Evaluation of Subcutaneous Immunotherapy in Asthmatics to Dermatophagoides Siboney and Dermatophagoides Pteronyssinus
Ra�l L�zaro Castro-Almarales, MD1*, Mercedes Ronquillo-D�az, MD2, Mirta �lvarez-Castell�, MD, PhD3, Jos� Rodr�guez-Canosa, MD4, Mayda Gonz�lez-Le�n5, Irene Enr�quez-Dom�nguez, MD6, B�rbara Ivonne Navarro-Viltre, MD7, Maytee Mateo-Morej�n, BSc8, Yunia Oliva-D�az, BSc9, Wendy Ram�rez-Gonz�lez, BSc10, Alexis Labrada-Rosado, PhD11
1 Second Degree Specialist in Allergy, Auxiliary Professor and Researcher.Centro Nacional de Biopreparados, Bejucal, Mayabeque, Cuba.University-
Hospital �General Calixto Garc�a�, Allergy and Immunology Service, Havana, Cuba.
2 Second Degree Specialist in Allergy, Auxiliary Professor; University Hospital �General Calixto Garc�a�, Allergy and Immunology Service, Havana, Cuba.
3 Second Degree Specialist in Allergy, Auxiliary Professor and Researcher; University Hospital �General Calixto Garc�a�, Allergy and Immunology Service, Havana, Cuba.
4 Second Degree Specialist in Allergy, Auxiliary Professor; UniversityHospital �General Calixto Garc�a�, Allergy and Immunology Service, Havana, Cuba.
5 Second Degree Specialist in Integral General Medicine, Teaching Polyclinic �Pedro Fonseca �lvarez�, La Lisa, Havana, Cuba.
6 First Degree Specialist in Allergy and Integral General Medicine, University Hospital �General Calixto Garc�a�, Allergy and Immunology Service, Havana, Cuba.
7 First Degree Specialist in Allergy, Master in Immunology, Teaching Polyclinic �Capdevila�, Boyeros, Havana, Cuba.
8 Centro Nacional de Biopreparados, Allergens Department, Bejucal, Mayabeque, Cuba.
9 Centro Nacional de Biopreparados, Allergens Department, Bejucal, Mayabeque, Cuba.
10 Centro Nacional de Biopreparados, Allergens Department, Bejucal, Mayabeque, Cuba.
11 Director of Research and Development. Centro Nacional de Biopreparados, Bejucal, Mayabeque, Cuba.
*Corresponding Author
Ra�l L�zaro Castro-Almarales, MD,
National Center of Bioproducts, Division of Research and Development, Clinical Trials Group. Carretera Beltr�n km 11/2, Bejucal, Zip postal 32600, Mayabeque, Cuba.
Tel/Fax: +53-47682441
E-mail: rcastro@biocen.cu
Received: October 22, 2021; Accepted: January 24, 2022; Published: January 28, 2022
Citation: Ra�l L�zaro Castro-Almarales, MD, Mercedes Ronquillo-D�az, MD, Mirta �lvarez-Castell�, MD, PhD, Jos� Rodr�guez-Canosa, MD, Mayda Gonz�lez-Le�n, Irene Enr�quez - Dom�nguez, MD, et al., Evaluation of Subcutaneous Immunotherapy in Asthmatics to Dermatophagoides Siboney and Dermatophagoides Pteronyssinus. Int J Clin Med
Allergy. 2022;07(01):73-80. doi: dx.doi.org/10.19070/2332-2799-2200014
Copyright: Ra�l L�zaro Castro-Almarales, MD�2021. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
2.Introduction
3.Discussion
4.References
Introduction
Prevalence of allergic diseases such as allergic rhinitis and asthma
is increasing to epidemic proportions in the world. [1-3] In Latin
America, asthma prevalence is higher in tropical regions. [2-4]
Particularly, in Cuba, asthma affects from 8.5% to 10% of adults
and up to 17.8% of children, [5-8] and it is associated with high
morbidity and profound social and economic impact, including
reduced quality of life of patients, lower productivity and increase
of indirect and direct costs. [9-10]
Increase of prevalence of allergic asthma is commonly associated
with changes in lifestyle and environment. Particularly, allergic
asthma is strongly linked to IgE sensitization to House Dust
Mites(HDM).[2, 3, 11, 12] Epidemiologic studies performed in
Cuba have reported that the more relevant HDMspecies are Dermatophagoides
pteronyssinus (Dp), Dermatophagoides siboney
(Ds) and Blomiatropicalis (Bt).13-16D. siboney is a tropical species
described first in Cuba and found also in the Caribbean [11]
[12], phylogenetically very close to D. farinae, which is absent in
Cuba. The allergenic relevance of D. siboney and its strong association
with asthma in Cuba has been reported previously. [13-15]
D. siboney shares extensive cross-reactivity with D. farina and D.
pteronyssinus, which is also common in Cuba.[15]
Management of allergic asthma is based on pharmacological
symptomatic treatment, allergen avoidance and allergen-specific
immunotherapy.[17-20] Allergen-specific immunotherapy (AIT)
is presently acknowledged as a biological response modifier, inducing
immunological tolerance towards the allergen.
It is the only available treatment able to influence the natural
course of allergic disease. Extensive clinical evidence supports
the efficacy of AIT.[21] In addition, this etiological therapeutic
approach can be used also in a preventive setting, avoiding development
of new sensitizations and the aggravation of the disease.
The clinical effect of AIT is based on modulation of the Th1/
Th2 balance of the allergen-specific immune response, inducing
also T regulatory cells and IgG4 blocking antibodies.[17-20]
Safety concerns about injection AIT are related to the risks of
anaphylactic reactions during treatment. In this regard, the standardization
of the allergen products, in terms of allergenic activity,
is very relevant for the accurate control of the administered doses.
