Paravertebral Block: A Comparative Study Evaluating Performance of Direct Surgical And Classic Technique
Ahmad R1*, Ahad B2, Zahoor SA3, Bhat MA4
1 Department of Anaesthesia and Critical care, Sher-i-Kashmir Institute of Medical Science, Jammu and Kashmir, Soura, Srinagar, India.
2 Department of Anaesthesia and Critical Care, Sher-i-Kashmir Institute of Medical Science, Medical College and Hospital, Bemina, Jammu and Kashmir, Bemina, Srinagar, India.
3 Department of Cardiovascular Thoracic Surgery, Sher-i-Kashmir Institute of Medical Science, Jammu and Kashmir, Srinagar, Soura, India.
*Corresponding Author
Riyaz Ahmad,
Deparment of Anaesthesia and Critical Care,
Sher-i-Kashmir Institute of Medical Sciences,
Jammu and Kashmir, Srinagar, India.
Tel: +919419548565
E-mail: riyazali88@gmail.com
Received: April 11, 2017; Accepted: May 24, 2017; Published: May 29, 2017
Citation: Ahmad R, Ahad B, Zahoor SA, Bhat MA (2017) Paravertebral Block: A Comparative Study Evaluating Performance of Direct Surgical And Classic Technique. Int J Anesth Res. 5(5), 446-451. doi: dx.doi.org/10.19070/2332-2780-1700092
Copyright: Ahmad R© 2017. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Thoracic paravertebral block (TPVB) has been employed for post operative analgesia in thoracotomy. Paravertebral block can be given by various techniques, two most commonly used techniques include direct surgical and classic technique yet the relative efficacy of one technique over alternate has not been accessed so far. This study has been intended to look at the performance of both these techniques on different imperative criteria’s. namely analgesic efficacy, technical difficulties and procedure time.
Methodology: This study was conducted in a prospective, randomized, double blind fashion. Sixty adult patients of ASA I & II status underwent unilateral thoracotomy. divided into two groups, Group S and Group C and randomly allocated. Paravertebral catheter was placed at an appropriate level just before the closure of thoracotomy either by surgical or classic technique and was activated by 15 ml of ropivacaine 0.3%. Time taken to perform the block was noted down. After recovery from GA, pain was assessed by VAS at pre-fixed intervals. The patients were administered top up dose through paravertebral catheter as soon as VAS score exceeded 4. Total requirement of ropivacaine and rescue analgesia (Morphine) consumption in 24 hours were noted down. Technical problems of both the techniques were documented by the surgeon or anesthetist who performed the block.
Result: Out of sixty patients three patients were excluded. Patients in Group C experienced better analgesia compared to Group S. Mean VAS scores at rest were lower at all measured intervals in Group C compared to Group S. Mean total consumption of ropivacaine was (91.72 ± 9.85) mg in Group C and (127.14 ± 9.58) mg in Group S (p<0.0001) over 24 hrs. Rescue analgesia (Morphine) consumption was higher in Group S vs. Group C (Group S 10.75 ± 2.6 vs. Group C 6.56 ± 1.44, p-value = 0.0001) over 24 hrs. Average procedure time in Group S 29.39±5.92 was almost double than Group C 15.90 ± 3. Technique related problems in Group S included difficult pleural stripping, local anesthetic leak, catheter fixation and pleural reattachment while in Group C difficult catheterization was the only problem encountered.
Conclusion: Paravertebral block by classic technique provides superior analgesia in thoracotomy, takes less time to execute than direct surgical technique and has fewer technical troubles.
2.Introduction
3.Materials And Methods
4.Results
5.Discussion
6.Conclusion
7.Acknowledgments
8.References
Keywords
Paravertebral Block; Paravertebral Technique; Procedure Time; Analgesic Efficacy; Technical Difficulty.
Introduction
Thoracotomy is widely recognized as being one of the most painful surgical procedures. Acute post-thoracotomy pain is aggravated by the constant movement of breathing. Pain relief is therefore, essential to facilitate coughing and deep breathing and to promote early mobilization [1]. Opioids alone are not potent enough to control post-thoracotomy pain without detrimental effects on respiratory outcome, epidural analgesia has been traditionally considered as the gold standard for thoracic pain control [2]. On the other hand, it has been shown that a paravertebral block (PVB) using a local anesthetic (LA) provides comparable pain relief to that of an epidural but with fewer side effects [3, 4].
