Observability of Minimal Cell Models
Motahareh Lotfzarie, Alireza Faghani Ghodrat*
Department of Electrical Engineering, Islamic Azad University, Damavand Branch, Tehran, Iran.
*Corresponding Author
Motahareh Lotfzarie,
Department of Electrical Engineering,
Islamic Azad University,
Damavand Branch, Tehran, Iran.
E-mail: alireza.faghani@chmail.ir
Article Type: Research Article
Received: February 29, 2014; Accepted: May 05, 2014; Published: May 28, 2014
Citation: Alireza Faghani Ghodrat, Motahareh Lotfzarie (2014) Observability of Minimal Cell Models. Int J Comput Neural Eng, 1(1) 1-5.
Copyright: Alireza Faghani Ghodrat© 2014. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Identifying and modeling of biological systems is very useful to understand cell's dynamic. To know what really happens inside the cell we need to observe the state of a cell. In fact observability is a structural property of a control system defined as the possibility to deduce the state of the system from observing its input-output behavior. Any complex cell model is a combination of some minimal models which are simpler than complex cell model because they have two dimensions. These models can describe the behavior of the cell. The property of observability for nonlinear systems is very useful in analyzing such systems. This paper deals with the observability of minimal cell models. Based on the fact that the minimal cell models are nonlinear, analyzing the property of these models need nonlinear methods. The method has been used for observability is Lie Derivative. The results indicate observability of minimal cell models.
2.Introduction
3.Problem Formulation
3.1 Hodgkin-Huxley Model
3.2 Minimal Cell Models
4.Method
4.1 Observability
5. Results
5.1 Observability of Minimal Cell Models
6. Conclusion
7. References
Keywords
Observability; Lie Derivative; Nonlinear Systems; Hodgkin-Huxley Model; Minimal Cell Models.
Introduction
Neurons are responsible for transmitting messages throughout the body via long distance electrical signals known as action potentials(AP). These depend on the active transport of sodium and potassium ions across the cell membrane. At the cellular level, the electrical signal excitable cells amplify and propagate is a change in the potential across a cell’s membrane, caused by different ion currents flowing through the membrane’s channels. In fact for each excitation event, the electrical signal is called an AP. For non-pacemaking excitable cells, an AP is an externally triggered all or nothing response to an external stimulus: if the stimulus is sufficiently strong, the cell fires an excitation AP [1]. The effect of various drugs on the process of neuron firing is a current research interest. The Hodgkin-Huxley equations mathematically model the influx and efflux of these ions across the cell membrane. One of the most important models in computational neuroscience is the Hodgkin-Huxley model of the squid giant axon [2]. The Hodgkin-Huxley model is a combination of minimal models. A mixture of one amplifying and one resonant gating variable results a minimal model. Each minimal model can oscillate at least from some values of its parameters. There are only few minimal models and understanding their dynamics can shed light on dynamics of more complicated electrophysiological models [3].
Using pioneering experimental techniques of that time, Hodgkin and Huxley in 1952 [8] determined that squid axon has three major current: voltage gated persistent K+ current with four activation gates (n4 term), voltage gated transient Na+ current with three activation gates and one inactivation gate (m3h term) and Ohmic leak current, IL, which is carried mostly by Cl- ions. The complete set of space clamped Hodgkin-Huxley equations is [3]:
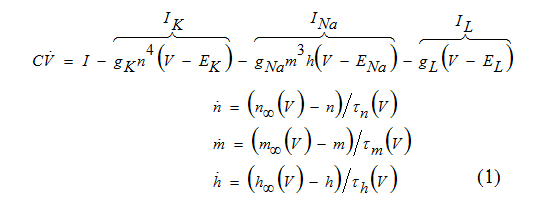
Where
By using this formulation, we obtain the minimal cell models.
Let us do the following thought experiment: Consider a conductance-based model capable of exhibiting periodic spiking and remove completely a current or one of its gating variables, and if the reduced model has a limit cycle attractor at least for some values of parameters, we arrive at the model that satisfies the following two properties:
• It has a limit cycle attractor, at least for some values of parameters.
