The Analgesic Efficacy and Safety of the Association Tramadol - Bupivacaïne Versus Morphine-Bupivacaïne In Epidural Thoracic Analgesia For Thoracotomy
Cheikhrouhou H1*, Kharrat A1, Derbel R1, Souissi I1, Jmal K1, Ben Jmaa H2, Hentati A2, Frikha M1, Frikha I2, Karoui A1
1 Department of Anesthesiology and Critical Care, Habib Bourguiba Hospital, Sfax, Tunisia.
2 Department of Cardiovascular and Thoracic Surgery, Habib Bourguiba; Sfax, Tunisia.
*Corresponding Author
Cheikhrouhou Hichem,
Service d’Anesthésie Réanimation Chirurgicale,
CHU Habib Bourguiba, Sfax, Tunisie.
Tel: 0021698418366
E-mail: cheikhrouhouhichem@yahoo.fr
Received: December 09, 2016; Accepted: January 09, 2017; Published: January 12, 2017
Citation: Cheikhrouhou H, Kharrat A, Ghrab B, Abdenadher M, Hriz MA, et al., (2017) The Analgesic Efficacy and Safety of the Association Tramadol - Bupivacaïne Versus Morphine-Bupivacaïne In Epidural Thoracic Analgesia For Thoracotomy. Int J Anesth Res. 5(1), 389-392. doi: http://dx.doi.org/10.19070/2332-2780-1600080
Copyright: Cheikhrouhou H© 2017. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: The aim of this study is to compare the analgesia efficacy and side effects in patients undergoing an electiv thoracotomy with thoracic epidural analgesia using tramadol or morphine in association with bupivacaine.
Patients and Methods: It is a prospective, randomized, double blind study, 40 patients scheduled for thoracotomy randomized after anesthesia induction in two equal groups. Each patient received in epidural analgesia using bupivacaine with morphine (Group 1) or bupivacaine with tramadol (Group 2).
In post-operative evaluation, pain was measured using a visual analog scale (VAS), arterial blood gas, post-operative sedation
and other side effects was done throughout 48 hours.
Results: All the groups were homogeneous with respect to their demographics. The total consumption of bupivacaïne, the number of boluses and analgesia scores at rest, during coughing and during respiratory physiotherapy were comparative in both groups. Patients in tramadol group were less sedated within the first 4 hours (p: 0.02) and had less respiratory depression within the first 6 post-operative hours (p :0.007). Nausea, vomiting, pruritus and urinary retention were comparative in both groups.
Conclusion: Our study showed that after thoracotomy, the post-operative analgesia provided by tramadol associated with bupivacaïne by epidural thoracic catheter was similar to that with morphine. The risks of sedation and respiratory depressionwere less for the patients receiving tramadol than morphine.
2.Background
3.Methods
4.Results
5.Discussion
6.Conclusion
7.References
Keywords
Postoperative Analgesia; Morphine; Epidural Thoracic Analgesia; Thoracotomy; Tramadol.
Background
Thoracotomy is widely recognized as one of the most painful surgical procedures acute [1] post thoracotomy pain is aggravated by the constant movement of breathing and causes discomfort and anxiety for patient. Pain relief is, therefore, essential to facilitate coughing and deep breathing and to promote early mobilization.
Shallow breathing and impaired coughing, resulting from thoracotomy pain are a major cause of atelectasis and retention of secretions, both of which can lead to hypoxemia, hypercapnia, and respiratory failure, especially in smoking patients with pre-existing lung disease [2]. It has been demonstrated that poor analgesia is associated with increased intensive care unit admissions and longer hospital stay. For this reason, different analgesic methods such as thoracic epidural analgesia, paravertebral blocks, and systemic analgesics have been tried. However, epidural analgesia is considered to be the gold standard for post thoracotomy pain relief [3].
The use of morphine as adjuvant to local anesthetics is associated with quite a few side effects, so various options including clonidine, dexmedetomidine, magnesium and tramadol are being extensively evaluated as an alternative with emphasis on opioidrelated side effects such as respiratory depression, nausea, urinary retention, and pruritus besides improving quality and duration of analgesia [4]. We conducted this clinical study to determine and compare the effects of adding a tramadol and morphine to epidural bupivacaine in patients undergoing an elective thoracotomy.