The thoracic paravertebral space (TPVS) is a wedge-shaped space [5] that lies on either side of the vertebral column. It is wider on the left than on the right [6]. The endothoracic fascia divides the TPVS into two potential fascial compartments, the anterior “extrapleural paravertebral compartment” and the posterior “subendothoracic paravertebral compartment” [7, 8]. The spinal nerves in the TPVS are segmented into small bundles lying freely among the fat and devoid of a fascial sheath, which makes them exceptionally susceptible to local anesthetic block [9]. The intercostal nerve and vessels are located behind the endothoracic fascia [10, 11]. While the sympathetic trunk is located anterior to it [11, 12] in the TPVS.
There are two approaches for thoracic PVBs; the paravertebral space can be approached percutaneously, or alternatively, a paravertebral catheter (PVC) can be placed under direct vision at thoracotomy [13, 14]. The classical percutaneous technique, which is most commonly used, involves eliciting loss of resistance [15]. Thoracic paravertebral catheters can also be safely, accurately, and easily placed under direct vision during thoracic surgery from within the chest [16-18].
These paravertebral block techniques have been studied extensively in terms of their complications, technical difficulties, safety, effectiveness, and failure rates [21-26] but none of the studies have proven the performance of these techniques comparable to each other. So we conducted this randomized controlled trial to compare these two techniques on some practically important criteria's namely analgesia, technical difficulty and procedure time.
Materials And Methods
The study entitled “Paravertebral block: A comparative study evaluating Performance of direct surgical and classic paravertebral technique.” was conducted in a prospective, randomized, double blind fashion by the Department of Anaesthesiology in Sher-i-Kashmir Institute of Medical Sciences (SKIMS). This study was approved by Institutional ethical committee and a written informed consent was obtained from all patients included in the study.
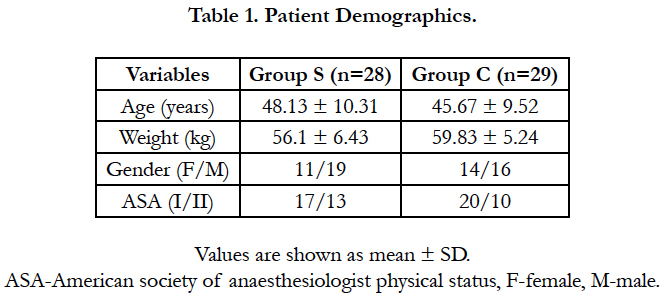
A total of 60 patients of ASA I and ASA II physical status, undergoing unilateral thoracotomy were recruited for the study and then randomly allocated in two Groups of 30 patients each using a sealed envelope technique. Both data collector and the patient were blinded. Group C: Included 30 patients who received total of 15ml of 0.3% Ropivacaine by classic technique Group S: Included 30 patients who received total of 15ml of 0.3% Ropivacaine by surgical technique.
Post operative mechanical ventilation, bilateral thoracotomy, total pleurectomy, ASA III and IV status, Complete heart block, AV Block, empyema, heamodynamic instability, ropivacaine allergy was kept as exclusion criteria.
Catheter based paravertebral technique was employed and was inserted by a scrubbed anesthetist or a surgeon depending on the technique. classic technique involved application of 18 G Tuhoy needle, just before the closure of the thoracotomy wound Tuhoy needle was inserted two intercostal spaces below the level of the thoracotomy and about 2.5 centimeters from the midline posteriorly. Tuhoy needle was advanced perpendicular to the skin upon contact with the tranverse process the needle was withdrawn slightly and redirected caudally an additional 1cm. The stylet was removed and local anesthesia was injected after checking for the absence of blood reflux by aspiration. This raised a bleb under theparietal pleura which, after injection of 15 ml of local anesthetic formed a pocket extending several levels above and below the surgical level and separate from the thoracotomy incision. The catheter was then fed into this newly formed pocket in a cephalad direction. Technical difficulties if encountered were documented by the concerned anesthetist.
