Urticaria and Atopic Dermatitis Related to Anisakis Simplex
Sugiura K*,Sugiura M
Department of Environmental Dermatology and Allergology, Daiichi Clinic, Nittochi Nagoya Bld. 2F, 1-1 Sakae 2, Nakaku, Nagoya, 468-0008, Japan.
*Corresponding Author
Keiji Sugiura,
Department of Environmental Dermatology and Allergology,
Daiichi Clinic, Nittochi Nagoya Bld. 2F,
1-1 Sakae 2, Nakaku, Nagoya,468-0008, Japan.
E-mail: ksguira@daiichiclinic.jp
Article Type: Research Article
Received: May 26, 2014; Accepted: June 21, 2014; Published: June 25, 2014
Citation: Sugiura K, Sugiura M (2014) Urticaria and Atopic Dermatitis Related to Anisakis Simplex. Int J Clin Dermatol Res. 2(3), 14-18, doi: dx.doi.org/10.19070/2332-2977-140004
Copyright: Keiji Sugiura© 2014. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Anisakis simplex (AS) is a parasite in seafood and this nematode is a known risk factor for gastro anisakiasis and dermatoses with itching, like urticaria and angioedema. Recently, Japanese cuisine received a UNESCO Intangible Cultural Heritage of Humanity designation. The number of people consuming raw fish is increasing around the world, and so too are diseases and dermatoses related to AS. We speculated that some cases of urticaria are due to AS allergy rather than being simple atopic dermatitis (AD) cases. In this study, we investigated the sensitization profile of specific IgE against AS in urticaria and AD.
Object and methods: We investigated gender, age, score of specific IgE to AS, WBC and eosinophils in patients with urticaria, atopic
dermatitis, or both diseases.
Results: Our research produced the following interesting results: (1) more male than female patients had a positive score to AS; (2) the
number of patients in their 50s and 60s with positive AS score was significantly higher than that for other ages; (3) there were a larger
number of AD patients than urticaria patients with a positive score to AS.
Conclusion: AS should be considered in some cases of AD. Determining IgE score to AS is useful for treatment of AD and urticaria,
especially in older male patients with these diseases.
2.Introduction
3.Patients and Methods
3.1.Patients
3.2.Methods
4.Statistical analysis
5.Results
6.Discussion
7.Conclusion
8.References
Keywords
Urticaria; Atopic Dermatitis; Anisakis; Specific Ige; Parasite.
Introduction
In 2013, Washoku (Japanese traditional cuisine) was added to UNESCO’s Intangible Cultural Heritage list. As a result, more people than ever before—not only Japanese but also foreign people— would like to become an Itamae (washoku cooker). With washoku becoming more popular around the world, the number of people consuming raw fish is increasing. Japanese and Spanish people often eat raw fish (ex. sushi, sashimi, vinagreta), and some experience urticaria or itching afterwards. An anisakis allergy is one potential cause of these symptoms. A traditional Japanese proverb says, “Eating fish that looks blue or shiny sometimes may cause urticaria or itching”. This means that fish looking blue or shiny are often infected by the parasitic nematode, Anisakis simplex (AS), and eating these fish often cause dermatoses with itching (AS allergy). AS parasites infect fish and other seafood (mackerel, sardines, anchovies, tuna, squid, etc.), and are well known to cause allergic reactions in Japan and Spain.
The AS nematode is about 2-3 cm long, and white in color; usually, fish eat the parasite as larva. Larva cannot reach the imago stage in humans, so most are excreted. Often, however, they invade theb mucosa, causing gastric symptoms (ex. stomachache, nausea and vomiting) and allergic reactions. Previous studies found the frequency of chronic urticaria to be 0.1% of the population [1], and 1% of the general population [2]. Urticaria among persons with allergic disease was shown to be 15-25% [3]; of these, 2.5-2.9% represented the prevalence of urticaria [4], while 22% of outpatients in Spain had specific IgE to anisakis [5]. There have been reports of allergic reactions by AS, and of specific IgE to AS [6- 13]. We hypothesized that the number of urticaria patients with AS allergy would be higher than that of AD, and the score to AS in urticaria was high. We determined serum IgE against AS in the dermatoses, and presented the features and relationships between anisakis and urticaria, as well as between AD and both diseases.