[17, 18, 20, 22]
AIT using HDM allergens, is regarded an effective treatment for
mild to moderate asthma in sensitized patients.[21, 22] A key aspect
is the proper selection of the allergens and the assessment of
its relevance to the disease. Both, D. pteronyssinus and D. siboney
are regarded as important allergen sources and sensitizers for Cuban
asthmatics.[5, 7, 13, 16] Therefore, the aim of this work was
to assess the efficacy and safety of standardized allergen extracts
of Dermatophagoides pteronyssinus and Dermatophagoides siboney
by subcutaneous route in allergic asthmatic adults sensitized
to these mite species.
Methods
Study design
Two separate clinical trials, for each allergen product, Dp and
Ds, were conducted simultaneously; both with the same design:
Phase II (therapeutic effect), double-blind, placebo-controlled,
ramdomized trials, in 40 adult asthmatic patients each, with mild
to moderate persistent asthma. Half of the patients in each trial
received active treatment and the other half placebo. Both trials
were conducted at the Allergy and Immunology Service of
the �General Calixto Garc�a� University Hospital (Code in the
Cuban Public Registry of Clinical Trials: RPCEC00000027 and
RPCEC00000028).[23] The studies were designed and conducted
according with the international guidelines for Good Clinical
Practice and were approved by the Research Ethics Committee of
the clinical site and the Cuban regulatory authority (CECMED).
Treatment
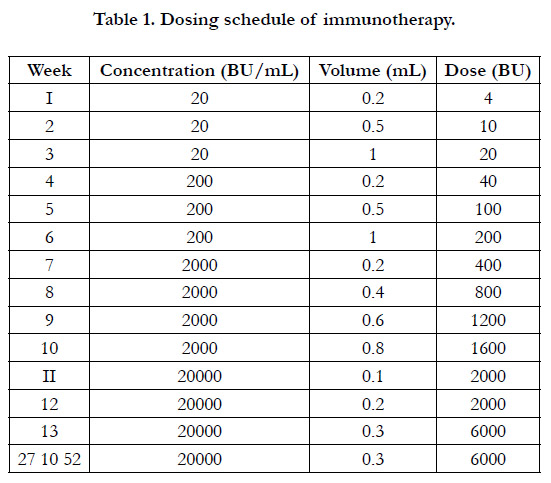
Allergen injections were administered by subcutaneous route in
a dose escalation scheme, first, aninduction phase with weekly
injections during the first 13 weeks, followed by a maintenance
phase ofmonthly injections up to 12 months (Table 1). The 100%
of included patients completing 12 months of AIT. Standardized
allergen extracts VALERGEN-DP (D. pteronyssinus) and
VALERGEN-DS (D. siboney), developed and manufactured by
BIOCEN (Bejucal, Cuba) were used. Both products are licensed
in Cuba as medicines. The standardization is expressed in Biological
Units (BU) according to the definition of Nordic Guidelines
for the Registration of Allergen Products.[24] The maintenance
dose was 6000 BU, which is equivalent to 12 �g of the major allergen
Der p 1 or 24 �g of Der s 1, respectively.[25]
Extracts are available as freeze-dried products, which are reconstituted
in an aqueous PBS albumin diluent. Both, the active and
control group, received concomitant symptomatic treatment
comprisingantihistamines, bronchodilators, �2-agonists and oral
steroids, as prescribed by blinded physicians.
Participants
The study involved men and women aged 18 to 45 years with clinical
diagnosis of mild to moderate persistent asthma diagnosed
according to Global Initiative for Asthma (GINA) guidelines, [26]
possiblyincluding concomitant manifestations of rhinitis, atopic
dermatitis and/or conjunctivitis. Diagnosis ofallergic asthma was
performed by an expert physician and took into account individual
patient history,physical examination and response upon bronchodilator
administration, with a minimum of 12% Peak.
Expiratory Flow increase. Patient�s allergic status was supported
by a positive skin prick test to D. pteronyssinus or D. siboney allergen
extracts (Centro Nacional de Biopreparados (BIOCEN),
Cuba, 20 000 BU/mL) and clinical assessment consistent with
allergic respiratory symptoms (cough, wheezing, chest tightness
and dyspnea) upon exposure to house dust. The skin test was
considered positive if the wheal diameter was greater or equal
to 3 mm. Since strict monosensitization in this case is practically
impossible due to the high degree of cross-reactivity between
both mite species, patients with predominant sensitization to one
of the two species were selected, i.e. with a reaction size larger
than 2 mm, with respect to the other mite species. In addition to
D. siboney and D. pteronyssinus, reactivity to Blomiatropicalis was
also tested. Patients were excluded from the trial if they had been
treated with allergen immunotherapy in the last two years, or if
they had had severe intermittent or persistent asthma or received
immunostimulant or immunosuppressive treatment one year prior
to the study.
Outcomes
The therapeutic effect was measured through symptom and medication
scores, and the combination of both as the main endpoint.
In addition, lung function, and skin reactivity relative to histamine
(Ch10) were used also as secondary outcomes. Symptom and
Medication (SM) scores were collected using a form filled by the
patient with a 4 level scale with the following values: 0 = absent,
1 = mild, 2 = moderate, 3 = severe, for each symptom manifestation
(cough, wheezing, chest tightness and dyspnea). In addition,
Medication Score measured the frequency of drug intake. Each
time a drug was consumed the score was incremented by 1 point,
except oral steroids that worth 2 points. At the end of the month,
all points were summed. [27, 28] For the semi annual partial evaluations
(at months 6 and 12) the values of the last two months
were averaged: 5-6, and 11-12 months, respectively. Baseline pretreatment
value was established measuring SM scores during one
month. For comparing with published meta-analysis the Standard
Mean Difference (SMD) was calculated (i.e. difference between
Active and Placebo, divided by the standard error) according to
Cochrane methodology.[29]
Allergen specific reactivity was assessed by Skin Prick Test (SPT)
to Dp, Ds and Bt allergen extractsat 20000 BU/mL. Stainless steel
lancets with tips of 1 mm (ALK, Denmark) were used according
toDreborg.[30] Histamine HCl 10 mg/mL was used as a positive
control, and as a negative control, PBS. Inorder to record the
test results, a line was drawn around the wheal and transferred
to a transparent adhesive tape, which was taped on to the data
recording book. The largest and the orthogonal diameters were
measured on the wheal drawing, and the mean diameter was calculated.