Surgical technique was conducted by a surgeon. The parietal pleura was raised from the posterior chest wall up to the vertebral bodies two intercostal spaces above and below the thoracotomy incision, exposing the paravertebral space. A small defect was made in the extrapleural fascia into the paravertebral space using a forceps. percutaneously inserted catheter was passed through the defect into the paravertebral space under direct vision and advanced 2 to 3 cm to lie against the costovertebral joints. The pleura was reattached, catheter was fixed by a purse string suture and the paravertebral space was infused with 0.3% ropivacaine. Surgeon conducting the procedure documented any technical difficulty encountered during the procedure.
All patients received a standardized general anesthesia technique, i.e. after transferring the patients into the operating room, standard monitors (ECG, SpO2, automated non invasive arterial blood pressure) were attached and baseline parameters were noted down. Patients were then induced with injection Propofol 2mg kg-1, injection Morphine 0.1mg kg-1 and injection Atracurium 0.8 mg kg-1. Injection paracetamol 20mg kg-1 were given intraoperatively towards the start of the case. Patients were intubated with double lumen ETT and one lung ventilation was started as and when required. Anesthesia was maintained with O2 in N2O and <1 MAC of Isoflurane. During the surgery the targeted SPO2 remained ≥ 91%. Incremental and additional anesthetic drugs or other medications were given as required. Intraoperatively before surgical incision, invasive intra arterial cannulation for BP monitoring and for blood sampling was done Post-operatively the patient were extubated after reversal of neuromuscular blockade. as a standard of care, all patients received supplemental oxygen therapy for 24 hours post-operatively.
The pain scoring was done using VAS which was explained to the patient preoperatively. The target pain scoring as per VAS was < 4 at rest. Pain scores at rest were recorded every 30 minutes in first hour, then hourly for next three hours and then four hourly till 24 hours. pain scores on coughing was recorded every 2 hour in first 4 hours, then 4 hourly upto 24 hours. total consumption of ropivacaine and rescue analgesics was calculated after 24 hrs. Rescue analgesics were given in a predetermined order i.e. First analgesia was Injection Ropivacaine 0.2% -10 ml, second was iv morphine given in 0.025mg kg-1 increments. Injection paracetamol 15mg kg-1 (max 4gm in 24hours) was given 6 hourly in both the groups. An appropriative interval of time was kept between the staggering of analgesics, keeping into account the onset of action and effectiveness of analgesics in treating acute thoracotomy pain and that to keep VAS at rest < 4. Procedure time was taken from needle prick or pleural stripping up to catheter fixation any technical difficulty encountered was noted down.
Monitoring included non-invasive blood pressure, electrocardiogram and arterial oxygen saturation (SpO2). Any complications of the technique or side effects of the drugs were noted and appropriately managed.
The statistical tests like independent t-test and Wilcoxan – Mann Whitney u test were employed for continuous variables after checking the normality condition of variables. The categorical variables were analyzed with chi–square and Fisher exact test to see the association of attributes. All the statistical result were discussed on the basis of 5% level of significance i.e. p value < 0.05 was considered significant.
Results
Sixty Patient’s were initially enrolled for the study three patients were excluded because of catheter dislodgement and block failure patient characteristics were comparable in the two groups (Table 1) as no significant difference was found between the two groups.
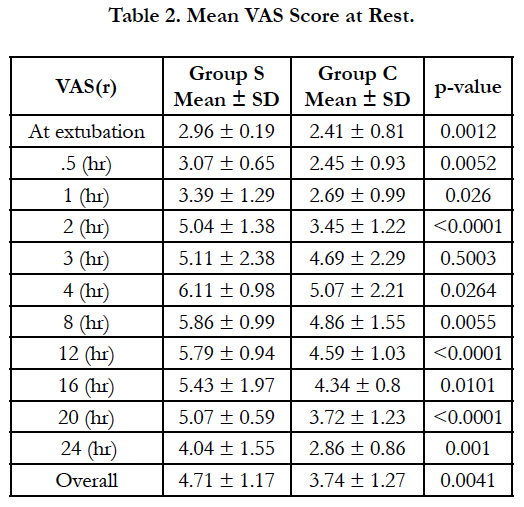
The mean VAS scores at rest in Group C were less at all intervals compared to Group S. The difference between the mean VAS scores per hour at rest were statistically significant almost at all intervals except at 3(hr). The data also revealed that VAS scores peaked at 4(hr) interval and then gradually declined in both groups (Table 2).