Patients and Methods
A total of 516 outpatients who visited our clinic between October 2004 and December 2013 were included in the study. Of these, 329 were female and 187 male. Patient age ranged from 0–87 years, with an average age of 36.91 years old. The average age of females was 36.15 years, while that of males was 38.42 years. Of the 516 cases, 258 cases were of urticaria, 226 cases of AD, and 32 patients had both diseases.
Data was collected on subjects’ specific IgE to AS, white blood cells (WBC), eosinophils (Eo), age, and gender. Specific IgE to AS of 0.34 UA/ml or less was converted to a CAP RAST score 0; 0.35-0.70 UA/ml was scored as 1; 0.71-3.50 UA/ml was scored as 2; 3.51-17.50 UA/ml was scored as 3; 17.51-50.00 UA/ml was scored as 4; 50.01-100.00 UA/ml was scored as 5; and 100.01UA/ml and more was scored 6. Specific IgE to AS 0.71 UA/ml and above (a score of 2 or more) was considered a positive result. Over 5.0% of Eos were positive, and less than 4.9% were negative; meanwhile, over 8000/mm3 of WBC were positive.
Statistical analysis
Statistical analyses were performed using t-test; P<0.5 was considered statistically significant.
Results
We obtained one significant and two interesting results: (1) the number of patients in their 50s and 60s with a positive score to AS was significantly higher than for other age groups; (2) a higher number of male than female patients had a positive score to AS; and (3) AD patients had the highest number of positive scores to AS in the study.
A total of 401 patients had a score of 0, and 39 patients a score of 1 for specific IgE to AS; these subjects were classified as negative. The frequency of patients with a score of 2 or higher was 14.55% (76 cases) (Table 1). In females, 36 out of 344 cases (10.46 %) had a score of 2 or more, as did 40 out of 172 cases in males (23.25%) (Table 1). The positive ratio of specific IgE score to AS in subjects in their 50s (10/38 cases; 26.31%) and 60s (8/35 cases; 22.85%) were higher than for other ages (Figure 1).
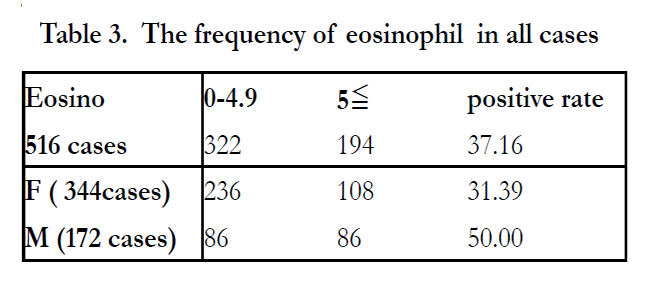
Analyses of the dermatoses and specific IgE score to AS found that frequency of cases with positive score to AS was 17.256% (39/226 cases) in AD, 12.4% (32/258 cases) in urticaria, and 15.625% (5/32 cases) with both diseases (Table 2). In considering disease, gender, and class, the highest ratio of positive score to AS (38.88%) was seen in males with AD (Figure 2). A total of 194 cases (37.16%) had a positive Eo number (Table 3). Of these 194 cases, 108 were female were (of 344 female Eo cases; 31.39%) and 86 were male (of 172 Eo cases; 50.0%; (Table 3). A total of 135 cases (59.73%) in AD had positive Eos, and 50 cases (19.38%) of urticaria were positive Eos; there were no patients with positive Eos in both diseases. Average fractionation of Eo was 6.81% in AD, 3.26% in urticaria, and 3.93% in both. In 258 patients with urticaria, 32 cases had a positive score to AS, and 8 out of 32 cases had 5% or more Eos (Table 4). In 226 AD cases, there were 39 cases with positive score to AS, and 27 of these cases were 5% or more Eos (Table 4). In patients with both diseases, there were 5 cases with a positive score to AS, and all had 4.9% or less Eo (Table 4). In patients with a score of 2 of IgE to AS, 27 had a positive Eos larger than patients with 4.9% or less of Eos. The rate of positive specific IgE score to AS in the patients with 8000/mm3 or more of WBC was higher than for those with 8000/mm3 or less of WBC. Patients with class 3 of specific IgE to AS possessed the highest number of WBC and rate of Eos (Table 5). Age, WBC and Eos of patients with a positive specific IgE score to AS were higher than for those with a negative score (Table 6).