• If one removes any current or gating variable, the model has only equilibrium attractors for any values of parameters.
We refer to such models as being minimal or irreducible for spiking. Thus, minimal models can exhibit periodic activity, but their reductions cannot. According to this definition, any space-clamped conductance-based model is a combination of some minimal ones. If not, then it can be reduced to a new minimal model. For example, the Hodgkin-Huxley model considered is not minimal for spiking. This model consists of three current: leakage IL, transient sodium INa,t (gating variables m and h) and persistent potassium Ik (gating variable n).
For definition of the minimal models we employ here a bottom-up approach, which is based on the following rule: A mixture of one amplifying and one resonant gating variable (plus an ohmic leak current) results in a minimal model. Indeed neither of the variables alone can produce oscillation but both are enough [3].
The amplifying gating variable is the activation variable m for voltage gated inward current or inactivation variable h for voltage gated outward current. Thesevariables amplify voltage changes via a positive feedback loop. Indeed a small depolarization increases m and decreases h which in turn increases inward and decrease outward current and produce more depolarization. Similarly a small hyper polarization decreases m and increases h, resulting in less inward and more outward current and hence in more hyper polarization. The resonant gating variable is the inactivation variable h for an inward current or activation variable n for an outward current. These variables resist voltage changes via negative feedback loop. A small depolarization decreases h and increases n which in turn decreasesinward and increases outward current and produce a net outward current that resists the depolarization. Similarly a small hyper polarization produces inward current and possibly rebound depolarization.
A typical neuronal model consists of at least on amplifying and at least one resonant gating variable. Two amplifying and two resonant gating variables produce four different combinations depicted in Figure. However, the number of minimal models is not four, but six. The additional models arise due to the fact that a pair of gating variables may describe activation/inactivation properties of the same current or of two different currents [3].
Figure 1: Any combination of one amplifying and one resonant gating variables results in a spiking model [3].
One of the most fundamental models in computational neuroscience is the INa,p+ Ik-model consisting of a fast Na+ current and a relatively slower K+ current
Here m(t) is much faster than the voltage variableV(t), so that m approaches the asymptotic value m∞ (V ) instantaneously. In this case we can substitute m = m∞(V ).
An interesting example of a spiking mechanism is given by the INa,t-model which consisting only of an ohmic leak current and a transient voltage-gated inward Na+ current.
The upstroke of an action potential is generated because of the regenerative process involving the activation gate m. This mechanism is similar to the one in the Hodgkin-Huxley model or in the INa,p+ Ik-model: Increase of m results in increase of the inward current, hence more depolarization and more increase of m until the excited state is achieved. At the excited state there is a balance of the Na+ inward current and the leak outward current.
Assuming that activation dynamics is instantaneous,we use m = m∞ (V ) in the voltage equation and obtain:
The system describes the essence of the mechanism of slow sub threshold voltage oscillations in some cortical,thalamic, and hippocampal neurons.
We assume that the activation kinetics of the Na+ current is instantaneous, and use m = m∞(V ) in the voltage equation to obtain a two-dimensional system:
The persistent Na+ current, which amplifies damped oscillations in the INa,p+Ih-model, can be substituted by the K+ inwardly rectifying current Ikir to achieve thesame amplifying effect.
Since kinetics of Ikir is practically instantaneous, we can use hKir=hKir,∞(V), in the voltage equation above and consider the two-dimensional system.
formula
In the Ik+Ikir-model
The amplifying current is Ikir with inactivation gating variable h, and the resonant current is Ik with activation variable n.
The kinetics of the amplifying current Ikir is relatively fast so that h = h∞(V ) can be used in the voltage equation to reduce the three-dimensional system above to a two dimensional system.