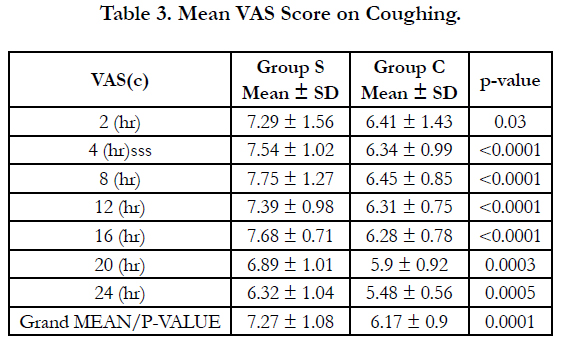
Both techniques did not provide satisfactory analgesia on coughing. Mean VAS score on coughing remained beyond 5 throughout the study however it remained lower in Group C compared to Group S with statistically significant difference (Table 3).
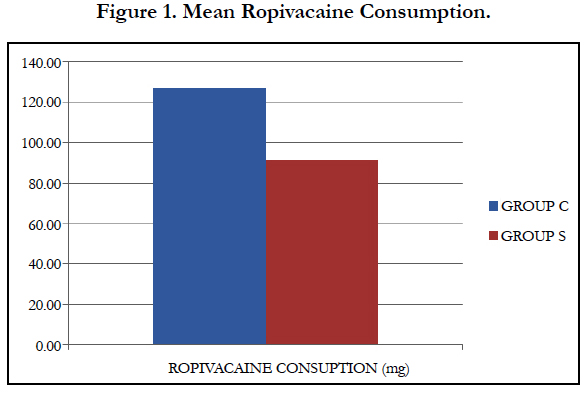
Ropivacaine consumption varied between the groups, Group S required repeated doses of ropivacaine compared to Group C(127.14 ± 9.58 versus 91.72 ± 9.85, P =< 0.0001) (Figure 1).
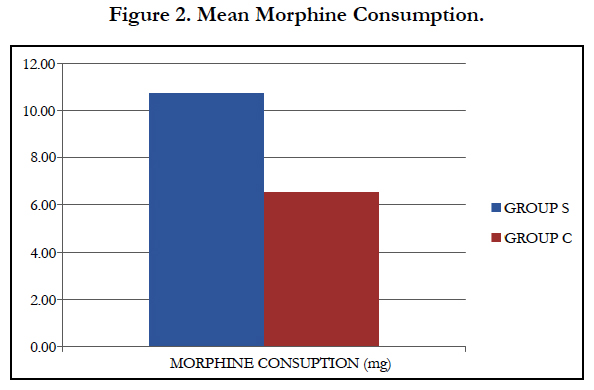
Rescue analgesia (morphine) was much more needed in Group S compared to Group C. Total morphine consumption in both the groups showed a statistically significant difference (Group S 10.75 ± 2.6 vs Group C 6.56 ± 1.44, P =< 0.0001) (Figure 2).
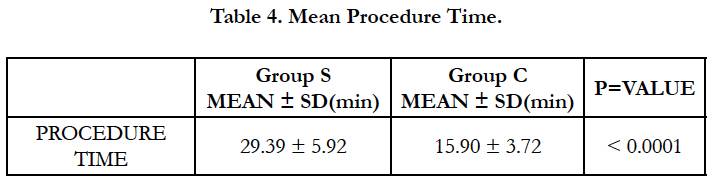
Surgical paravertebral technique took double the time to complete than classic technique (Group S 29.39 ± 5.92min vs Group C 15.90 ± 3.72) (Table 4).
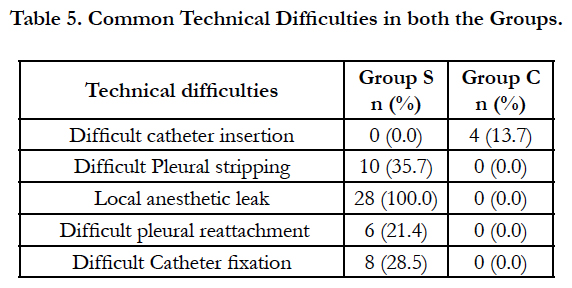
Common Techniqual difficulties encountered during surgical technique include difficult catheter fixation, pleural reattachment, local anesthetic leak and pleural stripping while in classic technique difficult catheterization was the most frequent problem observed by anesthetists (Table 5).
