Toxic Compounds Analysis by High Performance Liquid Chromatography
Hideharu Shintani1*
1* 1Chuo University, School of Science, Kasuga Bunkyo, Tokyo, Japan.
*Corresponding Author
Hideharu Shintani,
Chuo University,
School of Science,
Kasuga Bunkyo, Tokyo, Japan.
Tel: +81425922336
E-mail: shintani@mail.hinocatv.ne.jp
Article Type: Editorial
Received: September 17, 2014; Published: October 02, 2014
Citation: Hideharu Shintani (2014) Toxic Compounds Analysis By High Performance Liquid Chromatography. Int J Clin Pharmacol Toxicol. 3(3), 1-2. doi: dx.doi.org/10.19070/2167-910X-140005e
Copyright: Hideharu Shintani© 2014. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
I have studied the characteristics of toxic compounds from the sterilized medical devices or dental material, which migrated into patient’s body fluid and evaluated if the migrated amount is risky or not. I have studied several sorts of toxic compounds. They are 4,4’-methylenedianiline (MDA), bisphenol A, phthalate esters, methylmethacrylate (MMA), N,N’-dimethyl p-toluidine (DMPT), benzoly peroxide (BPO), 4,4’-dihydroxydiphenyl sulfone, 4-hydroxy 4’-chloro diphenyl sulfone and so on. Mostly they are from medical devices and dental materials. Sorts of sterilization are gamma-ray irradiation, moist-heating, ethylene oxide (EOG) exposure. Detection of MDA and bisphenol A was by electro chemical detector (ECD) at 900 mV. BY applying 900 mV, MDA change to diimino compound and bisphenol A changes to keto compound.These amine-imino or keto-enol changeable tautomerization at relatively lower oxidation-reduction potential is essential factor to detect selectively by ECD.
MDA can be produced from the gamma-ray sterilized thermosetting polyurethane (PU) and was found about 0.6 ppm for gammaray, 0.3 ppm for moist heat and 0.10 ppm for EOG, respectively [1]. This amount is from average three specimens. The virtual safe dose (VSD) was mathematically calculated by the one-hit model. Based on the VSD of 10-6 risk, estimation of the increased cancer risk from the highest MDA level of 0.6 ppm indicated a 1x10-7 increase for 50 kg patient exposed to dialysis three times per week for 70 years. This indicates virtually no serious risk. ECD detection for MDA from gamma-ray sterilized PU is the first report in the world. The formation mechanism of MDA by sterilization is that cleavage or hydrolysis at the urethane linkage adjacent to the terminal amino group by sterilization (Figure 1) [1]. The formation mechanism of MDA by sterilization is not simultaneous cleavage of consecutive urethane linkage, but only one cleavage at the urethane linkage adjacent to the terminal amino group as shown in Figure 1 [1].
Figure 1. Proposed mechanism of MDA formation by cleavage. The arrows indicate the site of possible cleavage.
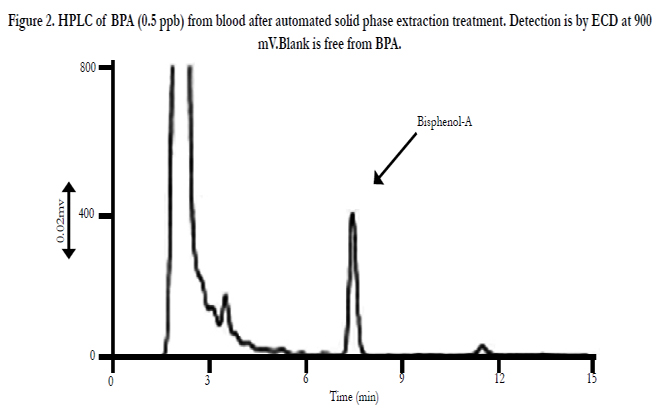
Second is the story about bisphenol A. Bisphenol A (BPA) is known carcinogen and endocrine disrupter. BPA is used to prepare polysulfone (PS) or polycarbonate (OC). PS is used for hollow fiber of dialyzer and PC is for housing of dialyzer. By gammaray or moist heat (121.1°C for 20 min) sterilization, bisphenol A is not detected by gamma-ray and 0.5 ppb was detected by moist heat [2]. This is quite low level detection, so no serious risk.HPLC chromatogram detected by ECD is presented in Figure 2.