Figure 2. Gender and dermatoses AD: atopic dermatitis, U: urticaria, AU: atopic dermatitis and urticaria, M:male, F: female
Discussion
Anisakis simplex (AS) is parasitic nematode that infects fish. Previous studies reported the rate of anisakis infection in mackerel was 94-100% in Japan [14], and 40-80% (depending on the species) in Spain and other European countries [15]. AS does not infect most fish cultured in aquatic plants, except for those fed raw fish. The custom of consuming raw fish (as sushi, sashimi, or vinagreta) is common in Japan and Spain, as are gastric anisakiasis and other anisakis-related diseases [16-20]. As transportation technology has evolved and Japanese cuisine has become more popular with the UNESCO Intangible Cultural Heritage designation, raw fish consumption worldwide is on the rise, and cases of anisakis-related disease are likely to increase.
The most common anisakis-related cutaneous disease is urticaria. Since the 1980s, many cases of allergic reactions by anisakis and the results of specific IgE to anisakis have been reported [6-9]. There have been various reports about the positive ratio of specific IgE to anisakis. One Spanish report found that 4.7% of normal subjects had specific IgE to AS [21]; a second reported that 15.23% of healthy donors had specific IgE to AS [22], while a third found that 22% of Spanish people had specific IgE to anisakis [5]. Yet another report detected occupation-related data specific to IgE to AS in 20.2% of professional populations [23]. We speculated that the frequency of positive IgE score to AS might be higher in urticaria patients of older age, but this was not borne out by actual data. The results of our study showed that 14.55% patients (76 out of 516 cases) had positive IgE score to AS, and this result was not high compared to other data.
The number of patients with positive IgE score to AS was as follows: AD, urticaria and both were 17.256% (39 cases), 12.4% (32 cases) and 15.625% (5 cases), respectively. It is unclear why the ratios of AD patients with positive IgE score to AS were higher than that for urticaria or both diseases. There have been reports about cross-reactivity between AS and both dust mites [24,25] and chironomids [22]. Reports on this cross-reactivity between AS and dust mites suggested certain proteins, including 35-50kd and 100kd were responsible [25]. One reason for the highest ratio of positive IgE score to AS in AD patients may have been cross-reactivity to other allergens. Recently, 13 AS allergens have been established, with allergens detected varying depending on country and cases. Anisakis 3, a notable allergen, is a tropomyosin and could be related to cross-reactivity. All animals contain tropomyosins, and the tropomyosins of invertebrate animals in particular are recognized as important and major allergens [26]. Because tropomyosins are similar to the molecules in dust-mites [27] and believed to possess cross-reactivity with anisakis, shrimp, cockroach and mites [22,24,28,29] some AD patients have crossreactivity between AS and mites.