We did not witness any complications due to post-operative pain (pneumonia, atelectasis, secreation retention), drug related (intravascular injection, allergies) or technique related (pneumothorax, epidural, intrathecal injection) problems in our study.
Discussion
Thoracotomy frequently causes severe postoperative pain and significant morbidity [24, 27]. Atelectasis, pneumonia, pulmonary embolism, and emergency intensive care admission have all been found to be related to poor analgesia and consequent immobility [24, 28, 29]. Postoperative pain is thought to be the single most important factor leading to ineffective ventilation and impaired secretion clearance after thoracotomy [4]. Severe or inadequately treated acute pain after thoracotomy also predicts conversion to chronic post-thoracotomy pain [24] and long-term post-surgical fatigue [31]. Thoracic paravertebral block (TPVB) has been shown to provide superior post-thoracotomy analgesia and lung function, compared with systemic opioids or Interpleural local anesthetics (LA) [30, 31]. Three systematic reviews have compared the efficacy of Thoracic paravertebral block and Thoracic epidural analgesia (TEA) after thoracotomy [2, 24, 30]. Detterbeck [30] found that Paravertebral block provided equivalent pain relief to Thoracic epidural analgesia, but did not quantitatively compare complications between the two techniques. A meta-analysis by Davies and colleagues [24] showed that Paravertebral block provides pain relief as good as thoracic epidural analgesia, using local anesthetics with or without opioid, but with fewer side-effects, technical problems, and failed blocks.
The classical technique, which is most commonly used, involves eliciting loss of resistance [15]. Unlike epidural space location, where a definite give is felt as the needle tip traverses the firm ligamentum flavum, TPVS location using loss of resistance is subjective and indefinite [20, 21] and may not be appreciated as a definite give [19]. The needle may be advanced by a fixed predetermined distance (1-2 cm) [12, 35] once the needle is walked off the transverse process without eliciting loss of resistance [35-37]. Difficulty is also commonly encountered during catheter insertion and may require manipulation of the needle [15, 19] or injection of saline to create a saline-filled cavity before passing a catheter. Very easy passage of the catheter may indicate interpleural placement.
Thoracic paravertebral catheters can also be safely, accurately, placed under direct vision during thoracic surgery from within the chest [16-18]. The original description by Sabanathan et al., [18] involved reflecting the parietal pleura from the posterior wound margin on to the vertebral bodies to create an extrapleural paravertebral pocket into which a percutanously inserted catheter is placed against the angles of the exposed ribs.
Ropivacaine dose used in our study has been chosen from other relevant studies as there are no specific guidelines regarding the ropivacaine dosage in paravertebral block.
The main purpose of this study was to compare the effectiveness and practicality of the two techniques, so we tested these techniques on some practically important parameters that’s analgesic efficacy, technical difficulty and procedure time.
With regards to analgesic efficacy classic technique showed better results compared to surgical technique, however both these techniques provided inadequate analgesia on coughing. The mean VAS scores at rest in Group S were less at all intervals compared to Group C. The difference between the mean VAS scores per hour at rest were statistically significant at all intervals except at 3(hr). VAS scores on coughing revealed inadequate analgesia however it was lower in Group C. These scores showed a similar trend with other studies but with a considerable variation in magnitude of scores which can be attributed to local anesthetic concentration, technical variations and magnitude of local anesthetic leak into pleural cavity.
Ropivacaine boluses had to be given repeatedly in Group S compared to Group C. That lead to more ropivacaine consumption in Group C (127.14 ± 9.58 vs 91.72 ± 9.85, P==<0.0001). Morphine also followed similar pattern with a statistically significant difference these findings can be attributed to higher VAS scores in Group S compared to Group R (Group S 10.75 ± 2.6 vs Group C 6.56 ± 1.44, P =< 0.0001).
Surgical technique took more time to execute than classic technique because former involved much more steps starting from pleural stripping, catheter placement, catheter fixation and pleural reattachment it took almost double the time for surgical technique to be completed (Group S vs Group C 29.39 ± 5.92 vs 15.90 ± 3.72).
