 mcg/ ml (10 – 20 mcg/ml). Ophthalmologic examination including a slit lamp examination was normal. EEG showed predominantly alpha wave of 8-10 Hz measuring 60-112 microvolts in the background which was reported to be normal.
The patient was treated with albendazole, steroids for cysticercosis and anticonvulsant changed to sodium valproate. He showed significant improvement of symptoms and was discharged after a period of ten days and is on regular follow up.
Discussion
Drug induced movement disorders are primarily associated with drugs that block dopamine receptors (neuroleptics) or central dopaminergic transmission but may be observed occasionally due to anticonvulsants also [4]. Phenytoin has been reported to induce orofacial dyskinesias, choreoathetosis, ballismus, myoclonus and dystonia [5,6,7] In animal experiments phenytoin has been shown to possess dopamine receptor blocking properties which can lead to dyskinesias [4].
Phenytoin has been shown both clinically and experimentally to be toxic to the cerebellum and degeneration of Purkinje cells has been seen after single large overdoses in man [8,9]. These involuntary movements occur in patients with high plasma phenytoin levels as was found in our case where the plasma phenytoin levels was in toxic range. Cerebellar atrophy has been noted in patients with long term phenytoin exposure [10,11]. Moreover the toxic level of phenytoin in blood at the time of presentation and disappearance of involuntary movements within 3 days of reducing the dose of phenytoin virtually rules out the possibility of these abnormal movements as part of disease process of cysticercosis.
Widespread dissemination of cysticerci throughout the body was reported as early as 1912 by the British army medical officers stationed in India [12]. Kumar et al and Wadia et al reviewed 22 cases each [13,14].
The larvae form cysts commonly in the brain, meninges, and eyes, which together constitute 86% of the cases. The other locations are muscle, heart, lungs, and peritoneum. Cases in the maxillofacial region, including the tongue and cheek muscles, are rarely reported. [15] The incidence of spinal neurocysticercosis reported in most series is only 1.5 to 3%.[16] Cerebral cysts usually number 7 to 10 per patient.[17] Our patient had innumerable number of cerebral cysts, and subcutaneous nodules, intramuscular cysts in the masseter and genioglossus along with involvement of the spinal cord.
Conclusion
This case emphasizes the importance of periodic anticonvulsant drug level monitoring and importance of workup for the cause of seizures before starting anticonvulsants.
References
- Browne TR. Phenytoin and other hydantoins. In: Engel J Jr, Pedley TA, eds. Epilepsy: A Comprehensive Textbook. Philadelphia: Lippincott - Raven, 1987: 1557-79.
- Kumar A, Bhagwani DK, Sharma RK, Kavita, Sharma S, Datar S, et al. Disseminated cysticercosis. Indian Pediatr 1996; 33: 337-9.
- Takayanagui OM, Odashima NS. Clinical aspects of neurocysticercosis. Parasito Int 2006; 55 Suppl: S111 - 5. Epub 2005 Dec 5.
- Tolosa E, Alom I, Marti MJ. Drug-induced dyskinesias. In: Parkinson’s Disease and Movement Disorders, 2nd edn. Eds. Jankovic J, Tolosa E, Eds. Williams and Baltimore, Wilkins, 1992; pp 375-397.
- Chadwick J, Reynolds EH, Marsden CD. Anticonvulsant-induced dyskinesia: A comparison with dyskinesias induced by neuroleptics.J Neurol Neurosurg Psychiatry 1976; 39:1210-1218.
- Duarte J, Sempere AP, Cabezas MC, Marcos J, Claveria LE. Postural myoclonus induced by phenytoin. Clin Neuropharmacol 1996; 19:536-8.
- Filloux F, Thompson JA. Transient chorea induced by phenytoin. J Pediatr 1987; 110:639-41. Erratum in: J Pediatr 1987; 110:1000.
- F. Buchtal et al., Clinical and electroencephalographic correlations with serum levels of diphenylhydantoin, Arch. Neurol. 1960; 2: 624- 631.
- Kokenge R, Kutt H, McDowell F. Neurological sequelae following Dilantin overdose in a patient and in experimental animals. Neurology. 1965; 15: 823-859.
- Ghatak NR, Santoso RA, McKinney WM. Cerebellar degeneration following long-term phenytoin therapy. Neurology. 1976; 26: 818- 820.
- Ney GC, Lantos G, Barr WB, Schaul N. Cerebellar atrophy in patients with long-term phenytoin exposure and epilepsy. Arch Neurol. 1994 Aug; 51(8): 767-71.
- Bhalla A, Sood A, Sachdeve A, Varma V. Disseminated Cysticercosis: A case report and review of the literature, J Med Case Rep 2008; 2: 137.
- Kumar A et al. Disseminated cysticercosis. Indian Pediatr 1196; 33: 337-9.
- Wadia N, Desai S, Bhatt M. Disseminated cysticercosis: New observations, including CT scan findings and experience with treatment by praziqauntel. Brain 1988; 111: 597-614.
- Sidhu R, Nada R, Palta A , Mohan H, Suri S. Maxillofacial cysticercosis. Uncommon appearance of a common disease. J Ultrasound Med 2002; 21: 199-202.
- King CH, et al. Cestodes (tapeworms). In: Mandell GL, et al. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 7th ed. Philadelphia, Pa.: Churchill Livingstone Elsevier;2010.
- Alsina GA, Johnson JP, Mc Bride DQ, et al. Spinal neurocysticercosis. Neurosurg Focus 2002: 12(6): e8.