In the case of treatment for AD patients, determination of specific IgE against AS is helpful. If patients have positive IgE score to AS, it is important to recommend that patients not eat raw seafood. As to why the prevalence of positive IgE score to AS was significantly higher in patients in their 50s and 60s, previous studies found that Anisakiasis were frequent in Japanese people between 20-50 years [30], and that IgE sensitization to AS increases with age [31,32]. The cases of patients sensitized to AS were higher in patients aged >65 than that those aged 65 > [2]. The mean age of AS allergy was older than that of food allergy, and the most frequent age of IgE-mediated allergy development was middle age or older in non-atopic patients [31]. Japanese people 20-50 years old eat fish as much as older people, and they eat foods from western countries. In the case of older patients with itching, determination of specific IgE score to AS is meaningful for patients. The question of why positive IgE score to AS was more prevalent in males than in females is more difficult. There are a number of possible answers, including: (1) males often eat raw fish at business events, and gastro-anisakiasis is higher in males than in females; (2) most fishermen are male; and (3) generally, males eat a larger volume of food than females. While we did not find evidence for these answers, we nevertheless recommend determining IgE score to AS for male patients with AD, urticaria or itching when determining treatment.
Conclusion
Anisakis simplex is an important allergen, and may be a hidden allergen in allergic food reactions. The prevalence of positive IgE score to AS was not high, but physicians should recommend that patients with AS allergy avoid raw fish. More research is needed to determine specific IgE against AS in not only urticaria but also AD patients, especially those who are male and older.
References
- Greaves M (2000). Chronic urticaria. J Allergy Clin Immunol 105: 664- 672.
- Ventura MT, Napolitano S, Menga R, Cecere R, Asro R(2013). Anisakis simplex hypersensitivity is associated with chronic urticaria in endemic areas. Int Arch Allergy Immunol 160: 297-300.
- Meynadier J, Meynadier JM (1990). Urticares. Dermatologie et Vénéréologie,Paris, Masson.
- Gaig P, Olona M, Muñoz Lejarazu D, Caballero MT, Dominguez FJ, et al (2004). Epidemiology of urticaria in Spain. J Investig Allergol Clin Immunol 14: 214-220.
- Maria Pilar Lopez-Saez, JM Zubeldia, M Caloto, S Olalde, R pelta, et al (2003). Is anisakis simplex responsible for chronic urticaria? Allergy and Asthma Proc 24: 339-345.
- Alonso A, Daschner A, Moreno-Ancillo A (1997). Anaphylaxis with anisakis simplex in the gastric mucosa. N Engl J Med 337: 351-352.
- Audicana MT, Fernández de Corres L, Muñoz D, Fernández E, Navarro JA, et al (1995). Recurrent anaphylaxis caused by anisakis simplex parasitizing fish. J Allergy Clin Immunol 96: 558-560.
- Sugimachi K, Inokuchi K, Ooiwa T, Fujino T, Ishii Y (1985). Acute gastric anisakiasis. Analysis of 178 cases. JAMA 253: 1012-1213.
- A Moreno-Ancillo, MT Caballero, R Cabañas, J Contreras, JA Martin-Barroso, et al (1997). Allergic reactions to anisakis simplex parasitizing seafood. Ann Allergy, Asthma & immunology 79: 246-250.
- A Daschner, Christina-Yolanda P.(2005) Anisakis simplex: sensitization and clinical allergy. Curr Opin Allergy Clin Immunolol 5: 281-285.
- Miguel GM, Rosario L, Frans N, Ignacio M (2005). Deection of anisakis simplex-induced basophil activation by flow cytometry. International Society for analytical cytology 68B: 31-36.
- Fernando G, Juan GB, Mar Garces, Sonsoles J, Mercedes F, et al. (2001) Frezing protects against allergy to anisakis simplex. J invest allergol clin immunol 11: 49-52.
- H Falcao, N Lunet, E Neves, I iglesias, H Barros (2008). Anisakis simplex as a risk factor for relapsing acute urticaria: a case- control study. J Epidemiol Community Health 62: 634-637.
- Murata I (1992). Detection rate of anisakis larvae, paragonimus miyazakii and Gnathostoma hispidum from several kinds of fish and shellfishes at Tokyo Central Market (1985-1990). Jpn J Parasitol 41: 80-81.
- Sanmartín ML, Quintero P, Iglesias R, MT Santamaria, J Leiro (1994). Nemátodos parásitos en peces de las costas gallegas. Ed. Díaz de Santos, S.A., Madrid (España).