Furthermore, the mean diameter (d) between the two
arms was calculated. The test was considered valid if the difference
between both arms wheals was less than 2 mm for wheals
between 3 and 6 mm, or less than 3 mm for larger wheals; besides
being positive for Histamine (positive control) and negative for the diluent solution (negative control). The test was considered
positive for d = 3 mm. Wheal area was calculated from the mean
diameter (d) according to the expression: A = p (d2/ 4). Allergen
specific reactivity was calculated relative to the Histamine 10 mg/
mL, using this expression:[30]
Ch10 = (Aa/Ah) 2.5
(where Aa: allergen wheal area; Ah: histamine wheal area).
Pulmonary function was measured through Peak Expiratory Flow
(PEF) with the use of a portable PEF meter (Ferraris, UK). This
measurement was performed daily by the patient at home, whereas
daily PEF variability was calculated by the physician.[31, 32]
A secondary dichotomical variable was used to assess the overall
clinical improvement (OCI) of eachpatient, with two levels:
�better� and �not better�. The �better� value was assigned when
the symptom/medication score was reduced below 60%, 27 with
respect to the baseline and the skin reactivity (Ch10) and lung
function category did not worsen with respect to the beginning
of the treatment.
Safety assessment
Adverse reactions were recorded and classified as local or systemic,
or immediate or delayed.[33] Local reactions were assessed
by erythema diameter. Local immediate reactions with a diameter
of less than 5 cm and delayed reactions of less than 10 cm were
considered mild. Systemic reactions were graded in accordance
with the World Allergy Organization.[34]
Statistical methods
The statistical package GlaxoWellcome C4-SDP was used for the
calculation of the sample size and randomization to intervention
groups. Thus, the sample size needed to obtain a relevant preset
minimum difference, between two proportions from two groups
P1 (Placebo) and P2 (Active), was calculated. The following input
data were used: P2-P1 = 53%; a = 0.05; � = 0.10. Output:
required subjects n = 14. Simple random assignment was used
with equal probability to each study arm: placebo or active. For
data processing and hypothesis testing the statistical package
STATISTICA v.5.0 (Statsoft, USA) was used. Outcome data was
analyzed in a blinded manner at the two planned evaluative points:
6 and 12 months, with all patients, without exclusions. Testing to
normality of variables was assessed by the Kolmogorov-Smirnov
and Shapiro-Wilks tests. Non-parametric methods were applied
to all variables (except height, weight and skin reactivity, Ch10).
Thus, the central values were expressed by the median and its
95% Confidence Interval and comparisons between the Active
and Placebo groups were performed using the Mann-Whitney U
test. Comparisons within each group with respect to pre-treatment
values were performed using the Wilconxon test for paired
samples. For Ch10, a log transformation was performed and, after
verification of its adjustment to normality. The following parametric
methods were used for height, weight and Ch10: t-test for
the comparison between groups; and t-test with paired data for
the comparison within the same group with respect to the baseline
values. For comparison of frequencies between groups the
Pearson's chi-squared test (?2) was used. Finally, the correlation
analysis between the variables was performed using from the nonparametric
Spearman correlation coefficient.
Results
Of 393 patients evaluated with asthma symptoms, presumably related
to indoor allergens, and whoexpressed their consent to participate
in the trial, only 80 patients (20.4%) were selected (Fig. 1).
The major limitation in this sense was the requirement of the preponderant
skin reactivity towards theinvestigated allergens (Dp
and Ds). Roughly 72% of patients showed polysensitization to
the three evaluated species Dp, Ds and Bt, and only 8.6% did not
show a positive response to any of them. In addition to asthma,
most patients also showed rhinitis symptoms: for the Ds trial 30
out of 40 included patients (75%), and for the Dp trial 34 out of
40 patients (85%).
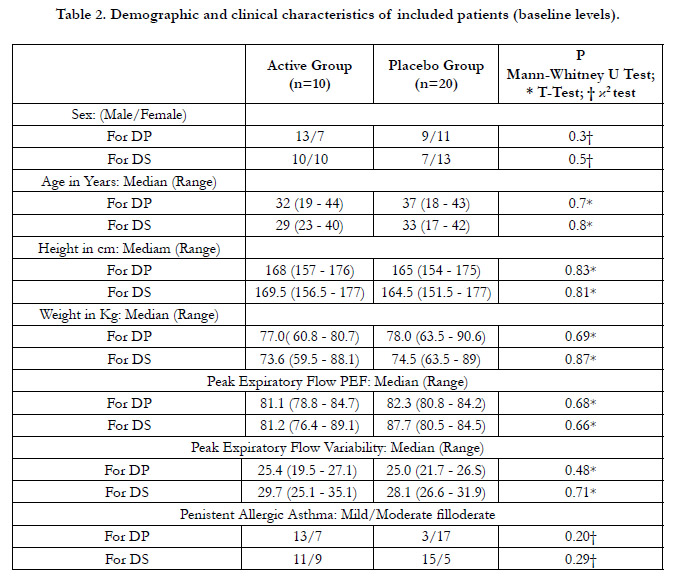
As expected by design, the Active and Placebo groups were comparable
in terms of demographic and clinical variables in both
trials with no statistical differences (p> 0.05) (Table 2). All 80 included
patients completed the trials. No dropouts were recorded
and no patient developed criteria for leaving the study.