Difficult catheterization was the only problem that we faced with classic technique 4(13.79%). While surgical technique was technically more complex and was riddled with much more problems that included pleural stripping problems, local anesthetic leak, pleural reattachment, and catheter fixation problems from these observations it is quite clear that surgical technique variation used by Sabanathan et al., [18] suffers from many techniqual problems which leads to inferior performance than classic technique.
Conclusion
Patients receiving paravertebral block by classic technique experience superior analgesia in thoracotomy than direct surgical technique in addition other advantages of classic technique include faster execution and fewer technical problems, however it gets limited by blind method of catheter placement.
Acknowledgments
The authors would like to acknowledge all kind cooperation of the physicians and nurses, Cardiac Surgery Operating Room, CVTS Ward, and SICU of Sher-i-kashmir institute of medical science, jammu and Kashmir, Srinagar, Soura, India. for their kind assistance and impressive cooperation.
References
- Hughes R, Gao F (2005) Pain control for thoracotomy. Conti Educ in Anaesth Crit Care Pain. 5 (2): 56–60.
- Joshi GP, Bonnet F, Shah R, Wilkinson RC, Camu F, et al., (2008) A systematic review of randomized trials evaluating regional techniques for post thoracotomy analgesia. Anesth Analg. 107(3): 1026-40.
- Gulbahar G, Kocer B, Muratli SN, Yildirim E, Gulbahar O, et al., (2010) A comparison of epidural and paravertebral catheterisation techniques in postthoracotomy pain management. Eur J Cardiothorac Surg. 37(2): 467–72.
- Richardson J, Sabanathan S, Jones J, Shah RD, Cheema S, et al., (1999) A prospective, randomized comparison of preoperative and continuous balanced epidural or paravertebral bupivacaine on post-thoracotomy pain, pulmonary function and stress responses. Br J Anaesth. 83(3): 387–92.
- MacIntosh R, Bryce-Smith R (1962) Local Analgesia: Abdominal Surgery. Edinburgh, E&S Livingstone, 26-32.
- Kittredge RD (1983) Computed tomographic evaluation of the thoracic prevertebral and paravertebral spaces. J Comput Tomogr. 7(3): 239–50.
- Karmakar MK, Kwok WH, Kew J (2000) Thoracic paravertebral block: Radiological evidence of contralateral spread anterior to the vertebral bodies. Br J Anaesth. 84(2): 263-5.
- Karmakar MK, Chung DC (2000) Variability of a thoracic paravertebral block: Are we ignoring the endothoracic fascia? Reg Anesth Pain Med. 25(3): 325–7.
- Nunn JF, Slavin G (1980) Posterior intercostal nerve block for pain relief aftercholecystectomy: Anatomical basis and efficacy. Br J Anaesth. 52(3): 253–60.
- Moore DC (1981) Intercostal nerve block: Spread of india ink injected to the rib’s costal groove. Br J Anaesth. 53(4): 325–9.
- Pernkopf E (1980) Atlas of Topographical andApplied Human Anatomy, Vol 2: Thorax, abdomen and extremities. Urban and Schwarzenberg. 127-9.
- Moore DC, Bush WH, Scurlock JE (1980) Intercostal nerve block: A roentgenographic anatomic study of technique and absorption in humans. Anesth Analg. 59(11): 815-25.
- Berrisford RG, Sabanathan SS (1990) Direct access to the paravertebral space at thoracotomy. Ann Thorac Surg. 49(5): 854.
- Daly DJ, Myles PS (2009) Update on the role of paravertebral blocks for thoracic surgery: are they worth it? Curr Opin Anaesthesiol. 22(1): 38-43.
- Eason MJ, Wyatt R (1979) Paravertebral thoracic block-a reappraisal. Anaesthesia. 34(7): 638-42.
- Downs CS, Cooper MG (1997) Continuous extrapleural intercostal nerve block for post thoracotomy analgesia in children. Anaesth Intensive Care. 25(4): 390–7 22.
- Karmakar MK, Booker PD, Franks R, Pozzi M (1996) Continuous extrapleural paravertebral infusion of bupivacaine for post-thoracotomy analgesia in young infants. Br J Anaesth. 76(6): 811–5.