The last minimal voltage-gated model has only one transient K+ current, often referred to as being A-current IA, yet it can also generate sustained oscillations. In some sense, the model is similar to the INa,t -model. Indeed, each consists of only one transient current and an Ohmic leak current. The only difference is that Acurrent is outward, and as a result, the action potentials are fired downward. The A-current has activation and inactivation variables m, and h, respectively, and the model has the form:
We are tempted to substitute m = m∞(V ) into the voltage equation above and reduce the IA -model to a twodimensional system, which hope-fully would have the right kind of nullclines and a limit cycle attractor.
formula
Then we define the observability property of systems.
Consider the nonlinear system [5]:
Two states x0 and x1 are distinguishable if there exists an input function μ such that:
The system is locally observable at x0 if exists a neighborhood of x0 such that every x in that neighborhood other than x0 is distinguishable from x0. A test for local observability is that [6]:
Must have rank n, where n is the rank of x and:
Where the output of nonlinear system is:
Generally h1 to hP are nonlinear functions of x, and:
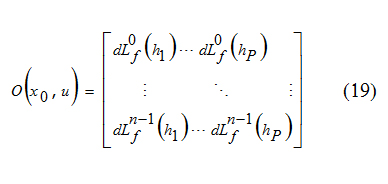
Here Lf(hi) is the Lie derivative of h with respect to f which is defined below [6]:
By definition, L0f (h )=h.
We can also define higher-order Lie Derivatives [7]:
By this test method, we check the observability of each minimal cell model.
For each minimal model first we construct l matrix then O matrix and then calculate the rank of O matrix. For some conditions the O matrix is not full rank. So we determine the condition that the O matrix is full rank and then the system is observable.
For example in the following we calculate the O matrix for INa,p+Ik-model and write the details for checking the observability condition.
Equation (3) describes the INa,p+Ik-mode. By using these equations, ƒ1 and ƒ2 can be written as below:
Here I=0.
After calculation the Lie derivatives we can construct the l matrix as below:
And
For observability O must be full rank. So:
With this condition the system is observable. We can run above procedure for other minimal cell model. The results are given in TABLE.
Conclusion
Using the results table, all minimal cell models are observable except on one line in state plane. By this condition we can consider these minimal models observable. Here we analyzed the observability property of minimal cell models. As a result we can use observers like Kalman Filter [8], in this case Extended Kalman Filter to observe the states of this nonlinear systems and via using a state feedback we can control the nonlinear behavior of a cell. In next research we'll focus on controllability property [5],[7],[9] of minimal cell models.
References
- Pei Ye, Emilia Entcheva, Scott A. Smolka, Radu Grosu, “Symbolic Analysis of the Neuron Action Potential”, State University of New York at Stony Brook Stony Brook, Department of BioMedical Engineering, 2008.
- A. L. Hodgkin, A. F. Huxley, “A quantitative description of membrane current and its application to conduction and excitation in nerve”, J. physiol, 500-544, 1952.
- Eugene. M. Izhikevich, “Dynamical Systems in Neuroscience: The Geometry of Excitability and Bursting”, Springer Verlog, 2004.
- Milena Anguelova, “Nonlinear Observability and Identifiability: General Theory and a Case Study of a Kinetic Model”, Chalmers University of technology and G¨oteborg University, 2004.
- R. Hermann, A. J. Krener, “Nonlinear Controllability and Observability”, IEEE Transaction on Automatic Control, vol. AC-22, No. 5, pp. 728-740, 1977.
- J. K. Hedrik, A. Girard, “A Control of Nonlinear Dynamic Systems: Theory and Application” ,2005.
- M. R. James, “Controllablity and Observability of Nonlinear Systems”, Institude for Systems Research, T.R. 87-195, 1987.
- MOHINDER S. GREWAL, ANGUS P. ANDREWS, “KALMAN FILTERING, Theory and Practice Using MATLAB”, Third Edition, California State University at Fullerton, 2008.
- K. Röbenack, “Computation of Controllability and Observability Matrices – Duality and Automatic Differentiation”, proceeding in Applied Mathematics and Mechanics, 2003.