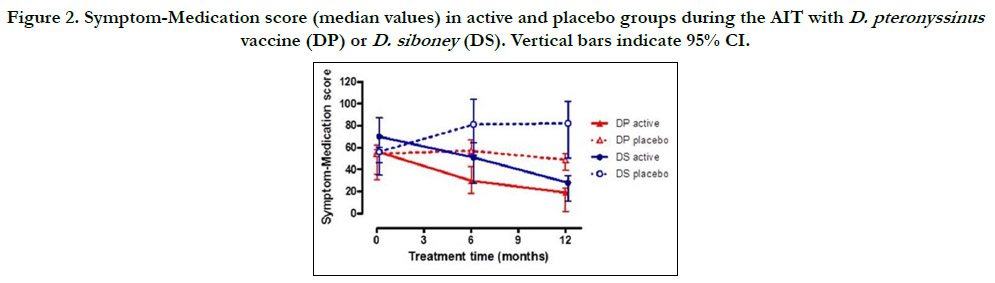
Regarding outcomes, clinical symptoms significantly decreased
down to 36.5% (95% CI: 28-36%) and medication, down to 23%
(95% CI: 18-28%) as compared to placebo values, as an average
of the two trials (p<0.001). The combined symptom/medication
score significantly decreased down to 39% for Dp and 34% for
Ds. The difference between the Active and Placebo groups was
significant (p <0.05) even at 6 months, and more pronounced at
12 months (p<0.001). The decreasing effect was also significant
(p<0.01) as compared to the beginning of the treatment within
the active group (Fig 2). As for lung function in the active group,
an apparent, although non-significant, slight increase in PEF value
was noted: 2.3% and 0.6% for Dp and Ds, respectively (p<0.05).
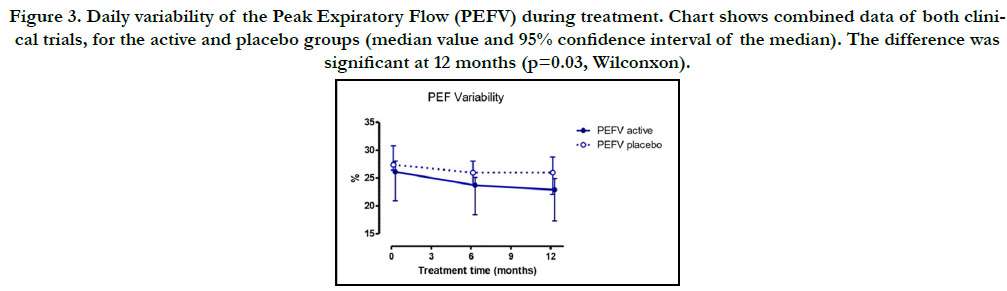
Nevertheless, PEF daily variability did show a significant decrease
(p<0.05) at 12 months when combining data of both trials (Fig. 3)
as well as, when comparing to pretreatment values. On the other
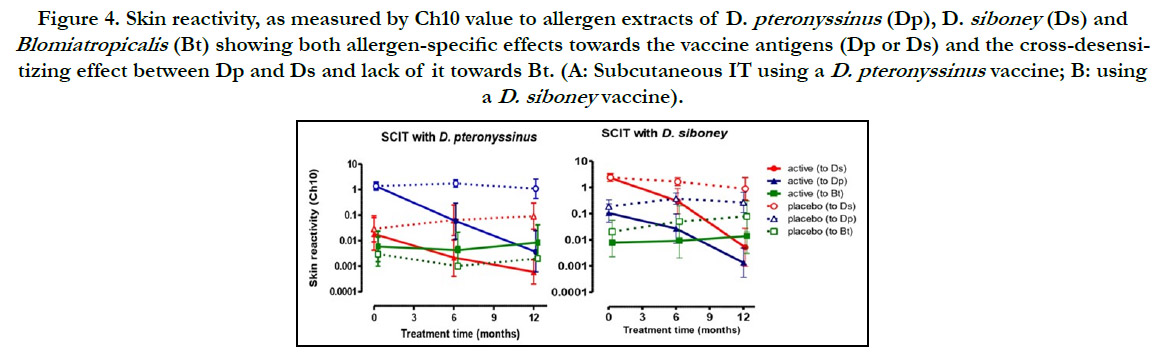
hand, skin reactivity to each allergen did show a remarked decrease.
The Ch10 value decreased significantly (p <0.05), both,
in comparison with the onset and with respect to the placebo
group, even more significant at 12 months (Fig 4). Moreover, the
treatment with Dp induced, not only the reduction of sensitivity
to Dp, but also to Ds and vice versa. In contrast, skin reactivity
towards the mite Blomiatropicalis did not changed. Combining
the results of both trials, most patients (52%) completely abolished
the skin reactivity to the causative allergen (i.e. they showed
no wheals or below 3 mm diameter) or decreased it remarkably
in more of 1 logarithm of Ch10 (78% of patients), i.e, at least a
10-fold increase in the allergen threshold.
According to the Overall Clinical Improvement (OCI) variable,
70% of the patients in the Active group were reported as �better�
relative to the treatment onset, both for Dp and Ds. Overall, OR
of improvement was 28.8 (95%CI: 7.4 to 111.8). The decrease of
skin reactivity to the administered allergens (Dp or Ds) correlated
significantly with the change in primary clinical outcome: the SM
score (r=0.514; p<0.05). Furthermore, there was a significant correlation
(r=0.462; p <0.05) between the decrease in daily variability
of PEF and change of skin reactivity.
During this study 1638 injections were given, 819 in the active group. All patients in the active groupreached the expected maintenance
dose of 6000 BU and the planned treatment duration of
12 months.All adverse events were classified as local immediate
allergic reactions. There were nine and ten reactions for Dp and
Ds, respectively. All these events occurred with the build-up dose
(0.5 mL of vial number 2, 100 BU). The observed local reactions
consisted of pruritus and wheals smaller than 5 cm at the injection
site and were considered mild. No systemic reactions were
reported.