Methods
Forty patients with physical status American Society of Anesthesiologists (ASA) I-III more than 20 years admitted for elective unilateral thoracic surgery were enrolled for the study. The patients undergoing an elective unilateral thoracotomy were divided into two groups (Group 1 and Group 2). Patients with ASA score IV or more, body mass index >30 kg/m2, hypersensitivity to morphine , tramadol or bupivacaine, severe renal, hepatic, cardiac or neurologic disease, haemostasis disorder and those using opioid or systemic analgesic preoperatively were excluded from the study. At the anesthesia consultation, patients were instructed about pain evaluation using visual analog scale (VAS) of 0-10 cm (0 cm = no pain and 10 cm = the worst pain). On arrival to the operating room, all patients were preloaded with 500 ml of crystalloid. In the operating room, electrocardiography, oxygen saturation and continuous arterial blood pressure monitoring were started.
Epidural catheter (20G) was inserted through 18G Tuohy needle at T6-T9 intervertebral space in sitting position depending. The epidural space was identified using loss of resistance to saline technique. Epidural catheter was inserted and fixed at 3-4 cm inside the space. A test dose of 3 ml of 1.5% lidocaine with epinephrine was given to rule out intravascular or intrathecal placement of the catheter. Thiopental 5 à 7 mg /kg, fentanyl 2 μg /kg and cisatracrium 0.15 mg/kg were used intravenously for induction of anesthesia; patients were intubated with a simple lumen tube. Anesthesia was maintained with equal volumes of oxygen and air in isoflurane (0.5-1.5%). Intraoperative rescue analgesia with fentanyl 1 μg/kg was used. After induction, patients were divided into two groups to receive following drug mixtures epidurally:
• Group 1: Bupivacaine 0.25% 2.5 ml + morphine 3 mg in 6 ml 0.9% saline as a bolus then bupivacaine 0.125% + morphine 0.01/ml using infusion pump 6ml/h.
• Group 2: Bupivacaine 0.25% 2.5 ml + tramadol 100 mg in 6 ml 0.9% saline as a bolus then bupivacaine 0.125% + tramadol 0.5 mg/ml using infusion pump 6ml/h.
Epidural infusion with 6 ml/h was started and it was continued during the per and postoperative period via epidural catheter using an infusion pump. After extubation, patients were shifted to the post anesthesia care unit for 2 h and then shifted to the postoperative ward for 48 h. The intensity of postoperative pain was measured using VAS. Pain scores and sensory level was assessed H0, H2, H4, H12, H24, H36, H48 post-operative. Patients complaining of pain in the postoperative period with VAS score ≥4, received a bolus 5 ml of epidural infusion as rescue analgesia and time to the first request for analgesia was noted. Postoperative monitoring included heart rate, blood pressure, pulse oxymetry, respiratory rate, oxygen arterial pressure and sensory block using pin prick sensation (T2-T12 dermatome). The degree of sedation was assessed after admission to the recovery room every 30 min for 2 h using modified Ramsay sedation scale. Side effects such as hypotension, nausea, vomiting, pruritus, and shivering were recorded.
All statistical analyses were performed using SPSS for windows versions 18.0. Continuous variables were tested for a normal distribution by the Kolmogorov–Smirnov test. Parametric data were compared using analysis of variance. Comparison between groups at different time intervals was assessed using paired t test. All the categorical data were compared by using the Chi-square test. A power analysis based on 95% confidence interval and β error of 20% revealed a sample size of 40 subjects (20 subjects in each group). A sample size of 20 patients in each group was needed to detect an intergroup difference of at least 20% with two-sample t-test. A P < 0.05 was considered to be significant.
Results
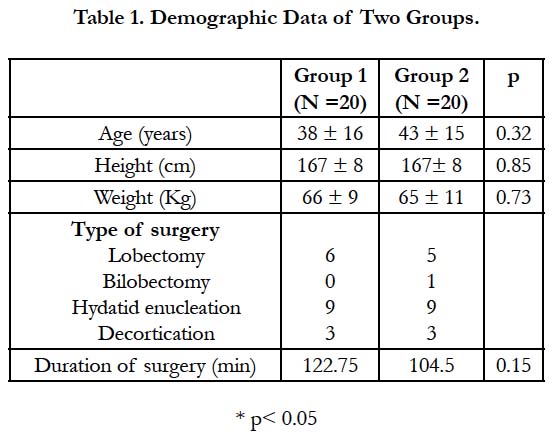
The two groups were homogenous with reference to age, sex, body weight, duration of surgery, and surgical procedures (Table 1). The hemodynamic variables (mean ± SD) between the groups at different study intervals were comparable.