Figure 2. HPLC of BPA (0.5 ppb) from blood after automated solid phase extraction treatment. Detection is by ECD at 900 mV.Blank is free from BPA.
Third is the phthalate ester compound. These are diethylhexyl phthalate (DEHP), monoethylhexyl phthalate (MEHP) and phthalic acid (PA).These are eluted from PVC blood bags into plasma and the migrated amount of DEHP, MEHP and PA to plasma was 270 ppm, 40 ppm and 0.6 ppm, respectively [3]. PA is non toxic, but DEHP and MEHP is seriously toxic and MEHP is more toxic than DEHP, which was first reported by the author. Their toxicity is serious, therefore must avoid to use DEHP in PVC. Currently safer plasticizer in place of DEHP is under study and preparation.
Next is the elution amount of MMA, DMPT, BPO to saliva. MMA, DMPT and BPO are the raw material of the preparation of methyl methacrylate polymer. MMA, DMPT and BPO are eluted into saliva at the amount of 32 ppm and 66 ppm, respectively. BPO immediately change to benzoic acid (BA), so BPO is hard to determine as BPO by HPLC. BPO can be determined as BA. The determined amount of BA was 2.2 ppm. Cytotoxicity data of IC50 (μg/mL) of BPO, BA, DMPT and MMA using Balb 3T3 cell were 22, 28, 1500 and 4400, respectively [4]. BPO is most cytotoxic, but it cannot determine cytotoxicity as is because cell culture medium contains blood serum, so BPO immediately changes to BA, which information was obtained by the author at the first, BA is difficult to consider serious cytotoxic, but IC 50 data is shown to be seriously cytotoxic.This is hard to understand and must speculate the fact [4].
The last is the 4, 4’-dihydroxydiphenyl sulfone (bisphenol S) and 4-chloro 4’-hydroxy diphenyl sulfone. They are the raw material of polysulfone and they also present toxicity. Toxicity can be evaluated from the inhibition degree of microorganisms. Sterilization is by moist-heat (121.1°C for 15 min) or ozone gas sterilization (3000 ppm at 90 % RH). Microorganisms are Geobacillus stearothermophilus ATCC 12980 as a representative spore and Micrococcus luteus. The sterilization result was cultivated in SCDA medium at 55°C for G. stearothermophilus ATCC 12980 and SCDA medium at 30°C for M.luteus and was scoured colony forming unit (CFU) in SCDA plate. G. stearothermophilus ATCC 12980 was around half sterilized and M.luteus. was totally sterilized by both sterillization methods. The produced amount of 4-chloro 4’-hydroxy diphenyl sulfone, Bisphenol S and Bisphenol A was 625 ppm, 355 ppm and 43 ppm. As the amount of bisphenol A was less than others, so bisphenol A may not be a major factor for inhibitors [5].
References
- Shintani H. (1995) The Relative Safety of Gamma-Ray, Autoclave, and Ethylene oxide Gas Sterilization of Thermosetting Polyurethane. Biomed. Instrument. Technol 29: 513-519.
- Shintani H. (2001) Determination of the Endocrine Disrupter Bisphenol-A in the Blood of Uremia Patients Treated by Dialysis. Chromatographia 53:331-333.
- Shintani H. (2000) Pretreatment and Chromatographic Analysis of Phthalate Esters, and their Biochemical Behavior in Blood products. Chromatographia 52:721-726.
- Shintani H. (1993) Solid Phase Extraction and HPLC Analysis of Toxic Components Eluted from Methyl Methacrylate Dental Materials. J. Anal.Toxicol 17:73-78.
- Shintani H. (2003) Determination of Compounds Inhibitors Bacterial Growth in Sterilized Medical Devices. Chromatographia 58:193-199.