In the Dp trial, the frequency of patients with local adverse reactions
in the Active group was 30 % (6 out of 20). Three patients
showed repeated reactions at two administrations. Only
one patient had a nonspecific local reaction (not associated to the
product), a mild pain and cramp sensation at the site of injection,
which disappeared spontaneously. Therefore, the frequency of patients with reactions attributable to the product was 25% (5
out of 20). The frequency of adverse reactions with respect to the
number of injections administered was 2.2% in the Active group
versus 0.2% with Placebo. Similarly, in the Ds trial, the frequency
of patients with local adverse reactions in the Active group was 30
%. Four patients suffered from repeated reactions at two distinct
administrations. The frequency of patients with reactions linked
to the product was 25%. Finally, the frequency of adverse reactions
with respect to the amount of injections administered was
2.5% in the Active group versus 0.5% with Placebo.
Figure 2. Symptom-Medication score (median values) in active and placebo groups during the AIT with D. pteronyssinus vaccine (DP) or D. siboney (DS). Vertical bars indicate 95% CI.
Figure 3. Daily variability of the Peak Expiratory Flow (PEFV) during treatment. Chart shows combined data of both clinical trials, for the active and placebo groups (median value and 95% confidence interval of the median). The difference was significant at 12 months (p=0.03, Wilconxon).
Figure 4. Skin reactivity, as measured by Ch10 value to allergen extracts of D. pteronyssinus (Dp), D. siboney (Ds) and Blomiatropicalis (Bt) showing both allergen-specific effects towards the vaccine antigens (Dp or Ds) and the cross-desensitizing effect between Dp and Ds and lack of it towards Bt. (A: Subcutaneous IT using a D. pteronyssinus vaccine; B: using a D. siboney vaccine).
Discussion
This work encompasses two clinical trials of allergen immunotherapy
using House Dust Mite extracts of the closely related
species Dermatophagoides pteronyssinus and Dermatophagoides
siboney. The last species is prevalent in Cuba and the Caribbean
and could be considered a tropical evolutionary descent
of D. farinae. This work describes the first double-blind placebo
controlled trial for a D. siboney allergen extract. Both allergen
products Dp and Ds were standardized sharing the same potency.
The dosage schedule, as well as, the study methodology, clinical
site and research team coincided. The study results were highly satisfactory in terms of clinical improvement and, particularly,
of the decrease of allergen specific skin reactivity, as well as, of
the cross-desensitization effect between Ds and Dp. Notably, the
AIT with Ds performed as well, or may be slightly better, than
the well-studied Dp. The decrease of symptoms was of a similar
magnitude that the Standard Mean Difference (SMD) reported
in a well-known meta-analysis of subcutaneous immunotherapy
using HDM allergens of D. pteronyssinus and D. farinae species
(SMD for symptoms: -0.85 (Dp) to -1.30 (Ds) versus -1.13
in the Abramson metaanalysis); whereas the effect on medication
was apparently higher here: SMD for medication: -0.82 (Dp) to
-1.26 (Ds) versus -0.51 in the Abramson review.21 Nevertheless,
it should be noted that medication scores are far to be homogeneous
between different authors.[22]
The results regarding PEF and its variability coincide with that
reported by Abramson in the sense that IT induces small or none
effect on the respiratory function; however, in this case the effect
on PEF variability, although small, was significant.[21] In
contrast, allergen specific skin reactivity decreased dramatically.
The magnitude of this effect, which by the Ch10 definition can
be translated into a 277-fold increase of the allergen threshold
able to elicit a positive skin reaction, was higher than the reported
in most publications.[17, 18, 20, 22] It is notorious that Ds immunotherapy
induced not only reduction of skin reactivity to Ds
extract; but also to Dp and vice versa, which was expected due to
the high cross-reactivity between both Dermatophagoides species,
aspreviously described.[35] As far as we know, this is the first
factual report of this cross-effect during IT between Dp and Ds
obtained in a controlled trial, since Ds is not found outside of the
Caribbean region.[11, 12] As expected, this cross-desensitization
effect was absent for Bt, since this species has only very limited
cross-reactivity to Dermatophagoides.[2] In full agreement with
this finding, a recent work has described the opposite, i.e, the lack
of cross-desensitization effect of AIT using Bt extracts towards
Dp or Ds.[36]
The question of correlation between change of allergen skin reactivity
and clinical improvement has been controversial.[37-39]
Confirming this correlation can be relevant for the proposed
use of skin test as surrogates markers of clinical efficacy during
AIT, a point that has been recently raised by several authors.
[40, 41] This study has, indeed, shown a significant correlation
between skin reactivity and SM score, which supports the use of
carefully measured skin reactivity by means of Ch10 (relative to
Histamine standard) as a marker of efficacy.[42] Lack of relevant
correlations in other studies could be due to the inaccuracy of the
used SPT methodology and poorer performance, or the use of
absolute variable values and not the shift occurred during treatment
period (i.e. the differences between final and initial values).