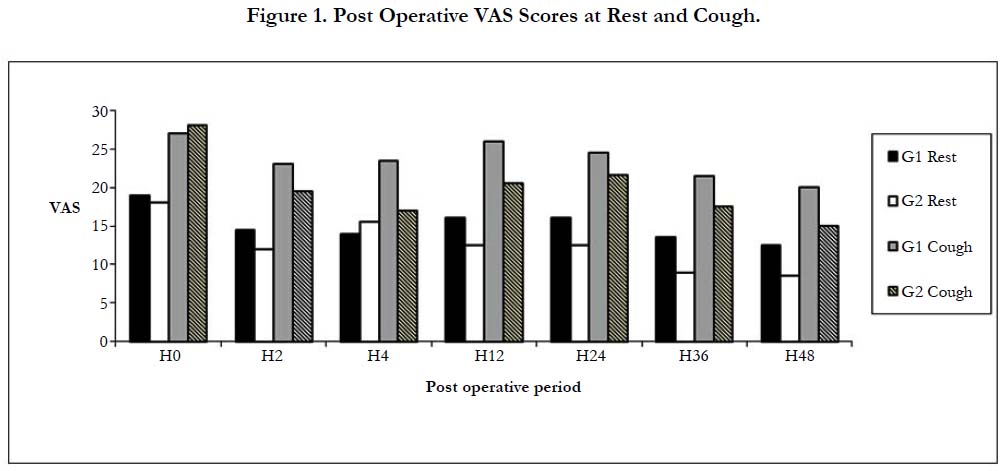
All subjects in our study groups assessed at various intervals during the study period had a good sensory block extending from T2 to T12 dermatomes, depending on the level of epidural. On comparing, the VAS scores at rest and cough between the groups at various intervals, no significant difference was found between two groups (Figure 1).
The total consumption of bupivacaine and the number of bolus were comparable. The respiratory frequency was greater in patients in group 1 at H1, H2, H4, H24 et H48 post-operative. The rate of pulse oximetry was lower in morphine group without significant difference. The oxygen blood pressure was higher in group 2 at first 48 first hours post-operative, however, the PaCO2 was higher in morphine group in H12, H24 and H4 post-operative.
Respiratory depression was reported to 9 patients in morphine group and none in tramadol group. The incidence of nausea, vomiting and pruritus was similar in two groups.
Discussion
Epidural analgesia with local anesthetics is one of the most effective techniques used for postoperative pain relief and may improve patient outcome [5]. Opiods used as adjuvant to local anesthesics in epidural analgesia was the common practice to have best analgesia but this practice is related to more side effects [6]. Adding other drugs to the neuraxial block to improve the quality of anesthesia and prolong the duration of analgesia without any added side effects. The faster onset of both sensory and motor blockade, prolonged duration of analgesia, dose-sparing action of local anesthetics and stable cardiovascular parameters makes these agents a very effective adjuvant in regional anesthesia.
The present study was undertaken to compare the postoperative analgesic efficacy and safety of morphine and tramadol as adjuvants to bupivacaine in thoracic epidurals in patients undergoing an elective thoracotomy. The results emerged from this clinical study: morphine and tramadol have the same beneficial effect on postoperative pain intensity and analgesic requirements in the postoperative period; however tramadol decreases the incidence of postoperative sedation and respiratory depression without increasing incidence of nausea, vomiting and prurit.
The efficacy of tramadol has been proven for adults after surgeries that have moderate to severe pain. Tramadol was used for analgesia after caesarean section under epidural anesthesia and 100 mg of tramadol administered by the epidural catheter has provided adequate postoperative analgesia of 10 hours without respiratory depression and the dose of 100 mg was as effective as 200 mg [7]. Delilkan [8] noted a long duration of action of a dose of 100 mg tramadol per epidural compared to the intravenous administration in adults (9.4 versus 6 h) with satisficing analgesia after abdominal surgery.
In thoracic surgery, tramadol was compared to lumbar epidural morphine in thoracotomy in the study of Turker with similar analgesic efficacy [9]. In our study the analgesia was comparable between the two groups with the VAS scores below 40 mm at rest and cough, and same consumption of bupivacaine and a total number of boluses. Respiratory depression after epidural morphine is a rare but potentially dramatic complication and is a limiting factor in the absence of rational monitoring [10]. The risk is not correlated with the dose administered. In a Swedish multicentre study [11], the risk was evaluated at 1/1000 for an epidural dose of morphine 4 mg.