- S Kasuya, H Hamano, S Izumi (1990). Mackerel-induced urticaria and anisakis. Lancet 335: 665.
- Oshima T, Kliks MM (1987). Effects of marine mammal parasites on human health. Int J Parasitol 17: 415-21.
- Armentia A, Martin=Gil FJ, Pascual C, Martín-Esteban M, Callejo A, et al (2006). Anisakis simplex allergy after eating chiken meat. J Investig Allergol Immunol 16: 258-263.
- Pecquet C, Danis M, Leynadier F (2002). Anisakis simplex and Immediate hypersensitivity reactions. Ann Dermatol Venereol 129: 303-305.
- Nieuwenhuizen N, Lopata Al, Jeebhay MF, Herbert DR, Robins TG, et al(2006). Exposure to the fish parasite Anisakis causes allergic airway hypersensitivityand dermatitis. J Allergy Clin Immunol 117: 1098-1105.
- Garcia-Palacios L, Gonzalez ML, Esteban MI, Mirabent E, Perteguer MJ, et al (1996). Enzyme-linked immunosorbent assay, immunoblot analysis and RAST fluoroimmunoassay analysis of serum responses against crude larval antigens of anisakis simplex in a Spanish random population. J Helminthol 70: 281-289.
- ascul CY, Crespo JF, San Martin SS, Ornia N, Ortega N, et al (1997). Cross-reactivity between IgE-binding proteins from anisakis, German cockroach, and chironomids. Allergy 52: 514-520.
- Mazzucco W, Lacca G, Cusimano R, Proverzani A, Costa A, et al (2012). Prevalence of sensitization to Anisakis simplex among proffessinally exposed populations in Sicily. Arch Environ Occup Health 67: 9-17.
- Johansson E, Aponno M, Lundberg M, van Hage-Hamsten M (2001). Antigenic cross-reactivity between the nematode anisakis simplex and the dust mites acarus siro, lepidoglyphus destructor, tyrophagus putrescentiae and dermatophagoides pteronyssinus. Allergy 56: 660-666.
- Bernardini R, Mistrello G, Novembre E Roncarolo D, Zanotta S, Lombardi E, et al (2005). Cross-reactivity between IgE-binding proteins from anisakis simplex and Dermatophagoides pteronyssnus. Int J Immunopathol Pharmacol 18: 671-675.
- Reese G, Ayuso R, Lehrer SB (1999). Tropomyosin: An Invertebrate Pan- Allergen. Int Arch Allergy Immunol 119: 247-258.
- Fitzsimmons CM, Falcone FH, Dunne DW (2014). Hel,inth Allergens, Parasite-Specific IgE, and Its Protective role in human immunity. Fron Immunol 2014, 14: 5: 61 ecollection 2014 review.
- Santos ARB, Chapman MD, Aalberse RC, Vailes LD, Ferriani VPL, et al(1999). Cockroach allergens and asthma in Brazil: Identification of tropomyosin as a major allergen with potential cross-reactivity with mite and shrimp allergens. JAllergy Clin Immunol 104: 329-337.
- Ayuso R, Reese G, Leong-Kee S, Plante M, Lehrer SB (2002). Molecular basis of arthropod cross-reactivity: IgE-binding cross-reactive epitopes of shrimp, house dust mite and cockroach tropomyosins. Int Arch Allergy Immunol 129: 38-48
- Smith JW, R Wootten (1978). Anisakis and anisakiasis. Adv Parasitol 16: 93-163
- MD Del Pozo, M Audicana, JM Diez, D Munoz, IJ Ansotegui, E Fernandez, et al (1997). Anisakis simplex, a relevant etiologic factor in acute urticaria. Allergy 52: 576-579.
- Fernández de Corres L, Audícana M, del Pozo MD, Muñoz D, Fernández E, et al (1996). Anisakis simplex induces not only anisakiasis: report on 28 cases of allergy caused by this nematode. J Investig Allergol Clin Immunol 6: 315-319.