Moreover, the Ch10 parameter eliminates the non-specific effect
related to Histamine reactivity, thus raising accuracy. In addition,
achievement of significant correlation values can be benefitted
by the large effect noticed in this work for both variables: SM
and Ch10. Skin tests have the advantages of simplicity and relative
safety over other gold-standard diagnostic procedures, such
as bronchial provocation tests, whereas a strong association has
been reported between boths.[39]
Although subcutaneous AIT has been widely used and acknowledged
as an effective treatment, demonstrating clear benefits in
reducing allergic symptoms, safety has been a major concern. In
fact, the main disadvantage of this type of treatment is the occurrence
of adverse events, some of them with a systemic character,
which can be very inconvenient for the patient and, even, pose a
life-threatening risk. In this study, the frequency of adverse reactions
attributable to the product was comparable to that reported
internationally, both with respect to the total number of injections
and to the number of patients.[17, 18, 20, 22]
However, here all reactions were local; noteworthy, no systemic
reactions were observed in this study. It has been reported that
systemic allergic reactions during AIT occurs in approximately 0.1
to 0.2% of injections and roughly 2% to 5% of patients.[18, 32,
33, 43-45] However, the number of patients in this study may
be too small to support conclusions on this issue. Factors favoring
minimization of systemic reactions in this study could be: the
accurate standardization of the allergen extracts, proper patient
selection and careful clinical examination prior to the injection
administration, as well as, surveillance during the injection procedure.[
17, 18, 20, 22, 44]
The current study was performed in adults with a range of ages
from 17 to 45 years in a population sample representative of
Cuban adult allergic asthmatic population. Asthma is the most
relevant allergic disease in terms of social and healthcare impact
worldwide; nevertheless, HDM sensitization is also a common
cause for allergic rhinitis. In agreement with this fact, rhinitis was
a frequent comorbidity in asthmatic patients recruited to these
clinical trials. The design of these trials addressed only asthma
endpoints, therefore, no data was collected regarding the evolution
of rhinitis symptoms, although it is likely that the same also
improved during the treatment. More studies should be conducted
to expand and confirm these results, particularly in children.
Summarizing, AIT with D. pteronyssinus and, remarkably, D. siboney
standardized allergen extracts, was effective and safe for the
control and amelioration of the allergic asthma in the Cuban
population commonly exposed to these allergens. This is the first
published report worldwide of AIT using D. siboney allergens.
Subcutaneous AIT should be applied in specialized allergy/immunology
services, taking into account internationally recommended
safety standards.
List of abbreviations
AIT: Allergen-Specific Immunotherapy
Dp: Dermatophagoides Pteronyssinus
Ds: Dermatophagoides Siboney
SCIT: Subcutaneous Immunotherapy
BU: Biological Units
PEF: Peak expiratory Flow
HDM: House Dust Mites
Bt: Blomiatropicalis
CECMED: Cuban Regulatory Authority
GINA: Global InitiativeforAsthma
BIOCEN: Centro Nacional de Biopreparados
SM: Symptom and Medication
PEF: Peak Expiratory Flow
OCI: Overall Clinical Improvement
SMD: Standard Mean Difference
PEFV: Daily variability of the Peak Expiratory Flow
Declarations
Ethics Approval and Consent to Participate
The study was conducted following the ethical principles contained
in the Declaration of Helsinki (64th WMA General Assembly,
Fortaleza, Brazil, October 2013. Subjects were asked for
their written informed consent to participate in the study. The
study was approved by the Ethics Committee of the "Calixto
Garc�a" University Hospitaland was approved by the Cuban medicine
regulatory authority (CECMED).
Consent for publication
All authors gave its written consent for publication.
Availability of data and materials
Please contact author for primary data requests.
Conflict interests
The authors have no conflicts of interest.
Funding
The research work was funded by the Cuban government: Ministry
of Science, Technology and Environment and Ministry of
Public Health. Forum SYD and Swedish International Development
Cooperation Agency contributed to partially funding this
work at the early stages.
Authors' contributions
Contributions of authors to the manuscript:
Ra�l L�zaro Castro-Almarales (AD, AE, CI, DE, TM).
Mercedes Ronquillo-D�az (AD, AE, CI, DE, IE).
Mirta �lvarez-Castell� (AD, AE, CI, DE, IE)
Jos� Rodr�guez-Canosa (AD, DE, CI, IE)
Mayda Gonz�lez-Le�n (AD, DE, CI, IE)
Irene Enr�quez-Dom�nguez (AD, DE, CI, IE)
B�rbara Ivonne Navarro-Viltre (AD, DE, CI, IE)
Mayte� Mateo-Morej�n (AD, DE, IE)
Yunia Oliva-D�az (AD, DE, IE)
Wendy Ram�rez-Gonz�lez(AD, DE, IE)
Alexis Labrada-Rosado, PhD (AD, AE, CI, DE)
AD: Acquisition of data, or analysis and interpretation of data.
AE: Performed the statistical analysis.
CI: Have been involved in drafting the manuscript or revising it
critically for important intellectual content.
DE: Conceived of the study, and participated in its design and performed of study.
IE: carried out the immunoassays and skin tests.
TM: Trial monitoring.
Acknowledgements
To Professor StenDreborg for its valuable training and advice in
Skin Tests and Immunotherapy, and to Forum SYD and Swedish
International Development Cooperation Agency.
References
- Pawankar R, Canonica GW, Holgate ST, Lockey RF. WAO White Book on Allergy 2011-2012.
- Caraballo L, Zakzuk J, Lee BW, Acevedo N, Soh JY, S�nchez-Borges M, et al. Particularities of allergy in the Tropics. World Allergy Organ J. 2016 Jun 27;9:20. Pubmed PMID: 27386040.
- Mallol J, Crane J, von Mutius E, Odhiambo J, Keil U, Stewart A. ISAAC Phase Three Study Group. The International Study of Asthma and Allergies in Childhood (ISAAC) Phase Three: a global synthesis. Allergol Immunopathol (Madr). 2013 Mar-Apr;41(2):73-85. Pubmed PMID: 22771150.
- Marks G, Pearce N, Strachan D, Asher I. The Global Asthma Report 2014.
- D�az-Rodr�guez A. Allergology in Cuba. General overview and future perspectives. Junior Member News 2013.
- Mallol J, Sol� D, Baeza-Bacab M, Aguirre-Camposano V, Soto-Quiros M, Baena-Cagnani C. Latin American ISAAC Group. Regional variation in asthma symptom prevalence in Latin American children. J Asthma. 2010 Aug;47(6):644-50. Pubmed PMID: 20642377.