Baraka [7] studied the effects of epidural 100 mg of tramadol compared with 4 mg of morphine for abdominal surgery: he concluded in favor of tramadol for the absence of respiratory depression with similar pain scores in both groups. This very low incidence of the effects of tramadol on respiratory function has been found in other studies comparing this product with pethidine either in the postoperative period in orthopedic surgery [12] or even under general anesthesia in spontaneous ventilation, absence of repercussion on the elimination of CO2 is a constant likely to be related to the conservation of the tidal volume [13]. In general, with tramadol there is a decrease in respiratory frequency but not in tidal volume, and to a lesser extent than in morphine, the elimination of CO2 in particular is not disturbed [13, 14]. Our results are consistent with those of the literature. Respiratory frequency was higher at 60 min, 90 min, H2, H4, H24 and H48 postoperatively in the tramadol group than morphine group. SpO2 was better in the tramadol group throughout the study without statistical significance. PaOM2 values were higher in this group during the 48-hour postoperative period. PaCO2 was higher in morphine group at the 12th, 24th and 48th postoperative hours with statistically significant differences. In our work, lower sedation scores were noted with tramadol during the first four hours postoperatively.
Conclusion
Our study showed that the epidural tramadol-bupivacaine combination provides analgesia comparable to that obtained by the morphine-bupivacaine combination after thoracotomy. Tramadol reduces the incidences of respiratory depression and causes less sedation episodes.
References
- CL Bong, M Samuel, JM Ng, C Ip-Yam (2005) Effects of Preemptive Epidural Analgesia on Post-thoracotomy Pain. J Cardiothorac Vasc Anesth. 19(6): 786–793.
- I McGovern, C Walker, F Cox, A Ng, J Swanevelder (2007) Pain relief after thoracotomy: Br J Anaesth. 98(6): 844-5.
- A Ng, J Swanevelder (2007) Pain relief after thoracotomy: is epidural analgesia the optimal technique? Br J Anaesth. 98(2): 159–62.
- SR Arain, RM Ruehlow, TD Uhrich, TJ Ebert (2004) The efficacy of dexmedetomidine versus morphine for postoperative analgesia after major inpatient surgery. Anesth Analg. 98(1): 153–8.
- A Rodgers, N Walker, S Schug, A McKee, H Kehlet, et al., (2000) Reduction of postoperative mortality and morbidity with epidural or spinal anaesthesia: results from overview of randomised trials. BMJ. 321(7275): 1493.
- PD Slinger (1999) Pro: every postthoracotomy patient deserves thoracic epidural analgesia. J Cardiothorac Vasc Anesth.13(3): 350–4.
- S Siddik-Sayyid, M Aouad-Maroun, D Sleiman, M Sfeir, A Baraka (1999) Epidural tramadol for postoperative pain after Cesarean section. Can J Anaesth. 46(8): 731–5.
- AE Delilkan, R Vijayan (1993) Epidural tramadol for postoperative pain relief. Anaesthesia. 48(4): 328–31.
- G Turker, S Goren, S Bayram, S Sahin, G Korfali (2005) Comparison of Lumbar Epidural Tramadol and Lumbar Epidural Morphine for Pain Relief After Thoracotomy: A Repeated-Dose Study. J Cardiothorac Vasc Anesth. 19(4): 468–474.
- N Meylan, N Elia, C Lysakowski, MR Tramer (2009) Benefit and risk of intrathecal morphine without local anaesthetic in patients undergoing major surgery: meta-analysis of randomized trials. Br J Anaesth. 102(2): 156–167.
- N Rawal, S Arnér, LL Gustafsson, R Allvin (1987) Present state of extradural and intrathecal opioid analgesia in Sweden. A nationwide follow-up survey. Br J Anaesth. 59(6): 791–9.
- R Tarradell, O Pol, M Farré, E Barrera, MM Puig (1996) Respiratory and analgesic effects of meperidine and tramadol in patients undergoing orthopedic surgery. Methods Find Exp Clin Pharmacol.18(3): 211–8.
- P Tarkkila, M Tuominen, L Lindgren (1998) Comparison of respiratory effects of tramadol and pethidine. Eur J Anaesthesiol.15(1): 64–8.
- BD Beakley, AM Kaye, AD Kaye (2015) Tramadol, Pharmacology, Side Effects, and Serotonin Syndrome: A Review. Pain Physician. 18(4): 395–400.