- Sabanathan S, Bickford-Smith PJ, Pradhan GN, Hashimi H, Eng J-B, et al., (1988) Continuous intercostal nerve block for pain relief after thoracotomy. Ann Thorac Surg. 46(4): 4256.
- Johnson CM (1993) Continuous paravertebral block in children (letter). Anaesthesia. 48(1): 93.
- Richardson J, Lo¨nnqvist PA (1998) Thoracic paravertebral block. Br J Anaesth 81(2): 230–8.
- Richardson J, Cheema SP, Hawkins J, Sabanathan S (1996) Thoracic paravertebral space location: A new method using pressure measurement. Anaesthesia. 51(2): 137–9.
- Mowbray A, Wong KK, Murray JM (1987) Intercostal catheterisation: An alternative approach to the paravertebral space. Anaesthesia. 42(9): 958–61.
- Lonnqvist PA, MacKenzie J, Soni AK, Conacher ID (1995) Paravertebral blockade. Failure rate and complications. Anaesthesia. 50(9): 813-5.
- Davies RG, Myles PS and Graham JM (2006) A comparison of the analgesic efficacy and the side effects of paravertebral vs. epidural blockade for thoracotomy - a systematic review and meta-analysis of randomized trials. Br J. Anaesth. 96(4): 418-426.
- Fibla JJ, Molins L, Mier JM, Sierra A, Carranza D, Vidal G (2011) The efficacy of paravertebral block using a catheter technique for postoperative analgesia in thoracoscopic surgery: a randomized trial. Eur J Cardiothorac Surg.40 (4): 907-911.
- Kotzé A, Scally A, Howell S (2009) "Efficacy and safety of different techniques of paravertebral block for analgesia after thoracotomy: a systematic review and metaregression." Br J Anaesth. 103(5): 626-636.
- Bimston DN, McGee JP, Liptay MJ, Fry WA (1999) Continuous paravertebral extrapleural infusion for post-thoracotomy pain management. Surgery. 126(4): 650-657.
- Pluijms WA, Steegers MA, Verhagen AF, Scheffer GJ, Wilder-Smith OH (2006) Chronic post-thoracotomy pain: a retrospective study. Acta Anaesthesiol Scand. 50(7): 804-808.
- Kruger M, Sandler AN (1998) Post-thoracotomy pain control. Curr Opin Anaesthesiol. 12: 55-58.
- Detterbeck FC (2005) Efficacy of methods of intercostal nerve blockade for pain relief after thoracotomy. Ann Thorac Surg. 80(4): 1550-1559.
- Richardson J, Sabanathan S, Shah R (1999) Post-thoracotomy spirometric lung function: the effect of analgesia. A review. J Cardiovasc Surg. 40(3): 445-446.
- Gilbert J, Hultman J (1989) Thoracic paravertebral block: A method of paincontrol. Acta Anaesthesiol Scand. 33(2): 142–5.
- Coveney E, Weltz CR, Greengrass R, Iglehart JD, Leight GS, et al., (1998) Use of paravertebral block anesthesia in the surgical management of breast cancer: Experience in 156 cases. Ann Surg. 227(4): 496–501.
- Greengrass R, O’Brien F, Lyerly K, Hardman D, Gleason D, et al., (1996) Paravertebral block for breast cancer surgery. Can J Anaesth. 43(8): 858–61.
- Marret E, Bazelly B, Taylor G, Lembert N, Deleuze A, et al., (2005) Paravertebral block with ropivacaine 0.5% versus systemic analgesia for pain relief after thoracotomy. Ann Thorac Surg. 79(6): 2109-13.
- Fibla JJ, Molins L, Mier JM, Sierra A, Vidal G (2008) Comparative analysis of analgesic quality in the postoperative of thoracotomy: paravertebral block with bupivacaine 0.5% vs ropivacaine 0.2%. Eur J Cardiothorac Surg. 33(3): 430-4.
- Helms O, Mariano J, Hentz JG, Santelmo N, Falcoz PE, et al., (2011) "Intra-operative paravertebral block for postoperative analgesia in thoracotomy patients: a randomized, double-blind, placebo-controlled study." Eur J Cardiothorac Surg. 40(4): 902-906.