- Diaz Rodriguez A, Rosado AL, Almarales RL, Castell� M�. Current status and future perspectives of immunotherapy in Latin America and Cuba. World Allergy Organ J. 2014 Nov 12;7(1):28. Pubmed PMID: 25829980.
- Fabr� Ortiz D, Varona P�rez P, Su�rez Medina R. Prevalencia de asma en adolescentes cubanos por diferentes criterios cl�nicos [Prevalence of asthma in Cuban adolescents by different clinical criteria]. Rev Alerg Mex. 2005 May-Jun;52(3):132-7. Spanish. Pubmed PMID: 16158787.
- de Salud AE. Direcci�n Nacional de Registros M�dicos y Estad�sticas de Salud. La Habana: Ministerio de Salud P�blica. 2011.
- Varona P�rez P, Garc�a Roche R, D�az-Perera Fern�ndez G, Mar�a G�lvez A, Bonet Gorbea M, Gonz�lez C, Van Der Stuyft P. Uso de servicios de salud y gastos de bolsillo por asma en ni�os de 5 a 14 a�os de edad: Ciudad de La Habana, 2002. Revista Cubana de Higiene y Epidemiolog�a. 2005;43(3):38- 41.11.
- van Hage-Hamsten M. Dermatophagoides siboney and Blomia tropicalis-- dust mites of subtropical and tropical areas. Clin Exp Allergy. 1995 Oct;25(10):905-7. Pubmed PMID: 8556557.
- Fern�ndez-Duro BI, Cuervo-Pineda N, Rodr�guez-Alv�zar JA, Celio-Murillo R, Ju�rez-Anaya D, P�rez-Ortiz TM. Identificaci�n de la fauna de �caros del polvo dom�stico en colchones de pacientes al�rgicos de seis estados mexicanos [Identification of house dust mite fauna from allergic patients' mattress in six Mexican states]. Rev Alerg Mex. 2013 Jul-Sep;60(3):87-92. Spanish. Pubmed PMID: 24274602.
- Ferrandiz R, Casas R, Dreborg S. Sensitization to Dermatophagoides siboney, Blomia tropicalis, and other domestic mites in asthmatic patients. Allergy. 1996 Jul;51(7):501-5.
- Cuervo N, Dusbabek F, de la Cruz J, Abreu R. Los �caros (acarina: Pyroglyphidae, Cheyleitidae, Saproglyphidae y Glycyphagidae) de los polvos dom�sticos en Cuba [Acarids (Acarina: Pyroglyphidae, Cheyletidae, Saproglyphidae and Glycyphagidae) of house dust in Cuba]. Rev Cubana Med Trop. 1983 Feb-Apr;35(1):83-103. Spanish. PMID: 6351195.
- Astro Almarales RL, Mateo Morej�n M, Naranjo Robalino RM, Navarro Viltre BI, Alvarez Castell� M, Ronquillo D�az M, Garc�a G�mez I, et al. Correlation between skin tests to Dermatophagoides pteronyssinus, Dermatophagoides siboney and Blomia tropicalis in Cuban asthmatics. Allergol Immunopathol (Madr). 2006 Jan-Feb;34(1):23-6. Pubmed PMID: 16540067.
- Ferr�ndiz R, Casas R, Dreborg S, Einarsson R, Fern�ndez B. Crossreactivity between Dermatophagoides siboney and other house dust mite allergens in sensitized asthmatic patients. Clin Exp Allergy. 1995 Oct;25(10):929-34. Pubmed PMID: 8556563.
- Bousquet J, Lockey R, Malling HJ. Allergen immunotherapy: therapeutic vaccines for allergic diseases A WHO position paper. Journal of Allergy and Clinical Immunology. 1998 Oct 1;102(4):558-62.
- Cox L, Nelson H, Lockey R, Calabria C, Chacko T, Finegold I, Nelson M, et al. Allergen immunotherapy: a practice parameter third update. J Allergy Clin Immunol. 2011 Jan;127(1):S1-55. Pubmed PMID: 21122901.
- Akdis CA, Akdis M: Mechanisms of allergen-specific immunotherapy. J Allergy ClinImmunol 2011, 127, 1:18-27.
- Burks AW, Calderon MA, Casale T, Cox L, Demoly P, Jutel M, Nelson H, et al. Update on allergy immunotherapy: American Academy of Allergy, Asthma & Immunology/European Academy of Allergy and Clinical Immunology/ PRACTALL consensus report. J Allergy Clin Immunol. 2013 May;131(5):1288-96.e3. Pubmed PMID: 23498595.
- Abramson MJ, Puy RM, Weiner JM. Injection allergen immunotherapy for asthma. Cochrane Database Syst Rev. 2010 Aug 4;(8):CD001186. Pubmed PMID: 20687065.
- Jutel M, Agache I, Bonini S, Burks AW, Calderon M, Canonica W, Cox L, et al. International Consensus On (ICON). Allergy Immunotherapy (AIT). J Allergy ClinImmunol. 2015;136, 3:556-68.
- Jim�nez G, Pascual MA, Fors M, Cobas J, Torres A, Cachimaille Y, Rodr�guez J et al. The Cuban Public Registry of Clinical Trials: primary registry of World Health Organization. J Evid Based Med. 2011;4(3):161-4. Pubmed PMID: 21894615.
- Nordic Council on Medicines. Registration of allergen preparations: Nordic Guidelines. NLN Publication. 1989;23:1-34.
- Labrada Rosado A. Desarrollo a ciclo completo de las primeras vacunas estandarizadas de alergenos de �caros para la inmunoterapia del asma en Cuba. Tesis de Doctor en Ciencias de la Salud. Instituto Superior de Ciencias M�dicas de La Habana, 2009. Editorial Universitaria, La Habana 2012. ISBN 978-959-16-1711-8 � 193.
- Koshak EA. Classification of asthma according to revised 2006 GINA: Evolution from severity to control. Annals of thoracic medicine. 2007 Apr;2(2):45.
- Malling HJ. Criteria for clinical efficacy--readout and monitoring of clinical studies. Arbeiten aus dem Paul-ehrlich-institut (Bundesamt fur Sera und Impfstoffe) zu Frankfurt aM. 2003 Jan 1(94):119-23.
- Canonica GW, Baena-Cagnani CE, Bousquet J, Bousquet PJ, Lockey RF, Malling HJ, Passalacqua G, et al. Recommendations for standardization of clinical trials with Allergen Specific Immunotherapy for respiratory allergy. A statement of a World Allergy Organization (WAO) taskforce. Allergy. 2007 Mar;62(3):317-24. Pubmed PMID: 17298350.
- Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions v4.2.6 Sept 2006.
- Dreborg S. Skin testing. The safety of skin tests and the information obtained from using different methods and concentrations of allergen. Allergy. 1993 Oct;48(7):473-5. Pubmed PMID: 8238806.
- Lebowitz MD. The use of peak expiratory flow rate measurements in respiratory disease. Pediatric pulmonology. 1991;11(2):166-74.
- Quackenboss JJ, Lebowitz MD, Krzyzanowski M. The normal range of diurnal changes in peak expiratory flow rates. Relationship to symptoms and respiratory disease. Am Rev Respir Dis. 1991 Feb;143(2):323-30. Pubmed PMID: 1990947.
- Cox L, Larenas-Linnemann D, Lockey RF, Passalacqua G. Speaking the same language: The World Allergy Organization Subcutaneous Immunotherapy Systemic Reaction Grading System. J Allergy Clin Immunol. 2010 Mar;125(3):569-74, 574.e1-574.e7. Pubmed PMID: 20144472.
- Cox LS, Sanchez-Borges M, Lockey RF. World Allergy Organization Systemic Allergic Reaction Grading System: Is a Modification Needed? J Allergy Clin Immunol Pract. 2017 Jan-Feb;5(1):58-62.e5. Pubmed PMID: 28065342.
- Ferr�ndiz R, Casas R, Dreborg S. Cross-reactivity between Dermatophagoides siboney and other domestic mites. II. Analysis of individual crossreacting allergens after SDS-PAGE and Western blotting inhibition. Int Arch Allergy Immunol. 1998 Jul;116(3):206-14. Pubmed PMID: 9693268.
- Castro-Almarales RL, Ronquillo-D�az M, �lvarez-Castell� M, Rodr�guez- Canosa J, Gonz�lez-Le�n M, Enr�quez-Dom�nguez I, Navarro-Viltre BI, et al. Subcutaneous allergen immunotherapy for asthma: A randomized, double-blind, placebo-controlled study with a standardized Blomia tropicalis vaccine. World Allergy Organ J. 2020 Apr 10;13(4):100098. Pubmed PMID: 32308779.
- Calderon MA, Casale TB, Nelson HS, Demoly P. An evidence-based analysis of house dust mite allergen immunotherapy: a call for more rigorous clinical studies. J Allergy Clin Immunol. 2013 Dec;132(6):1322-36. Pubmed PMID: 24139829.
- Lee E, Kim MJ, Yang SI, Yu J, Hong SJ. Comparison of short-term effects between subcutaneous and sublingual immunotherapies in children with house dust mite-sensitized allergic rhinitis and asthma. Allergy, Asthma & Respiratory Disease. 2015 May 1;3(3):180-6.
- Son JY, Jung MH, Koh KW, Park EK, Heo JH, Choi GS, Kim HK. Changes in skin reactivity and associated factors in patients sensitized to house dust mites after 1 year of allergen-specific immunotherapy. Asia Pac Allergy. 2017 Apr;7(2):82-91. Pubmed PMID: 28487839.
- Cox LS. Sublingual Immunotherapy for Allergic Rhinitis: Is 2-Year Treatment Sufficient for Long-term Benefit? JAMA. 2017 Feb 14;317(6):591- 593. Pubmed PMID: 28196239..
- Scadding GW, Calderon MA, Shamji MH, Eifan AO, Penagos M, Dumitru F, Sever ML, et al. Immune Tolerance Network GRASS Study Team. Effect of 2 Years of Treatment With Sublingual Grass Pollen Immunotherapy on Nasal Response to Allergen Challenge at 3 Years Among Patients With Moderate to Severe Seasonal Allergic Rhinitis: The GRASS Randomized Clinical Trial. JAMA. 2017 Feb 14;317(6):615-625. Pubmed PMID: 28196255.
- Dreborg S. Allergen skin prick test should be adjusted by the histamine reactivity. Int Arch Allergy Immunol. 2015;166(1):77-80. Pubmed PMID: 25765942.
- Epstein TG, Liss GM, Murphy-Berendts K, Bernstein DI. AAAAI/ACAAI surveillance study of subcutaneous immunotherapy, years 2008-2012: an update on fatal and nonfatal systemic allergic reactions. J Allergy Clin Immunol Pract. 2014 Mar-Apr;2(2):161-7. Pubmed PMID: 24607043.
- Kowalski ML, Ansotegui I, Aberer W, Al-Ahmad M, Akdis M, Ballmer-Weber BK, Beyer K, et al. Risk and safety requirements for diagnostic and therapeutic procedures in allergology: World Allergy Organization Statement. World Allergy Organ J. 2016 Oct 12;9(1):33. Pubmed PMID: 27777642.
- Epstein TG, Liss GM, Murphy-Berendts K, Bernstein DI. Risk factors for fatal and nonfatal reactions to subcutaneous immunotherapy: National surveillance study on allergen immunotherapy (2008-2013). Ann Allergy Asthma Immunol. 2016 Apr;116(4):354-359.e2. Pubmed PMID: 26948485.